|
|
THE VETERINARY SURGEONS AND VETERINARY PARA-PROFESSIONALS REGULATIONS
ARRANGEMENT OF REGULATIONS
PART I – PRELIMINARY
PART II – ELECTION OF BOARD MEMBERS, THE DISCIPLINARY COMMITTEE AND OTHER MATTERS
| 3. |
Procedure for election of Board members
|
| 5. |
Nomination of candidates
|
| 6. |
Registrar to confirm nominees
|
| 7. |
Election of Board members
|
| 10. |
Creation of Registers
|
| 11. |
Appointment of inspectors
|
| 12. |
Establishment of a disciplinary committee
|
| 13. |
Summons to appear at inquiries
|
PART III – TRAINING AND REGISTRATION
| 14. |
Registration of Specialist or Consultants
|
| 15. |
Establishment of veterinary para-professional cadres
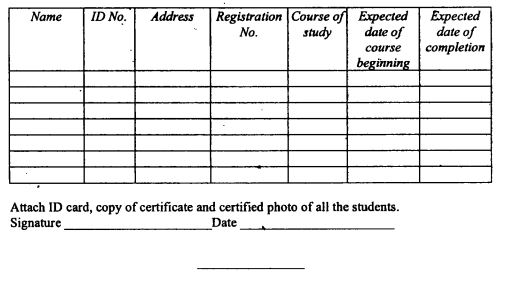
|
| 16. |
Temporary registration of a foreign veterinary surgeon
|
| 17. |
Continuous Professional Development
|
| 19. |
Fees for professional services
|
PART IV – REGISTRATION OF INSTITUTIONS AND ORGANISATIONS
| 20. |
Registration of institutions handling animals to safeguard their health and welfare
|
| 21. |
Approval for veterinary clinical centres, hospitals and ambulatory services
|
| 22. |
Approval for veterinary laboratory
|
PART V – STANDARDS
| 23. |
Standards for a veterinary clinical centre
|
| 24. |
Standards for a veterinary hospital
|
| 25. |
Standards for an ambulatory service
|
| 27. |
Standards of veterinary laboratory
|
| 28. |
Standards for advertisement of veterinary services
|
| 29. |
Animal breeding services
|
| 30. |
Standards for Training and curriculum for breeding services
|
| 31. |
Reporting of notifiable diseases
|
PART VI – TRAINING INSTITUTIONS AND CONDUCT OF EXAMINATIONS
| 32. |
Approval of training institution
|
| 33. |
Standards for institutions training for award of degree of veterinary medicine
|
| 34. |
Standards for institutions training for award of diploma in animal health
|
| 35. |
Standards for institutions training for the award of animal health certificates
|
| 37. |
Conduct of examinations in authorized institutions
|
| 38. |
Internship for graduates
|
SCHEDULES
| SECOND SCHEDULE [r.9(1)] — |
BOARD'S LOGO
|
THE VETERINARY SURGEONS AND VETERINARY PARAPROFESSIONALS REGULATIONS
PART I – PRELIMINARY
| 1. |
Citation
These Regulations may be cited as the Veterinary Surgeons and Veterinary Para-professionals Regulations, 2013.
|
| 2. |
Interpretation
In these Regulations, unless the context otherwise requires—
"Act" means the Veterinary Surgeons and Veterinary Para-professionals Act (No. 29 of 2011);
"advertisement" means the publication, through print or electronic means, of the following information to the public—
| (a) |
directions to a veterinary clinic;
|
| (b) |
name of the owner of a veterinary clinic;
|
| (c) |
description of services offered by a veterinary clinic;
|
| (d) |
qualifications of a practitioner;
|
"ambulatory facility" means a mobile veterinary clinical centre offering outpatient service from a contact point;
"animal health service" includes animal welfare service;
"animal hospital" means a veterinary clinical centre offering inpatient and outpatient service with facility for surgery, radiology, clinical pathology, dispensary, ward accommodation and provision for twenty four hour surveillance of patients;
"breeding service" means artificial insemination or fertilization, embryo transfer and genetic technologies in an animal;
"captive animal" means a domestic or wild animal confined within a space continuously or intermittently and includes an animal kept in a cage, zoo, wildlife sanctuary, an emerging livestock breeding centre or a farm;
"contact point" means a physical premise from which an ambulatory service is operated;
"continuous professional development" means a process or activity that provides added value to the capability of the professional through advancement in knowledge, skills and personal qualities necessary for the execution of professional and technical duties;
"credit point" means continuous professional development rating as determined by the Board;
"direction" with respect to internship, means instruction by a veterinary surgeon to an intern as to a procedure, test or treatment to be administered on an animal, but such veterinary surgeon is not necessarily physically present when the procedure test or treatment is being administered by the intern;
"faculty" means a division within a university comprising one subject area and includes schools and colleges;
"institution offering animal health services" means a non-governmental institution that provides services aimed at safeguarding animal health and welfare;
"internship" means a training period of not less than twelve months apprenticeship to present a graduate an opportunity to gain competence in veterinary practice;
"Registrar" means the chief executive officer appointed under section 12 of the Act;
"specialist college" means a group of veterinary surgeons who are experts in a sphere set out by the Board;
"supervision" with respect to internship, means instruction by a veterinary surgeon to an intern on a procedure, test or treatment to be administered and such veterinary surgeon is physically present when the procedure, test or treatment is being administered by the intern;
"teaching aid" includes an audio visual aid, computer, camera, liquid crystal display projector, television, monitor, video camera, video, digital versatile disk, specimen, model and skeleton;
"veterinary clinical centre" means an establishment that has facilities for outpatient examinations and treatment of animals and may include ambulatory services and boarding facilities.
"veterinary laboratory" means an institution where—
| (a) |
a specimen is received, processed and examined for diagnosis of animal diseases and for forensic purposes;
|
| (b) |
animal feedstuff, fertilizer, veterinary medicine, animal products, or animal production inputs are tested for quality and safety; or
|
| (c) |
veterinary research is conducted;
|
"veterinary student" means a student in an institution recognized by the Board offering veterinary medicine or animal health course.
|
PART II – ELECTION OF BOARD MEMBERS, THE DISCIPLINARY COMMITTEE AND OTHER MATTERS
| 3. |
Procedure for election of Board members
The members of the Board under section 4 (1) (f) and (g), shall be elected in accordance with the provisions of regulations 4, 5, 6, and 7 of these Regulations.
|
| 4. |
Eligibility of voters
| (1) |
A person registered as a veterinary surgeon under the Act shall be eligible to vote under section 4 (f) of the Act.
|
| (2) |
A person registered as a veterinary paraprofessional under the Act shall be eligible to vote under section 4 (g) of the Act.
|
|
| 5. |
Nomination of candidates
| (1) |
The Registrar shall, not less than twenty one days before the expiry of the term of the Board, publish a notice in two newspapers of national circulation, requiring the nomination of candidates for the election of Board members under section 4 of the Act.
|
| (2) |
The notice under sub-regulation (1) shall be—
| (a) |
in Form 1 set out in the First Schedule, for veterinary surgeons; and
|
| (b) |
in Form 2 set out in the First Schedule, for veterinary para-professionals.
|
|
| (3) |
The notice under sub-regulation (2) shall—
| (a) |
state the direction as to where a voter can obtain the nomination form; and
|
| (b) |
specify the date on which the nomination forms shall be submitted to the Registrar.
|
|
| (4) |
The nomination period shall, for the purposes of sub-regulation (1), be fourteen days from the date of publication.
|
| (5) |
Each voter shall be entitled to nominate as many candidates as the number of vacancies specified in sections 4 (f) and 4 (g) of the Act.
|
| (6) |
A separate form shall be used for the nomination of each candidate.
|
| (7) |
Each form under sub-regulation (6) shall be completed and signed by a nominee, a proposer and a seconder.
|
| (8) |
The nomination of a candidate shall be lodged with the Registrar before twelve noon on the nomination day specified.
|
|
| 6. |
Registrar to confirm nominees
| (1) |
The Registrar shall, on receipt of the nomination form, accept the nomination of a person as a candidate for an election, if he is satisfied that—
| (a) |
the nomination has been done in accordance with these Regulations; and
|
| (b) |
the nominated person is qualified in terms of the qualification criteria provided in section 4(3) of the Act.
|
|
| (2) |
If the Registrar refuses to accept the nomination of a person as a candidate for an election, he shall, within three days of receipt of the forms notify, in writing, the nominated person concerned and the persons by whom that person has been nominated, of the grounds of refusal.
|
| (3) |
A candidate whose application has been rejected by the Registrar may, within five days, lodge an appeal with the Board.
|
| (4) |
The Board shall hear and conclude the appeal within five days.
|
| (5) |
If the number of nominees is equal or less than the number of vacancies the nominees shall be declared elected.
|
| (6) |
Where the number of nominees is greater than the vacancies, the Registrar shall publish the names and particulars of the nominees in at least two newspapers of national circulation.
|
| (7) |
The publication under sub-regulation (6) shall specify the date and place for election of Board members.
|
|
| 7. |
Election of Board members
| (1) |
The ballot shall be as provided in—
| (a) |
Form 3 for veterinary surgeons; and
|
| (b) |
Form 4 for veterinary paraprofessionals.
|
|
| (2) |
The Registrar shall, on the day of issuance of notice of election, send the ballots to the voters by registered mail or electronic mail.
|
| (3) |
The elections shall be held twenty one days from the date of issuance of notice for election.
|
| (4) |
Every voter shall submit a marked ballot, either physically or electronically, to the Board or voting centres, on or before twelve noon of the twenty first day referred to in sub-regulation (3).
|
| (5) |
There shall be no voting by proxy.
|
| (6) |
The Board shall select a team to preside over elections headed by the Registrar who shall be the returning officer or his authorized nominee and any other persons as the Board may choose from persons who are not interested parties to the elections.
|
| (7) |
Counting of votes shall begin upon closure of voting and shall be completed within two days.
|
| (8) |
Each candidate for an election shall be entitled to have an observer in the counting hall during the counting of votes.
|
| (9) |
The Registrar shall publish the result of the count in at least two newspapers of national circulation.
|
|
| 8. |
Appeals
| (1) |
A candidate may challenge the result of an election by filing a petition with the Board within five days after the announcement of the result and which petition shall be accompanied by a fee of twenty thousand shillings.
|
| (2) |
The petition referred to in sub-regulation (l), shall be determined within seven days by the Board whose composition shall not include any member nominated for the election for which a petition has been filed.
|
|
| 9. |
Common Seal
| (1) |
The common seal of the Board is the Board's logo provided in the Second Schedule.
|
| (2) |
The common seal shall be attached to documents upon obtaining—
| (a) |
the authority of the Chairperson in writing; or
|
| (b) |
by a resolution of the Board.
|
|
|
| 10. |
Creation of Registers
The Board shall keep and maintain the following registers—
| (a) |
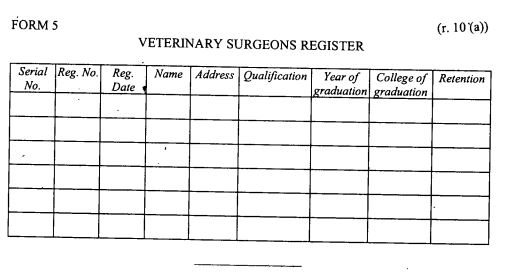
register of veterinary surgeons as provided in Form 5 set out in the First Schedule;
|
| (b) |
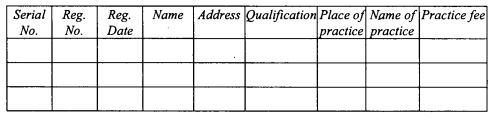
register of licensed veterinary surgeons as provided in Form 6 set out in the First Schedule;
|
| (c) |
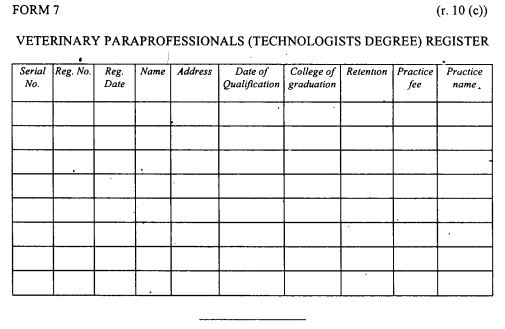
Register of veterinary para-professionals (veterinary technologists-degree holders) as provided in Form 7 set out in the First Schedule;
|
| (d) |
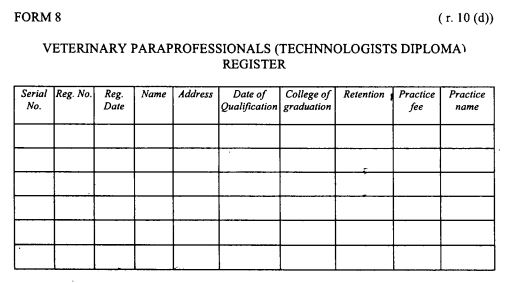
register of veterinary para-professionals (veterinary technologists-diploma holders) as provided in Form 8 set out in the First Schedule;
|
| (e) |
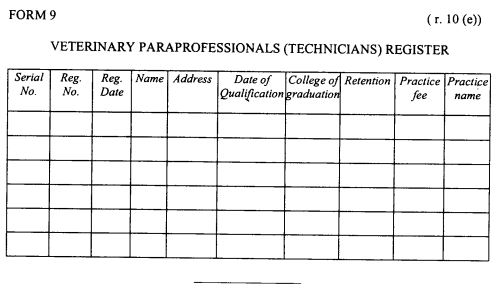
register of veterinary para-professionals (veterinary technicians) as provided in Form 9 set out in the First Schedule;
|
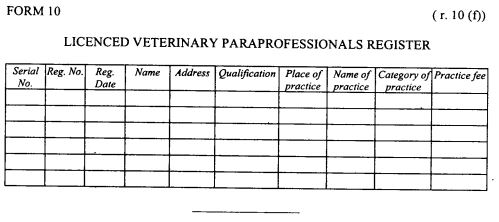
| (f) |
register of licensed veterinary para-professionals as provided in Form 10 set but in the First Schedule.
|
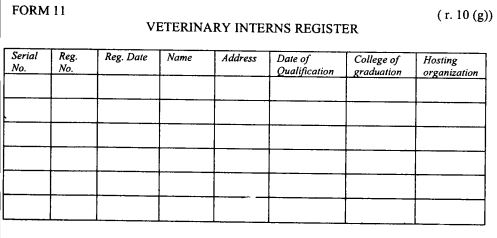
| (g) |
register of veterinary interns as provided in Form 11 set out in the First Schedule;
|
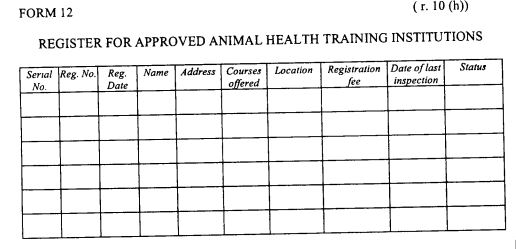
| (h) |
register of approved animal health training institutions as provided in Form 12 set out in the First Schedule;
|
| (i) |
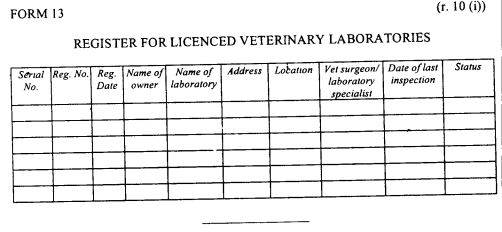
register of licensed veterinary laboratories as provided in Form 13 set out in the First Schedule;
|
| (j) |
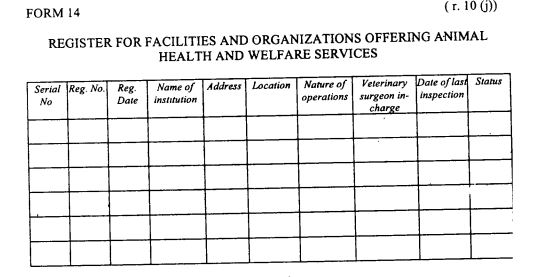
register of facilities and organizations offering animal health and welfare services as provided in Form 14 set out in the First Schedule;
|
| (k) |
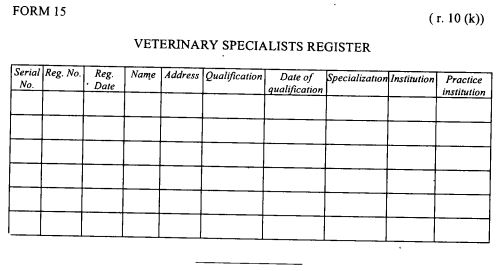
register of veterinary specialists as provided in Form 15 set out in the First Schedule;
|
| (l) |
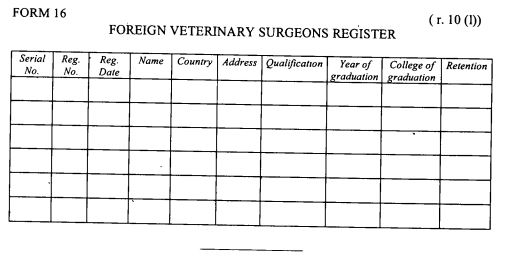
register of foreign veterinary surgeons as provided in Form 16 set out in the First Schedule;
|
| (m) |
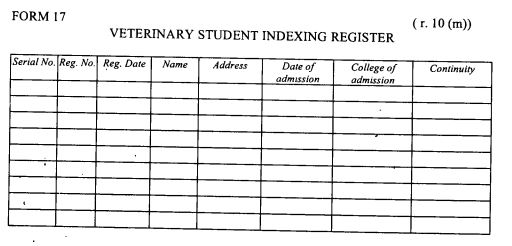
register of indexed veterinary students as provided in Form 17 set out in the First Schedule;
|
| (n) |
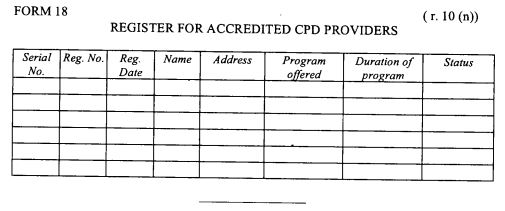
register of CPD providers as provided in Form 18 set out in the First Schedule;
|
| (o) |
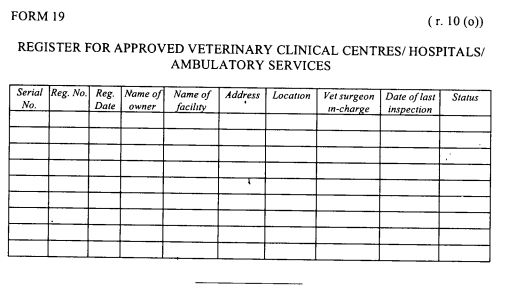
register of veterinary clinical centres, hospitals and ambulatory services as provided in Form 19 set out in the First Schedule; and
|
| (p) |
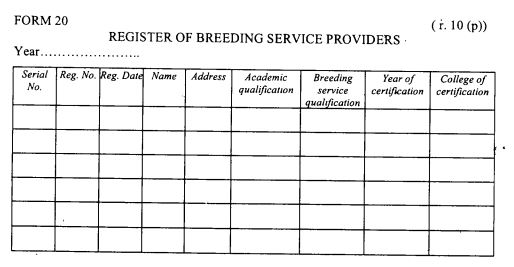
a register of breeding service providers as provided in Form 20 set out in the First Schedule.
|
|
| 11. |
Appointment of inspectors
| (1) |
There is established the Veterinary Inspectorate which shall be headed by a Chief Inspector and shall comprise a team of persons as the Board shall appoint.
|
| (2) |
The Board shall appoint qualified persons, whether by name or by virtue of their office to serve as inspectors for the purposes of this Act.
|
| (3) |
Without prejudice to the generality of sub-regulation (2), a person shall be qualified for appointment as an inspector if that person—
| (a) |
holds at least a degree in veterinary medicine or is qualified as a veterinary para-professional;
|
| (b) |
has at least ten years experience in the relevant field;
|
| (c) |
passes an integrity assessment; and
|
| (d) |
has undergone an appropriate course for inspectors offered by the Board.
|
|
| (4) |
The Board shall issue an inspector appointed under sub regulation (1) with an official photo identity card with inscription "Inspector of Kenya Veterinary Board" duly stamped and signed by the chief executive officer or his authorized agent.
|
| (5) |
Upon ceasing to be an inspector, a person shall return his identification card to the chief executive officer or his authorized agent.
|
| (6) |
An inspector shall have authority to conduct inspections for the purpose of—
| (a) |
confirming standards provided under these Regulations have been complied with before the Board approves an application;
|
| (b) |
safeguarding animal welfare by confirming the standards of any premises where animals are kept are being adhered to; and
|
| (c) |
enforcing the provisions of these Regulations.
|
|
| (7) |
An inspector appointed under these Regulations, for the purpose of ascertaining whether the provisions of the Act are being complied with, shall have the power—
| (a) |
to enter a veterinary clinic, veterinary laboratory or any other facility that has an animal health and welfare component to inspect any books, papers, records or writings, medicines and other inputs;
|
| (b) |
to enter any premises in which he has reasonable cause to suspect that a breach of the law has been or is being committed;
|
| (c) |
to make examination and inquiry; and
|
| (d) |
to do such other things, including the taking of samples, as may be necessary.
|
|
| (8) |
An inspector shall submit his report in the manner provided in Form 21 set out in the Second Schedule.
|
| (9) |
An inspector shall observe confidentiality in the findings of his inspection.
|
| (10) |
An inspector shall not be personally liable for performing his lawful duties provided under this Act.
|
| (11) |
Despite sub-regulation (10), an inspector shall be liable for any act of negligence committed in performance of his duties.
|
|
| 12. |
Establishment of a disciplinary committee
| (1) |
There is established a Disciplinary Committee of the Board which shall be a standing committee for the purposes of inquiries under this Act.
|
| (2) |
The Disciplinary Committee shall be responsible for making recommendation to the Board after conducting inquiry on disciplinary issues as provided under section 34 of the Act.
|
|
| 13. |
Summons to appear at inquiries
| (1) |
The Registrar may, on the direction of the Board, issue a written notice summoning a person who is the subject of an inquiry under the Act, to attend a disciplinary inquiry.
|
| (2) |
The notice under sub-regulation (1), shall be served by—
| (a) |
forwarding it by electronic mail or registered post to the person concerned;
|
| (b) |
delivering it to the person concerned in person or to his duly authorized representative;
|
| (c) |
delivering it at the residence or place of business of the person concerned to a person who is not less than eighteen years of age and resides or is employed by the person who is subject of the inquiry;
|
| (d) |
delivering it at the place of employment of the person concerned to a person who is not less than eighteen years of age and in authority over him or, in the absence of such person, to a person who is not less than eighteen years of age and is in charge at his place of employment;
|
| (e) |
delivering it, in the case of a legal person, at the registered office of the legal person; or
|
| (f) |
publishing it in a newspaper with wide national circulation.
|
|
|
PART III – TRAINING AND REGISTRATION
| 14. |
Registration of Specialist or Consultants
| (1) |
The Board shall register specialist colleges in accordance with such guidelines as it shall develop.
|
| (2) |
A veterinary surgeon may apply to the Board in Form 22 set out in the First Schedule for registration as a veterinary specialist in a college.
|
| (3) |
An applicant under sub-regulation (2) shall—
| (a) |
produce documentary evidence of the relevant expertise; and
|
|
| (4) |
The Board shall, following a recommendation from the specialist college, on approval of an application under sub-regulation (2), issue the applicant with a certificate of registration in Form 23 set out in the First Schedule.
|
| (5) |
A person registered by the Board as specialist or consultant shall use the titles with a client or colleague.
|
| (6) |
A person shall not hold himself or any other person as having expertise they cannot substantiate, or refer to himself or any other person as a ‘specialist’, where in doing so would be misleading or amount to misrepresentation.
|
|
| 15. |
Establishment of veterinary para-professional cadres
| (1) |
The following cadres of veterinary para-professionals are established—
| (a) |
veterinary technologist is a veterinary para-professional who has attained an academic diploma certificate in animal health or a degree in animal health, in a course of study as approved by the Board, but does not include a person with a degree in veterinary medicine;
|
| (b) |
veterinary technician is a veterinary para-professional who has attained an academic certificate in an animal health course of at least two years of study as approved by the Board.
|
|
| (2) |
Despite the provisions of sub-regulation 1(b), a person referred to in section 17(1)(b)(ii) of the Act, who has undergone a one year course in animal health as prescribed by the Board and has passed an exam set by the Board, shall be registered as a Veterinary Technician.
|
| (3) |
A person referred to in this Regulation shall be required to sit an exam set by the Board to qualify for registration under this Act.
|
|
| 16. |
Temporary registration of a foreign veterinary surgeon
| (1) |
A foreign person wishing to practice veterinary surgery in Kenya shall apply to the Board in accordance with Form 24 set out in the First Schedule.
|
| (2) |
The Board shall approve the application in accordance with the criteria set out under section 16 of the Act.
|
| (3) |
The Board shall, on approval of the application, issue the applicant with a temporary registration certificate in accordance with Form 25 set out in the First Schedule.
|
| (4) |
A foreign veterinary surgeon may practice if he is issued with an annual practicing license in Form 26 set out in the First Schedule.
|
|
| 17. |
Continuous Professional Development
| (1) |
Every veterinary surgeon and veterinary para-professional shall undertake continuous professional development activities in the manner provided by the Board.
|
| (2) |
A veterinary surgeon and a veterinary para-professional shall not be retained in the register until he has acquired at least twenty and fourteen credit points respectively in the year under consideration.
|
| (3) |
A veterinary surgeon or a veterinary para-professional may apply to the Board for a waiver of some or all the credit points and shall give reasons for the request.
|
| (4) |
The Board may, for valid reasons such as medical reasons, absence from the country or non-veterinary commitments, waive some or all the credit points for a registered person.
|
| (5) |
A veterinary surgeon and veterinary para-professional shall show evidence of the continuous professional development activities completed by thirty first December of every year in the manner provided in Form 27 set out in the First Schedule.
|
| (6) |
In addition to filling Form 27 a veterinary surgeon or veterinary para-professional shall submit—
| (a) |
a summary of continuous professional development activities attended, including the nature, scope and duration of such activities; and
|
| (b) |
a summary of credit points acquired.
|
|
| (7) |
The Board may discipline any veterinary surgeon and veterinary para-professional who has not acquired the minimum number of credit points by—
| (a) |
requiring the person to follow a remedial continuous professional development programme;
|
| (b) |
requiring the person to write an examination determined by the Board; or
|
| (c) |
removing the person's name from the relevant register.
|
|
|
| 18. |
Accreditation
| (1) |
A person wishing to be accredited as a continuous professional development service provider shall apply to the Board in the manner provided in Form 28 set out in the First Schedule.
|
| (2) |
The Board shall confirm accreditation of a continuous professional development service provider in the manner provided in Form 29 set out in the First Schedule.
|
| (3) |
Every continuous professional development provider shall apply for approval and determination of credit point worth by the Board, of the intended activity in the manner provided in Form 30 set out in the First Schedule.
|
|
| 19. |
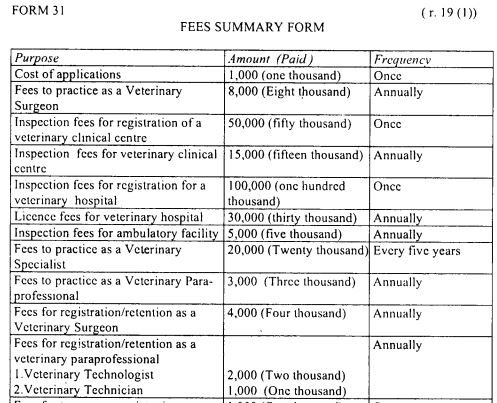
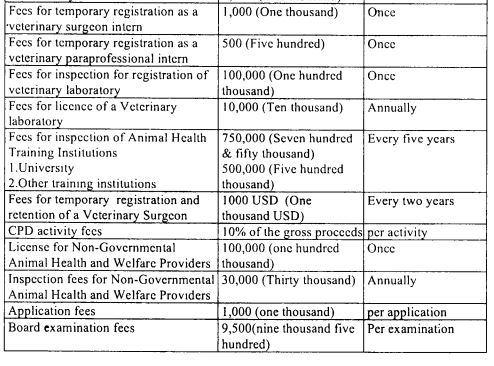
Fees for professional services
| (1) |
The Board shall from time to time determine in Form 31 set out in the First Schedule, the fees to be charged for the services it provides.
|
| (2) |
The Board shall, from time to time, set minimum fees for professional services offered by a person registered under the Act.
|
|
PART IV – REGISTRATION OF INSTITUTIONS AND ORGANISATIONS
| 20. |
Registration of institutions handling animals to safeguard their health and welfare
| (1) |
An institution intending to offer animal health services shall apply to the Board in Form 32 set out in the First Schedule and shall state—
| (a) |
the location and jurisdiction of the intended operation;
|
| (b) |
the type of service it intends to offer;
|
| (c) |
the name and registration status of the veterinary surgeon in the employment of the institution in charge of animal health matters; and
|
| (d) |
the facilities the institution has in order to offer the intended service.
|
|
| (2) |
Despite sub-regulation (1) (c), if the organization changes the veterinary surgeon, it shall notify the Board and shall suspend animal health services until it has recruited a veterinary surgeon in charge of the animal health services.
|
| (3) |
The Board may grant the institution an operation licence, with the necessary conditions, in accordance with Form 33 set out in the First Schedule.
|
|
| 21. |
Approval for veterinary clinical centres, hospitals and ambulatory services
| (1) |
A person intending to operate a veterinary clinical centre, veterinary hospital or an ambulatory service shall, before commencing such operation, apply to the Board for approval in accordance with Form 34 (a) set out in the First Schedule.
|
| (2) |
The Board shall on receipt of the application under sub-regulation (1), cause an inspection to be conducted of the veterinary clinical centre, veterinary hospital or an ambulatory service.
|
| (3) |
The Board shall, where the applicant complies with the standards set out in regulations 23, 24, or 25 and on payment of the requisite fee, approve the application and issue a licence in Form 34 (b) set out in the First Schedule.
|
| (4) |
A license issued under this Regulation shall be valid for a period of one year calculated from the date of issue.
|
| (5) |
Despite sub-regulation (4), a licence may be cancelled at any time when the Board establishes a breach of any provisions of these Regulations.
|
|
| 22. |
Approval for veterinary laboratory
| (1) |
A person shall apply to the Board for approval to operate a veterinary laboratory in the manner provided in Form 35 set out in the First schedule.
|
| (2) |
The Board shall inspect a veterinary laboratory for purposes of registration under sub-regulation (1).
|
| (3) |
The inspection under sub-regulation (2) shall be done once.
|
| (4) |
An applicant under this Regulation shall specify the laboratory services he intends to offer in the veterinary laboratory.
|
| (5) |
The Board shall inspect the veterinary laboratory to confirm that it satisfies the standards under regulation 27 and on approval issue an annual license in Form 36 set out in the First schedule.
|
| (6) |
A license may be cancelled by the Board any time an inspector establishes a breach of any provisions of these Regulations.
|
| (7) |
A veterinary clinical centre, veterinary hospital or an ambulatory service that has been licensed to operate under this Act shall not be required to make a separate application for its own in-house laboratory.
|
|
PART V – STANDARDS
| 23. |
Standards for a veterinary clinical centre
A veterinary centre shall satisfy the following minimum standards—
| (a) |
cleanliness, impervious walls and paved floors which are easy to clean and disinfect;
|
| (b) |
sufficient natural light supported by adequate artificial lighting;
|
| (c) |
clean protective equipment;
|
| (d) |
clean well groomed personnel;
|
| (e) |
facility for containment of micro-organisms and adequate protection against dissemination of diseases to personnel and animals;
|
| (g) |
facility for proper disposal of all waste materials such as incinerators or secure soak pits;
|
| (h) |
provision for proper disposal of unclaimed dead patients, on or off site;
|
| (i) |
sufficient ventilation and freedom from noxious odours;
|
| (j) |
sufficient species-specific restraining facilities;
|
| (k) |
sufficient refrigeration and storage facilities;
|
| (l) |
basic diagnostic and medical equipment including, thermometers, stethoscope, microscopes, surgical kit, lockable medicine kit, sampling tools and equipment;
|
| (m) |
implementation of Occupational Health and Safety Act (No. 15 of 2007) in the work place;
|
| (n) |
| (ii) |
diseases encountered; |
| (iv) |
inventory and stores; |
| (v) |
veterinary certificates which shall include the full name and signature of the attending veterinary surgeon, Kenya Veterinary Board registration number, the address. the full identification of the patient in terms of the name, species, sex, colour, age and any identification device number on the patient, date of vaccination or examination, and the generic and trade name of the vaccine used, batch number and the expiry date and manufacturer; and |
| (vi) |
any other record determined by the Board. |
|
|
| 24. |
Standards for a veterinary hospital
| (1) |
An animal hospital shall satisfy the minimum standards specified in regulation 23.
|
| (2) |
In addition to the provisions of regulation 23, an animal hospital shall have sufficient facilities including;
| (a) |
comfortable and safe accommodation with walls and floors that are easy to clean and disinfect with adequate bedding;
|
| (b) |
wholesome feeds and sufficient potable water;
|
| (c) |
diagnostic facilities including imaging and laboratory services;
|
| (d) |
at least one resident veterinary surgeon and one veterinary para-professional on duty at any given time; and
|
| (e) |
at least two well-equipped veterinary surgical theatres, a preparation room and a separate recovery room.
|
|
| (3) |
An animal hospital shall have adequate staff establishment, which shall comprise of at least three veterinary surgeons.
|
|
| 25. |
Standards for an ambulatory service
An ambulatory service shall have the following minimum standards—
| (a) |
sufficient means of transport;
|
| (b) |
clean protective equipment;
|
| (c) |
clean, well-groomed personnel;
|
| (d) |
a facility for containment of micro-organisms and adequate protection against dissemination of diseases to personnel and animals;
|
| (e) |
access to proper disposal of all waste materials such as incinerators or secure soak pits;
|
| (f) |
sufficient portable species-specific restraining facilities;
|
| (g) |
a contact point with adequate refrigeration and storage facilities for veterinary medicines, equipment and records; and
|
| (h) |
sampling tools and equipment.
|
|
| 26. |
Emergency service
A veterinary clinical centre, animal hospital or ambulatory facility shall offer twenty four hour emergency service.
|
| 27. |
Standards of veterinary laboratory
| (1) |
A veterinary laboratory shall—
| (a) |
be located away from known fire hazards;
|
| (b) |
be separated from non-related activities;
|
| (c) |
have restricted access by personnel: and
|
| (d) |
be vermin-proofed for pests including insects and rodents.
|
|
| (2) |
In addition to sub-regulation (1), a veterinary laboratory shall have the following minimum standards—
| (a) |
the floors and the walls of the building shall be constructed from materials that are easy to clean, impervious and resistant to corrosion by chemicals;
|
| (b) |
have access to an incinerator, pressure steam sterilizer or renderer for disposal of carcasses and waste;
|
| (c) |
provide a microbiological safety cabinet able to protect the operator, the product and the environment in laboratories dealing with microbial cultures;
|
| (d) |
sample collection and diagnostic equipment and facilities;
|
| (e) |
provide an autoclave or pressure steam sterilizer;
|
| (f) |
provide personnel with protective clothing and equipment for use only within the laboratory;
|
| (g) |
have a designated veterinary surgeon responsible for containment;
|
| (h) |
competent staff in the area of specialization;
|
| (i) |
provide warning notices for containment area indicating the type of hazard, the name and telephone number of the person responsible;
|
| (j) |
have laboratory alarm system, emergency lighting, fire fighting equipment and first aid kit;
|
| (k) |
post emergency protocols within the laboratory advising personnel on procedures to follow in case of emergencies: and
|
| (l) |
display standard operating procedures for the laboratory.
|
|
|
| 28. |
Standards for advertisement of veterinary services
| (1) |
An advertisement for services by a veterinary surgeon, a veterinary paraprofessional or a veterinary practice shall—
| (a) |
conform to the veterinary code of ethics:
|
| (b) |
comprise information that enriches the clients’ knowledge in animal health care;
|
| (d) |
be scientifically factual; and
|
| (e) |
not be an endorsement of a veterinary surgeon, veterinary para-professional, veterinary product or service.
|
|
| (2) |
A veterinary surgeon or veterinary para-professional shall display at the place at which he practices a sign bearing—
| (a) |
the name of the veterinary surgeon or veterinary para-professional;
|
| (b) |
the qualification as recorded in the register or veterinary surgeons, veterinary para-professionals or specialists;
|
| (c) |
the name, professional details and duration of visit of any foreign veterinary surgeon; and
|
|
| (3) |
A sign displayed at a veterinary premises may be illuminated by a light of constant intensity without causing an irritation to the general public.
|
| (4) |
A veterinary surgeon or veterinary para-professional whose premises are in a shopping complex or other place where a directory of tenants is provided may display on that directory a sign indicating that the veterinary surgeon or veterinary para-professional has a practice in that place.
|
| (5) |
An advertisement shall not contain any non-academic nominals.
|
| (6) |
Advertising in any telephone book, directory, website or other place or medium shall be accurate and shall comply with the standards set out in the Act.
|
|
| 29. |
Animal breeding services
| (1) |
A veterinary para-professional who has completed an artificial insemination course at an institution approved by the Board may apply for registration as a breeding service provider.
|
| (2) |
A person shall offer veterinary breeding services if he is registered under this Act.
|
|
| 30. |
Standards for Training and curriculum for breeding services
| (1) |
A training institution shall not offer training in breeding services unless the institution is licensed by the Board in accordance with Form 38 (b) set out in the First Schedule.
|
| (2) |
An institution training in breeding services shall have facilities approved by the Board and qualified personnel.
|
| (3) |
A trainee in breeding services shall have a minimum qualification of a veterinary para-professional.
|
| (4) |
A person shall not offer training in breeding services unless he uses the curriculum approved by the Board.
|
| (5) |
A breeding service provider for all species, other than cattle, shall have the physical address and facilities approved by the Board.
|
|
| 31. |
Reporting of notifiable diseases
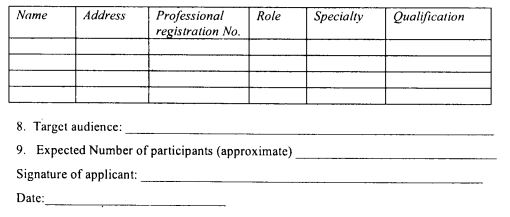
A notifiable disease shall be reported in the manner provided in Form 37 set out in the First Schedule.
|
PART VI – TRAINING INSTITUTIONS AND CONDUCT OF EXAMINATIONS
| 32. |
Approval of training institution
| (1) |
An animal health course or a veterinary medicine course shall be offered in a registered institution or in a faculty of veterinary medicine.
|
| (2) |
An institution desiring to offer courses of training leading to the award of animal health degree, diploma or certificate, or degree of Bachelor of Veterinary Medicine shall after being registered by the relevant body, apply to the Board for approval by submitting an application form in accordance with Form 38(a) set out in the First Schedule.
|
| (3) |
A person or institution shall not offer training in Animal Health or Veterinary Medicine unless they are registered by the Board.
|
| (4) |
An institution or person shall offer training courses in Animal Health or Veterinary Medicine using the curriculum approved by the Board.
|
| (5) |
The Board shall, on receipt of the application under sub-regulation (2), inspect the institution and if satisfied that it conforms to standards specified under these Regulations and upon payment of the requisite fees, approve the institution for the courses proposed.
|
| (6) |
The Board shall inspect approved training institutions from time to time.
|
| (7) |
The Board shall, in every five years, inspect institutions for purposes of approval.
|
|
| 33. |
Standards for institutions training for award of degree of veterinary medicine
| (1) |
A training institution desiring to offer courses for the award of degree of veterinary medicine shall have adequate physical facilities and equipment that meet the following minimum standards—
| (a) |
a lecture theatre for every class;
|
| (b) |
a laboratory for each major discipline;
|
| (c) |
seminar rooms and other teaching spaces which shall be adequate in number and size for the instructional purposes intended and the number of students enrolled;
|
| (d) |
administrative and faculty offices;
|
| (e) |
a teaching animal hospital with the minimum standards set out in regulation 24 and which has adequate teaching laboratory, research and clinical equipment for examination and diagnostics of all animals used by the institution;
|
| (f) |
an animal farm with the full spectrum of animal species to give the students adequate exposure in a ratio of at least two animals per student-admission for cattle, sheep, goat and pigs and one flock of two hundred poultry for ten student-admission and at least one fish farm and one rabbit farm;
|
| (g) |
a veterinary library; and
|
| (h) |
access to facilities offering food and accommodation for students in the vicinity.
|
|
| (2) |
The facilities and equipment specified in sub-regulation (1) shall be maintained clean and in good repair.
|
| (3) |
The number, qualification and terms of service for teaching and support staff shall conform to the minimum standards prescribed by the body responsible for higher education.
|
| (4) |
Without prejudice to sub-regulation (1), .an institution shall meet the following additional requirements—
| (a) |
the lecturers for animal health courses shall be veterinary surgeons;
|
| (b) |
the lecturers shall possess relevant qualifications for the subjects they teach;
|
| (c) |
the head of the faculty or school shall be a veterinary surgeon;
|
| (d) |
the head of veterinary academic section shall be a veterinary surgeon;
|
| (e) |
the ratio of permanently employed to part-time lecturers teaching the animal health programmes shall not be less than 3: 1; and
|
| (f) |
the staff establishment shall include sufficient numbers of technologists, technicians and other relevant staff.
|
|
| (5) |
In addition to the standards and equipment provided in regulation 24, the institutional surgical theatres and laboratory shall have the following additional equipments—
| (a) |
computed tomography scan;
|
| (b) |
magnetic resonance imaging;
|
| (c) |
fluoroscopy for cardiac catheterization and diagnostic imaging;
|
| (e) |
cardiopulmonary bypass equipment;
|
| (h) |
hemodynamic data services and software;
|
| (j) |
histopathology equipment;
|
| (m) |
light microscopes for each student;
|
| (o) |
ultrasound equipment; and
|
|
|
| 34. |
Standards for institutions training for award of diploma in animal health
| (1) |
A training institution desiring to offer courses tor the award of diploma in animal health shall have adequate physical facilities and equipment maintained clean and in good state of repair and shall meet the following minimum standards—
| (a) |
a lecture theatre for every class;
|
| (b) |
a laboratory for each major discipline;
|
| (d) |
administrative and staff offices;
|
| (e) |
an animal farm with the full spectrum of animal species to give the students adequate exposure in a ratio of at least two animals per student-admission for cattle, sheep, goat and pigs and one flock of two hundred poultry for ten student-admission and at least one fish farm and one rabbit farm;
|
| (f) |
a veterinary library; and
|
| (g) |
access to facilities offering food and accommodation for students in the vicinity.
|
|
| (2) |
The number, qualification and terms of service for teaching and support staff shall conform to the minimum standards prescribed by the body responsible for tertiary and higher education.
|
| (3) |
Without prejudice to sub-regulation (1), the institution shall meet the following additional requirements—
| (a) |
lecturers for animal health courses shall be veterinary surgeons;
|
| (b) |
lecturers shall possess relevant qualifications for the subjects they teach;
|
| (c) |
the head of the relevant section or department offering animal health course shall be a veterinary surgeon;
|
| (d) |
the ratio of permanently employed to part-time lecturers teaching the animal health programmes shall not be less than 3:1;
|
| (e) |
the staff establishment shall include sufficient numbers of technologists, technicians and other relevant staff;
|
| (f) |
minimum teaching aids shall be availed.
|
|
| (4) |
The standards of the laboratory and equipment in the institution shall conform to the standards in section 29 of the Act.
|
|
| 35. |
Standards for institutions training for the award of animal health certificates
| (1) |
A training institution desiring to offer courses for the award of certificate in animal health shall have adequate physical facilities and equipment maintained clean and in good state of repair and shall meet the following minimum standards—
| (a) |
a lecture theatre for every class;
|
| (b) |
two laboratories, one for basic sciences and one for applied sciences;
|
| (d) |
administrative and staff offices;
|
| (e) |
an animal farm with the full spectrum of animal species to give the students adequate exposure in a ratio of at least two animals per student-admission for cattle, sheep, goat and pigs and one flock of two hundred poultry for ten student-admission and at least one fish farm and one rabbit farm;
|
| (f) |
a veterinary library; and
|
| (g) |
food and accommodation for students.
|
|
| (2) |
The number, qualification and terms of service for teaching and support staff shall conform to the minimum standards prescribed v the body responsible for tertiary and higher education.
|
| (3) |
Without prejudice to sub-regulation (2), the institution shall set the following additional requirements—
| (a) |
lecturers for animal health courses shall be veterinary surgeons;
|
| (b) |
lecturers shall possess relevant qualifications for the subjects they teach;
|
| (c) |
the head of the relevant section or department offering animal health course shall be a veterinary surgeon;
|
| (d) |
the ratio of permanently employed to part-time lecturers teaching the animal health programmes shall not be less than 3: 1;
|
| (e) |
the staff establishment shall include sufficient numbers of technologists, technicians and other relevant staff; and
|
| (f) |
minimum number and types of teaching aids shall be availed.
|
|
| (4) |
The standards of the laboratory and equipment in the institution shall conform to the standards in section 29 of the Act.
|
|
| 36. |
Indexing of students
| (1) |
Every veterinary medicine or animal health student shall be indexed by the Board.
|
| (2) |
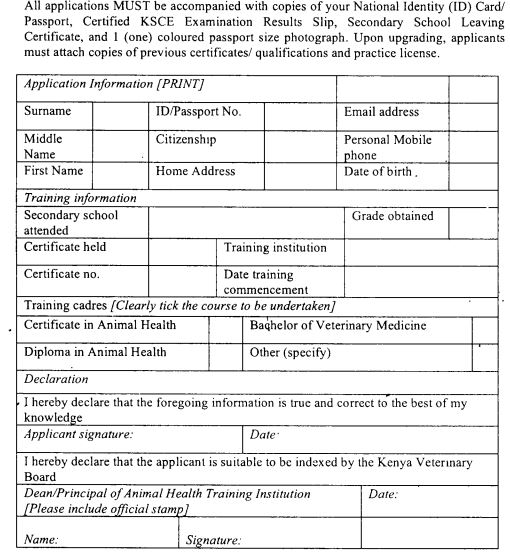
A student shall apply for indexing to the Board in Form 39(a) set out in the First Schedule.
|
| (3) |
A student shall submit the application to the institution where he is admitted for training.
|
| (4) |
An institution training students shall forward applications submitted under sub regulation (3), to the Board on behalf of the students.
|
| (5) |
An institution under sub-regulation (4), shall summarize the particulars of all the applications in Form 39 (b) set out in the First Schedule and shall submit the summary, together with the students' applications, to the Board.
|
|
| 37. |
Conduct of examinations in authorized institutions
| (1) |
The examinations for a veterinary medicine degree or animal health courses shall include examinations in all the subjects as specified by the Board.
|
| (2) |
At least two examiners, one of whom was not involved in the teaching of the subject to the candidate, shall participate in the evaluation of each subject examined.
|
| (3) |
A candidate for an examination for veterinary medicine degree or an animal health course shall pass an examination in a subject if he attains at least fifty per cent or the equivalent of fifty per cent of the maximum marks obtainable in the examination, in the marking system employed by the institution.
|
|
| 38. |
Internship for graduates
| (1) |
A veterinary medicine or animal health graduate shall apply to the Board for temporary registration for the period of internship in the manner provided in Form 40 set out in the First Schedule.
|
| (2) |
The Board shall, after payment of requisite fee, grant an applicant under sub-regulation (1) intern registration and issue him with a certificate in accordance with Form 41 set out in the First Schedule.
|
| (3) |
A veterinary medicine intern may examine animals, carry out diagnostic tests and administer treatment under the direction of a veterinary surgeon and perform surgical operation under the supervision of a veterinary surgeon.
|
| (4) |
A veterinary para-professional intern shall perform tasks and activities equal with his training under the direction of a veterinary surgeon.
|
| (5) |
A veterinary para-professional intern may examine animals and carry out diagnostic tests and administer treatment under the direction of a veterinary surgeon.
|
| (6) |
Upon successful completion of internship, the supervisor shall make recommendations to the Board for evaluation and registration of the intern in Form 42 set out in the First Schedule.
|
| (7) |
An intern or supervisor may refer any complaint during the internship to the Board in writing.
|
|
THE VETERINARY SURGEONS AND VETERINARY PARA-PROFESSIONALS REGULATIONS
PART I – PRELIMINARY
| 1. |
Citation
These Regulations may be cited as the Veterinary Surgeons and Veterinary Para-Professionals Regulations.
|
| 2. |
Interpretation
In these Regulations, unless the context otherwise requires—
"Act" means the Veterinary Surgeons and Veterinary Para-Professionals Act (Cap. 366);
"advertisement" means the publication, through print or electronic means, of the following information to the public—
| (a) |
directions to a veterinary clinic;
|
| (b) |
name of the owner of a veterinary clinic;
|
| (c) |
description of services offered by a veterinary clinic;
|
| (d) |
qualifications of a practitioner;
|
"ambulatory facility" means a mobile veterinary clinical centre offering outpatient service from a contact point;
"animal health service" includes animal welfare service;
"animal hospital" means a veterinary clinical centre offering inpatient and outpatient service with facility for surgery, radiology, clinical pathology, dispensary, ward accommodation and provision for twenty four hour surveillance of patients;
"breeding service" means artificial insemination or fertilization, embryo transfer and genetic technologies in an animal;
"captive animal" means a domestic or wild animal confined within a space continuously or intermittently and includes an animal kept in a cage, zoo, wildlife sanctuary, an emerging livestock breeding centre or a farm;
"contact point" means a physical premise from which an ambulatory service is operated;
"continuous professional development" means a process or activity that provides added value to the capability of the professional through advancement in knowledge, skills and personal qualities necessary for the execution of professional and technical duties;
"credit point" means continuous professional development rating as determined by the Board;
"direction" with respect to internship, means instruction by a veterinary surgeon to an intern as to a procedure, test or treatment to be administered on an animal, but such veterinary surgeon is not necessarily physically present when the procedure test or treatment is being administered by the intern;
"faculty" means a division within a university comprising one subject area and includes schools and colleges;
"institution offering animal health services" means a non-governmental institution that provides services aimed at safeguarding animal health and welfare;
"internship" means a training period of not less than twelve months apprenticeship to present a graduate an opportunity to gain competence in veterinary practice;
"Registrar" means the chief executive officer appointed under section 12 of the Act;
"specialist college" means a group of veterinary surgeons who are experts in a sphere set out by the Board;
"supervision" with respect to internship, means instruction by a veterinary surgeon to an intern on a procedure, test or treatment to be administered and such veterinary surgeon is physically present when the procedure, test or treatment is being administered by the intern;
"teaching aid" includes an audio visual aid, computer, camera, liquid crystal display projector, television, monitor, video camera, video, digital versatile disk, specimen, model and skeleton;
"veterinary clinical centre" means an establishment that has facilities for outpatient examinations and treatment of animals and may include ambulatory services and boarding facilities;
"veterinary laboratory" means an institution where—
| (a) |
a specimen is received, processed and examined for diagnosis of animal diseases and for forensic purposes;
|
| (b) |
animal feedstuff, fertilizer, veterinary medicine, animal products, or animal production inputs are tested for quality and safety; or
|
| (c) |
veterinary research is conducted;
|
"veterinary student" means a student in an institution recognized by the Board offering veterinary medicine or animal health course.
|
PART II – ELECTION OF BOARD MEMBERS, THE DISCIPLINARY COMMITTEE AND OTHER MATTERS
| 3. |
Procedure for election of Board members
The members of the Board under section 4 (1) (f) and (g), shall be elected in accordance with the provisions of regulations 4, 5, 6, and 7 of these Regulations.
|
| 4. |
Eligibility of voters
| (1) |
A person registered as a veterinary surgeon under the Act shall be eligible to vote under section 4 (f) of the Act.
|
| (2) |
A person registered as a veterinary para-professional under the Act shall be eligible to vote under section 4 (g) of the Act.
|
|
| 5. |
Nomination of candidates
| (1) |
The Registrar shall, not less than twenty one days before the expiry of the term of the Board, publish a notice in two newspapers of national circulation, requiring the nomination of candidates for the election of Board members under section 4 of the Act.
|
| (2) |
The notice under subregulation (1) shall be—
| (a) |
in Form 1 set out in the First Schedule, for veterinary surgeons; and
|
| (b) |
in Form 2 set out in the First Schedule, for veterinary para-professionals.
|
|
| (3) |
The notice under subregulation (2) shall—
| (a) |
state the direction as to where a voter can obtain the nomination form; and
|
| (b) |
specify the date on which the nomination forms shall be submitted to the Registrar.
|
|
| (4) |
The nomination period shall, for the purposes of subregulation (1), be fourteen days from the date of publication.
|
| (5) |
Each voter shall be entitled to nominate as many candidates as the number of vacancies specified in sections 4 (f) and 4 (g) of the Act.
|
| (6) |
A separate form shall be used for the nomination of each candidate.
|
| (7) |
Each form under subregulation (6) shall be completed and signed by a nominee, a proposer and a seconder.
|
| (8) |
The nomination of a candidate shall be lodged with the Registrar before twelve noon on the nomination day specified.
|
|
| 6. |
Registrar to confirm nominees
| (1) |
The Registrar shall, on receipt of the nomination form, accept the nomination of a person as a candidate for an election, if he is satisfied that—
| (a) |
the nomination has been done in accordance with these Regulations; and
|
| (b) |
the nominated person is qualified in terms of the qualification criteria provided in section 4(3) of the Act.
|
|
| (2) |
If the Registrar refuses to accept the nomination of a person as a candidate for an election, he shall, within three days of receipt of the forms notify, in writing, the nominated person concerned and the persons by whom that person has been nominated, of the grounds of refusal.
|
| (3) |
A candidate whose application has been rejected by the Registrar may, within five days, lodge an appeal with the Board.
|
| (4) |
The Board shall hear and conclude the appeal within five days.
|
| (5) |
If the number of nominees is equal or less than the number of vacancies the nominees shall be declared elected.
|
| (6) |
Where the number of nominees is greater than the vacancies, the Registrar shall publish the names and particulars of the nominees in at least two newspapers of national circulation.
|
| (7) |
The publication under subregulation (6) shall specify the date and place for election of Board members.
|
|
| 7. |
Election of Board members
| (1) |
The ballot shall be as provided in—
| (a) |
Form 3 for veterinary surgeons; and
|
| (b) |
Form 4 for veterinary paraprofessionals.
|
|
| (2) |
The Registrar shall, on the day of issuance of notice of election, send the ballots to the voters by registered mail or electronic mail.
|
| (3) |
The elections shall be held twenty one days from the date of issuance of notice for election.
|
| (4) |
Every voter shall submit a marked ballot, either physically or electronically, to the Board or voting centres, on or before twelve noon of the twenty first day referred to in subregulation (3).
|
| (5) |
There shall be no voting by proxy.
|
| (6) |
The Board shall select a team to preside over elections headed by the Registrar who shall be the returning officer or his authorized nominee and any other persons as the Board may choose from persons who are not interested parties to the elections.
|
| (7) |
Counting of votes shall begin upon closure of voting and shall be completed within two days.
|
| (8) |
Each candidate for an election shall be entitled to have an observer in the counting hall during the counting of votes.
|
| (9) |
The Registrar shall publish the result of the count in at least two newspapers of national circulation.
|
|
| 8. |
Appeals
| (1) |
A candidate may challenge the result of an election by filing a petition with the Board within five days after the announcement of the result and which petition shall be accompanied by a fee of twenty thousand shillings.
|
| (2) |
The petition referred to in subregulation (1), shall be determined within seven days by the Board whose composition shall not include any member nominated for the election for which a petition has been filed.
|
|
| 9. |
Common Seal
| (1) |
The common seal of the Board is the Board's logo provided in the Second Schedule.
|
| (2) |
The common seal shall be attached to documents upon obtaining—
| (a) |
the authority of the Chairperson in writing; or
|
| (b) |
by a resolution of the Board.
|
|
|
| 10. |
Creation of Registers
The Board shall keep and maintain the following registers—
| (a) |
register of veterinary surgeons as provided in Form 5 set out in the First Schedule;
|
| (b) |
register of licensed veterinary surgeons as provided in Form 6 set out in the First Schedule;
|
| (c) |
Register of veterinary para-professionals (veterinary technologists-degree holders) as provided in Form 7 set out in the First Schedule;
|
| (d) |
register of veterinary para-professionals (veterinary technologists-diploma holders) as provided in Form 8 set out in the First Schedule;
|
| (e) |
register of veterinary para-professionals (veterinary technicians) as provided in Form 9 set out in the First Schedule;
|
| (f) |
register of licensed veterinary para-professionals as provided in Form 10 set but in the First Schedule.
|
| (g) |
register of veterinary interns as provided in Form 11 set out in the First Schedule;
|
| (h) |
register of approved animal health training institutions as provided in Form 12 set out in the First Schedule;
|
| (i) |
register of licensed veterinary laboratories as provided in Form 13 set out in the First Schedule;
|
| (j) |
register of facilities and organizations offering animal health and welfare services as provided in Form 14 set out in the First Schedule;
|
| (k) |
register of veterinary specialists as provided in Form 15 set out in the First Schedule;
|
| (l) |
register of foreign veterinary surgeons as provided in Form 16 set out in the First Schedule;
|
| (m) |
register of indexed veterinary students as provided in Form 17 set out in the First Schedule;
|
| (n) |
register of CPD providers as provided in Form 18 set out in the First Schedule;
|
| (o) |
register of veterinary clinical centres, hospitals and ambulatory services as provided in Form 19 set out in the First Schedule; and
|
| (p) |
a register of breeding service providers as provided in Form 20 set out in the First Schedule.
|
|
| 11. |
Appointment of inspectors
| (1) |
There is established the Veterinary Inspectorate which shall be headed by a Chief Inspector and shall comprise a team of persons as the Board shall appoint.
|
| (2) |
The Board shall appoint qualified persons, whether by name or by virtue of their office to serve as inspectors for the purposes of this Act.
|
| (3) |
Without prejudice to the generality of subregulation (2), a person shall be qualified for appointment as an inspector if that person—
| (a) |
holds at least a degree in veterinary medicine or is qualified as a veterinary para-professional;
|
| (b) |
has at least ten years experience in the relevant field;
|
| (c) |
passes an integrity assessment; and
|
| (d) |
has undergone an appropriate course for inspectors offered by the Board.
|
|
| (4) |
The Board shall issue an inspector appointed under subregulation (1) with an official photo identity card with inscription "Inspector of Kenya Veterinary Board" duly stamped and signed by the chief executive officer or his authorized agent.
|
| (5) |
Upon ceasing to be an inspector, a person shall return his identification card to the chief executive officer or his authorized agent.
|
| (6) |
An inspector shall have authority to conduct inspections for the purpose of—
| (a) |
confirming standards provided under these Regulations have been complied with before the Board approves an application;
|
| (b) |
safeguarding animal welfare by confirming the standards of any premises where animals are kept are being adhered to; and
|
| (c) |
enforcing the provisions of these Regulations.
|
|
| (7) |
An inspector appointed under these Regulations, for the purpose of ascertaining whether the provisions of the Act are being complied with, shall have the power—
| (a) |
to enter a veterinary clinic, veterinary laboratory or any other facility that has an animal health and welfare component to inspect any books, papers, records or writings, medicines and other inputs;
|
| (b) |
to enter any premises in which he has reasonable cause to suspect that a breach of the law has been or is being committed;
|
| (c) |
to make examination and inquiry; and
|
| (d) |
to do such other things, including the taking of samples, as may be necessary.
|
|
| (8) |
An inspector shall submit his report in the manner provided in Form 21 set out in the Second Schedule.
|
| (9) |
An inspector shall observe confidentiality in the findings of his inspection.
|
| (10) |
An inspector shall not be personally liable for performing his lawful duties provided under this Act.
|
| (11) |
Despite subregulation (10), an inspector shall be liable for any act of negligence committed in performance of his duties.
|
|
| 12. |
Establishment of a disciplinary committee
| (1) |
There is established a Disciplinary Committee of the Board which shall be a standing committee for the purposes of inquiries under this Act.
|
| (2) |
The Disciplinary Committee shall be responsible for making recommendation to the Board after conducting inquiry on disciplinary issues as provided under section 34 of the Act.
|
|
| 13. |
Summons to appear at inquiries
| (1) |
The Registrar may, on the direction of the Board, issue a written notice summoning a person who is the subject of an inquiry under the Act, to attend a disciplinary inquiry.
|
| (2) |
The notice under subregulation (1), shall be served by—
| (a) |
forwarding it by electronic mail or registered post to the person concerned;
|
| (b) |
delivering it to the person concerned in person or to his duly authorized representative;
|
| (c) |
delivering it at the residence or place of business of the person concerned to a person who is not less than eighteen years of age and resides or is employed by the person who is subject of the inquiry;
|
| (d) |
delivering it at the place of employment of the person concerned to a person who is not less than eighteen years of age and in authority over him or, in the absence of such person, to a person who is not less than eighteen years of age and is in charge at his place of employment;
|
| (e) |
delivering it, in the case of a legal person, at the registered office of the legal person; or
|
| (f) |
publishing it in a newspaper with wide national circulation.
|
|
|
PART III – TRAINING AND REGISTRATION
| 14. |
Registration of Specialist or Consultants
| (1) |
The Board shall register specialist colleges in accordance with such guidelines as it shall develop.
|
| (2) |
A veterinary surgeon may apply to the Board in Form 22 set out in the First Schedule for registration as a veterinary specialist in a college.
|
| (3) |
An applicant under subregulation (2) shall—
| (a) |
produce documentary evidence of the relevant expertise; and
|
|
| (4) |
The Board shall, following a recommendation from the specialist college, on approval of an application under subregulation (2), issue the applicant with a certificate of registration in Form 23 set out in the First Schedule.
|
| (5) |
A person registered by the Board as specialist or consultant shall use the titles with a client or colleague.
|
| (6) |
A person shall not hold himself or any other person as having expertise they cannot substantiate, or refer to himself or any other person as a ‘specialist’, where in doing so would be misleading or amount to misrepresentation.
|
|
| 15. |
Establishment of veterinary para-professional cadres
| (1) |
The following cadres of veterinary para-professionals are established—
| (a) |
veterinary technologist is a veterinary para-professional who has attained an academic diploma certificate in animal health or a degree in animal health, in a course of study as approved by the Board, but does not include a person with a degree in veterinary medicine;
|
| (b) |
veterinary technician is a veterinary para-professional who has attained an academic certificate in an animal health course of at least two years of study as approved by the Board.
|
|
| (2) |
Despite the provisions of subregulation 1(b), a person referred to in section 17(1)(b)(ii) of the Act, who has undergone a one year course in animal health as prescribed by the Board and has passed an exam set by the Board, shall be registered as a Veterinary Technician.
|
| (3) |
A person referred to in this Regulation shall be required to sit an exam set by the Board to qualify for registration under this Act.
|
|
| 16. |
Temporary registration of a foreign veterinary surgeon
| (1) |
A foreign person wishing to practice veterinary surgery in Kenya shall apply to the Board in accordance with Form 24 set out in the First Schedule.
|
| (2) |
The Board shall approve the application in accordance with the criteria set out under section 16 of the Act.
|
| (3) |
The Board shall, on approval of the application, issue the applicant with a temporary registration certificate in accordance with Form 25 set out in the First Schedule.
|
| (4) |
A foreign veterinary surgeon may practice if he is issued with an annual practicing license in Form 26 set out in the First Schedule.
|
|
| 17. |
Continuous Professional Development
| (1) |
Every veterinary surgeon and veterinary para-professional shall undertake continuous professional development activities in the manner provided by the Board.
|
| (2) |
A veterinary surgeon and a veterinary para-professional shall not be retained in the register until he has acquired at least twenty and fourteen credit points respectively in the year under consideration.
|
| (3) |
A veterinary surgeon or a veterinary para-professional may apply to the Board for a waiver of some or all the credit points and shall give reasons for the request.
|
| (4) |
The Board may, for valid reasons such as medical reasons, absence from the country or non-veterinary commitments, waive some or all the credit points for a registered person.
|
| (5) |
A veterinary surgeon and veterinary para-professional shall show evidence of the continuous professional development activities completed by thirty first December of every year in the manner provided in Form 27 set out in the First Schedule.
|
| (6) |
In addition to filling Form 27 a veterinary surgeon or veterinary para-professional shall submit—
| (a) |
a summary of continuous professional development activities attended, including the nature, scope and duration of such activities; and
|
| (b) |
a summary of credit points acquired.
|
|
| (7) |
The Board may discipline any veterinary surgeon and veterinary para-professional who has not acquired the minimum number of credit points by—
| (a) |
requiring the person to follow a remedial continuous professional development programme;
|
| (b) |
requiring the person to write an examination determined by the Board; or
|
| (c) |
removing the person's name from the relevant register.
|
|
|
| 18. |
Accreditation
| (1) |
A person wishing to be accredited as a continuous professional development service provider shall apply to the Board in the manner provided in Form 28 set out in the First Schedule.
|
| (2) |
The Board shall confirm accreditation of a continuous professional development service provider in the manner provided in Form 29 set out in the First Schedule.
|
| (3) |
Every continuous professional development provider shall apply for approval and determination of credit point worth by the Board, of the intended activity in the manner provided in Form 30 set out in the First Schedule.
|
|
| 19. |
Fees for professional services
| (1) |
The Board shall from time to time determine in Form 31 set out in the First Schedule, the fees to be charged for the services it provides.
|
| (2) |
The Board shall, from time to time, set minimum fees for professional services offered by a person registered under the Act.
|
|
PART IV – REGISTRATION OF INSTITUTIONS AND ORGANISATIONS
| 20. |
Registration of institutions handling animals to safeguard their health and welfare
| (1) |
An institution intending to offer animal health services shall apply to the Board in Form 32 set out in the First Schedule and shall state—
| (a) |
the location and jurisdiction of the intended operation;
|
| (b) |
the type of service it intends to offer;
|
| (c) |
the name and registration status of the veterinary surgeon in the employment of the institution in charge of animal health matters; and
|
| (d) |
the facilities the institution has in order to offer the intended service.
|
|
| (2) |
Despite subregulation (1)(c), if the organization changes the veterinary surgeon, it shall notify the Board and shall suspend animal health services until it has recruited a veterinary surgeon in charge of the animal health services.
|
| (3) |
The Board may grant the institution an operation licence, with the necessary conditions, in accordance with Form 33 set out in the First Schedule.
|
|
| 21. |
Approval for veterinary clinical centres, hospitals and ambulatory services
| (1) |
A person intending to operate a veterinary clinical centre, veterinary hospital or an ambulatory service shall, before commencing such operation, apply to the Board for approval in accordance with Form 34 (a) set out in the First Schedule.
|
| (2) |
The Board shall on receipt of the application under subregulation (1), cause an inspection to be conducted of the veterinary clinical centre, veterinary hospital or an ambulatory service.
|
| (3) |
The Board shall, where the applicant complies with the standards set out in regulations 23, 24, or 25 and on payment of the requisite fee, approve the application and issue a licence in Form 34 (b) set out in the First Schedule.
|
| (4) |
A license issued under this Regulation shall be valid for a period of one year calculated from the date of issue.
|
| (5) |
Despite subregulation (4), a licence may be cancelled at any time when the Board establishes a breach of any provisions of these Regulations.
|
|
| 22. |
Approval for veterinary laboratory
| (1) |
A person shall apply to the Board for approval to operate a veterinary laboratory in the manner provided in Form 35 set out in the First schedule.
|
| (2) |
The Board shall inspect a veterinary laboratory for purposes of registration under subregulation (1).
|
| (3) |
The inspection under subregulation (2) shall be done once.
|
| (4) |
An applicant under this Regulation shall specify the laboratory services he intends to offer in the veterinary laboratory.
|
| (5) |
The Board shall inspect the veterinary laboratory to confirm that it satisfies the standards under regulation 27 and on approval issue an annual license in Form 36 set out in the First schedule.
|
| (6) |
A license may be cancelled by the Board any time an inspector establishes a breach of any provisions of these Regulations.
|
| (7) |
A veterinary clinical centre, veterinary hospital or an ambulatory service that has been licensed to operate under this Act shall not be required to make a separate application for its own in-house laboratory.
|
|
PART V – STANDARDS
| 23. |
Standards for a veterinary clinical centre
A veterinary centre shall satisfy the following minimum standards—
| (a) |
cleanliness, impervious walls and paved floors which are easy to clean and disinfect;
|
| (b) |
sufficient natural light supported by adequate artificial lighting;
|
| (c) |
clean protective equipment;
|
| (d) |
clean well groomed personnel;
|
| (e) |
facility for containment of micro-organisms and adequate protection against dissemination of diseases to personnel and animals;
|
| (g) |
facility for proper disposal of all waste materials such as incinerators or secure soak pits;
|
| (h) |
provision for proper disposal of unclaimed dead patients, on or off site;
|
| (i) |
sufficient ventilation and freedom from noxious odours;
|
| (j) |
sufficient species-specific restraining facilities;
|
| (k) |
sufficient refrigeration and storage facilities;
|
| (l) |
basic diagnostic and medical equipment including, thermometers, stethoscope, microscopes, surgical kit, lockable medicine kit, sampling tools and equipment;
|
| (m) |
implementation of Occupational Health and Safety Act (Cap. 236A) in the work place;
|
| (n) |
| (ii) |
diseases encountered; |
| (iv) |
inventory and stores; |
| (v) |
veterinary certificates which shall include the full name and signature of the attending veterinary surgeon, Kenya Veterinary Board registration number, the address. the full identification of the patient in terms of the name, species, sex, colour, age and any identification device number on the patient, date of vaccination or examination, and the generic and trade name of the vaccine used, batch number and the expiry date and manufacturer; and |
| (vi) |
any other record determined by the Board. |
|
|
| 24. |
Standards for a veterinary hospital
| (1) |
An animal hospital shall satisfy the minimum standards specified in regulation 23.
|
| (2) |
In addition to the provisions of regulation 23, an animal hospital shall have sufficient facilities including;
| (a) |
comfortable and safe accommodation with walls and floors that are easy to clean and disinfect with adequate bedding;
|
| (b) |
wholesome feeds and sufficient potable water;
|
| (c) |
diagnostic facilities including imaging and laboratory services;
|
| (d) |
at least one resident veterinary surgeon and one veterinary para-professional on duty at any given time; and
|
| (e) |
at least two well-equipped veterinary surgical theatres, a preparation room and a separate recovery room.
|
|
| (3) |
An animal hospital shall have adequate staff establishment, which shall comprise of at least three veterinary surgeons.
|
|
| 25. |
Standards for an ambulatory service
An ambulatory service shall have the following minimum standards—
| (a) |
sufficient means of transport;
|
| (b) |
clean protective equipment;
|
| (c) |
clean, well-groomed personnel;
|
| (d) |
a facility for containment of micro-organisms and adequate protection against dissemination of diseases to personnel and animals;
|
| (e) |
access to proper disposal of all waste materials such as incinerators or secure soak pits;
|
| (f) |
sufficient portable species-specific restraining facilities;
|
| (g) |
a contact point with adequate refrigeration and storage facilities for veterinary medicines, equipment and records; and
|
| (h) |
sampling tools and equipment.
|
|
| 26. |
Emergency service
A veterinary clinical centre, animal hospital or ambulatory facility shall offer twenty four hour emergency service.
|
| 27. |
Standards of veterinary laboratory
| (1) |
A veterinary laboratory shall—
| (a) |
be located away from known fire hazards;
|
| (b) |
be separated from non-related activities;
|
| (c) |
have restricted access by personnel; and
|
| (d) |
be vermin-proofed for pests including insects and rodents.
|
|
| (2) |
In addition to subregulation (1), a veterinary laboratory shall have the following minimum standards—
| (a) |
the floors and the walls of the building shall be constructed from materials that are easy to clean, impervious and resistant to corrosion by chemicals;
|
| (b) |
have access to an incinerator, pressure steam sterilizer or renderer for disposal of carcasses and waste;
|
| (c) |
provide a microbiological safety cabinet able to protect the operator, the product and the environment in laboratories dealing with microbial cultures;
|
| (d) |
sample collection and diagnostic equipment and facilities;
|
| (e) |
provide an autoclave or pressure steam sterilizer;
|
| (f) |
provide personnel with protective clothing and equipment for use only within the laboratory;
|
| (g) |
have a designated veterinary surgeon responsible for containment;
|
| (h) |
competent staff in the area of specialization;
|
| (i) |
provide warning notices for containment area indicating the type of hazard, the name and telephone number of the person responsible;
|
| (j) |
have laboratory alarm system, emergency lighting, fire fighting equipment and first aid kit;
|
| (k) |
post emergency protocols within the laboratory advising personnel on procedures to follow in case of emergencies; and
|
| (l) |
display standard operating procedures for the laboratory.
|
|
|
| 28. |
Standards for advertisement of veterinary services
| (1) |
An advertisement for services by a veterinary surgeon, a veterinary paraprofessional or a veterinary practice shall—
| (a) |
conform to the veterinary code of ethics:
|
| (b) |
comprise information that enriches the clients’ knowledge in animal health care;
|
| (d) |
be scientifically factual; and
|
| (e) |
not be an endorsement of a veterinary surgeon, veterinary para-professional, veterinary product or service.
|
|
| (2) |
A veterinary surgeon or veterinary para-professional shall display at the place at which he practices a sign bearing—
| (a) |
the name of the veterinary surgeon or veterinary para-professional;
|
| (b) |
the qualification as recorded in the register or veterinary surgeons, veterinary para-professionals or specialists;
|
| (c) |
the name, professional details and duration of visit of any foreign veterinary surgeon; and
|
|
| (3) |
A sign displayed at a veterinary premises may be illuminated by a light of constant intensity without causing an irritation to the general public.
|
| (4) |
A veterinary surgeon or veterinary para-professional whose premises are in a shopping complex or other place where a directory of tenants is provided may display on that directory a sign indicating that the veterinary surgeon or veterinary para-professional has a practice in that place.
|
| (5) |
An advertisement shall not contain any non-academic nominals.
|
| (6) |
Advertising in any telephone book, directory, website or other place or medium shall be accurate and shall comply with the standards set out in the Act.
|
|
| 29. |
Animal breeding services
| (1) |
A veterinary para-professional who has completed an artificial insemination course at an institution approved by the Board may apply for registration as a breeding service provider.
|
| (2) |
A person shall offer veterinary breeding services if he is registered under this Act.
|
|
| 30. |
Standards for Training and curriculum for breeding services
| (1) |
A training institution shall not offer training in breeding services unless the institution is licensed by the Board in accordance with Form 38 (b) set out in the First Schedule.
|
| (2) |
An institution training in breeding services shall have facilities approved by the Board and qualified personnel.
|
| (3) |
A trainee in breeding services shall have a minimum qualification of a veterinary para-professional.
|
| (4) |
A person shall not offer training in breeding services unless he uses the curriculum approved by the Board.
|
| (5) |
A breeding service provider for all species, other than cattle, shall have the physical address and facilities approved by the Board.
|
|
| 31. |
Reporting of notifiable diseases
A notifiable disease shall be reported in the manner provided in Form 37 set out in the First Schedule.
|
PART VI – TRAINING INSTITUTIONS AND CONDUCT OF EXAMINATIONS
| 32. |
Approval of training institution
| (1) |
An animal health course or a veterinary medicine course shall be offered in a registered institution or in a faculty of veterinary medicine.
|
| (2) |
An institution desiring to offer courses of training leading to the award of animal health degree, diploma or certificate, or degree of Bachelor of Veterinary Medicine shall after being registered by the relevant body, apply to the Board for approval by submitting an application form in accordance with Form 38(a) set out in the First Schedule.
|
| (3) |
A person or institution shall not offer training in Animal Health or Veterinary Medicine unless they are registered by the Board.
|
| (4) |
An institution or person shall offer training courses in Animal Health or Veterinary Medicine using the curriculum approved by the Board.
|
| (5) |
The Board shall, on receipt of the application under subregulation (2), inspect the institution and if satisfied that it conforms to standards specified under these Regulations and upon payment of the requisite fees, approve the institution for the courses proposed.
|
| (6) |
The Board shall inspect approved training institutions from time to time.
|
| (7) |
The Board shall, in every five years, inspect institutions for purposes of approval.
|
|
| 33. |
Standards for institutions training for award of degree of veterinary medicine
| (1) |
A training institution desiring to offer courses for the award of degree of veterinary medicine shall have adequate physical facilities and equipment that meet the following minimum standards—
| (a) |
a lecture theatre for every class;
|
| (b) |
a laboratory for each major discipline;
|
| (c) |
seminar rooms and other teaching spaces which shall be adequate in number and size for the instructional purposes intended and the number of students enrolled;
|
| (d) |
administrative and faculty offices;
|
| (e) |
a teaching animal hospital with the minimum standards set out in regulation 24 and which has adequate teaching laboratory, research and clinical equipment for examination and diagnostics of all animals used by the institution;
|
| (f) |
an animal farm with the full spectrum of animal species to give the students adequate exposure in a ratio of at least two animals per student-admission for cattle, sheep, goat and pigs and one flock of two hundred poultry for ten student-admission and at least one fish farm and one rabbit farm;
|
| (g) |
a veterinary library; and
|
| (h) |
access to facilities offering food and accommodation for students in the vicinity.
|
|
| (2) |
The facilities and equipment specified in subregulation (1) shall be maintained clean and in good repair.
|
| (3) |
The number, qualification and terms of service for teaching and support staff shall conform to the minimum standards prescribed by the body responsible for higher education.
|
| (4) |
Without prejudice to subregulation (1), an institution shall meet the following additional requirements—
| (a) |
the lecturers for animal health courses shall be veterinary surgeons;
|
| (b) |
the lecturers shall possess relevant qualifications for the subjects they teach;
|
| (c) |
the head of the faculty or school shall be a veterinary surgeon;
|
| (d) |
the head of veterinary academic section shall be a veterinary surgeon;
|
| (e) |
the ratio of permanently employed to part-time lecturers teaching the animal health programmes shall not be less than 3:1; and
|
| (f) |
the staff establishment shall include sufficient numbers of technologists, technicians and other relevant staff.
|
|
| (5) |
In addition to the standards and equipment provided in regulation 24, the institutional surgical theatres and laboratory shall have the following additional equipments—
| (a) |
computed tomography scan;
|
| (b) |
magnetic resonance imaging;
|
| (c) |
fluoroscopy for cardiac catheterization and diagnostic imaging;
|
| (e) |
cardiopulmonary bypass equipment;
|
| (h) |
hemodynamic data services and software;
|
| (j) |
histopathology equipment;
|
| (m) |
light microscopes for each student;
|
| (o) |
ultrasound equipment; and
|
|
|
| 34. |
Standards for institutions training for award of diploma in animal health
| (1) |
A training institution desiring to offer courses tor the award of diploma in animal health shall have adequate physical facilities and equipment maintained clean and in good state of repair and shall meet the following minimum standards—
| (a) |
a lecture theatre for every class;
|
| (b) |
a laboratory for each major discipline;
|
| (d) |
administrative and staff offices;
|
| (e) |
an animal farm with the full spectrum of animal species to give the students adequate exposure in a ratio of at least two animals per student-admission for cattle, sheep, goat and pigs and one flock of two hundred poultry for ten student-admission and at least one fish farm and one rabbit farm;
|
| (f) |
a veterinary library; and
|
| (g) |
access to facilities offering food and accommodation for students in the vicinity.
|
|
| (2) |
The number, qualification and terms of service for teaching and support staff shall conform to the minimum standards prescribed by the body responsible for tertiary and higher education.
|
| (3) |
Without prejudice to subregulation (1), the institution shall meet the following additional requirements—
| (a) |
lecturers for animal health courses shall be veterinary surgeons;
|
| (b) |
lecturers shall possess relevant qualifications for the subjects they teach;
|
| (c) |
the head of the relevant section or department offering animal health course shall be a veterinary surgeon;
|
| (d) |
the ratio of permanently employed to part-time lecturers teaching the animal health programmes shall not be less than 3:1; and
|
| (e) |
the staff establishment shall include sufficient numbers of technologists, technicians and other relevant staff;
|
| (f) |
minimum teaching aids shall be availed.
|
|
| (4) |
The standards of the laboratory and equipment in the institution shall conform to the standards in section 29 of the Act.
|
|
| 35. |
Standards for institutions training for the award of animal health certificates
| (1) |
A training institution desiring to offer courses for the award of certificate in animal health shall have adequate physical facilities and equipment maintained clean and in good state of repair and shall meet the following minimum standards—
| (a) |
a lecture theatre for every class;
|
| (b) |
two laboratories, one for basic sciences and one for applied sciences;
|
| (d) |
administrative and staff offices;
|
| (e) |
an animal farm with the full spectrum of animal species to give the students adequate exposure in a ratio of at least two animals per student-admission for cattle, sheep, goat and pigs and one flock of two hundred poultry for ten student-admission and at least one fish farm and one rabbit farm;
|
| (f) |
a veterinary library; and
|
| (g) |
food and accommodation for students.
|
|
| (2) |
The number, qualification and terms of service for teaching and support staff shall conform to the minimum standards prescribed v the body responsible for tertiary and higher education.
|
| (3) |
Without prejudice to subregulation (2), the institution shall set the following additional requirements—
| (a) |
lecturers for animal health courses shall be veterinary surgeons;
|
| (b) |
lecturers shall possess relevant qualifications for the subjects they teach;
|
| (c) |
the head of the relevant section or department offering animal health course shall be a veterinary surgeon;
|
| (d) |
the ratio of permanently employed to part-time lecturers teaching the animal health programmes shall not be less than 3:1;
|
| (e) |
the staff establishment shall include sufficient numbers of technologists, technicians and other relevant staff; and
|
| (f) |
minimum number and types of teaching aids shall be availed.
|
|
| (4) |
The standards of the laboratory and equipment in the institution shall conform to the standards in section 29 of the Act.
|
|
| 36. |
Indexing of students
| (1) |
Every veterinary medicine or animal health student shall be indexed by the Board.
|
| (2) |
A student shall apply for indexing to the Board in Form 39(a) set out in the First Schedule.
|
| (3) |
A student shall submit the application to the institution where he is admitted for training.
|
| (4) |
An institution training students shall forward applications submitted under subregulation (3), to the Board on behalf of the students.
|
| (5) |
An institution under subregulation (4), shall summarize the particulars of all the applications in Form 39 (b) set out in the First Schedule and shall submit the summary, together with the students' applications, to the Board.
|
|
| 37. |
Conduct of examinations in authorized institutions
| (1) |
The examinations for a veterinary medicine degree or animal health courses shall include examinations in all the subjects as specified by the Board.
|
| (2) |
At least two examiners, one of whom was not involved in the teaching of the subject to the candidate, shall participate in the evaluation of each subject examined.
|
| (3) |
A candidate for an examination for veterinary medicine degree or an animal health course shall pass an examination in a subject if he attains at least fifty per cent or the equivalent of fifty per cent of the maximum marks obtainable in the examination, in the marking system employed by the institution.
|
|
| 38. |
Internship for graduates
| (1) |
A veterinary medicine or animal health graduate shall apply to the Board for temporary registration for the period of internship in the manner provided in Form 40 set out in the First Schedule.
|
| (2) |
The Board shall, after payment of requisite fee, grant an applicant under subregulation (1) intern registration and issue him with a certificate in accordance with Form 41 set out in the First Schedule.
|
| (3) |
A veterinary medicine intern may examine animals, carry out diagnostic tests and administer treatment under the direction of a veterinary surgeon and perform surgical operation under the supervision of a veterinary surgeon.
|
| (4) |
A veterinary para-professional intern shall perform tasks and activities equal with his training under the direction of a veterinary surgeon.
|
| (5) |
A veterinary para-professional intern may examine animals and carry out diagnostic tests and administer treatment under the direction of a veterinary surgeon.
|
| (6) |
Upon successful completion of internship, the supervisor shall make recommendations to the Board for evaluation and registration of the intern in Form 42 set out in the First Schedule.
|
| (7) |
An intern or supervisor may refer any complaint during the internship to the Board in writing.
|
|
FIRST SCHEDULE
FORMS
|
FORM 1
|
(r. 5)
|
|
NOMINATION FORM
|
|
NOMINATION OF VETERINARY SURGEON CANDIDATES FOR ELECTION FOR APPOINTMENT TO THE BOARD ESTABLISHED BY SECTION 4 (F) OF THE VETERINARY SURGEONS AND VETERINARY PARA-PROFESSIONALS ACT, 2011
|
|
(to be filled in block letters)
|
Name of Candidate...................................................
Address..............................................................
Telephone ..................... e-mail................................
KVB regisration No...................................................
Academic qualifications .............................................
Career record........................................................
Special interests....................................................
Record in public and professional service............................
Self declaration of integrity and ethics ............................
Signature of the candidate ................... Date ..................
Name and address of proposer ......................................
Registration No. of proposer .........................................
Signature of proposer ............................... Date..............
Name and address of Seconder ........................................
Registration No. of Seconder.........................................
Signature of Seconder............................. Date...........
_________________________
|
FORM 2
|
|
(r. 5)
|
|
NOMINATION FORM
|
|
NOMINATION OF VETERINARY PARA-PROFESSIONAL CANDIDATES FOR ELECTION FOR APPOINTMENT TO THE BOARD ESTABLISHED BY SECTION 4 (G) OF THE VETERINARY SURGEONS AND VETERINARY PARA-PROFESSIONALS ACT, 2011.
|
|
(to be filled in block letters)
|
Name of Candidate...................................................
Address..............................................................
Telephone ...........................................................
E-mail...............................................................
KVB registration No...................................................
Academic qualifications .............................................
Career record........................................................
Special interests....................................................
Record in public and professional service............................
Self-declaration of integrity and ethics ............................
Signature of the candidate ...................Date ..................
Name and address of proposer ......................................
Registration No. of proposer .........................................
Signature of proposer ...............................Date..............
Name and address of Seconder ........................................
Registration No. of Seconder.........................................
Signature of Seconder ............................. Date...........
_________________________
|
FORM 3
|
|
(r. 7)
|
|
BALLOT FORM FOR VETERINARY SURGEONS ELECTIONS
|
|
Name of Candidates
|
Insert X in this column against the names of candidates for whom you wish to vote
|
|
|
|
|
|
|
|
|
|
|
|
|
Full name of voter (in block letter)..................................
Registration Number ..................................................
Signature of voter....................................................
Date...................................................................
_________________________
|
FORM 4
|
|
(r. 7)
|
|
BALLOT FORM FOR VETERINARY PARA-PROFESSIONALS ELECTIONS
|
|
Name of Candidates
|
Insert X in this column against the names of candidates for whom you wish to vote
|
|
|
|
|
|
|
|
|
|
|
|
|
Full name of voter (in block letter)..................................
Registration Number ..................................................
Signature of voter....................................................
Date...................................................................
_________________________
|
FORM 5
|
|
(r.10(a))
|
|
VETERINARY SURGEONS REGISTER
|
|
Serial No.
|
Reg. No.
|
Reg. Date
|
Name
|
Address
|
Qualification
|
Year of graduation
|
College of graduation
|
Retention
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
_________________________
|
FORM 6
|
(r. 10 (b))
|
|
LICENCED VETERINARY SURGEONS REGISTER
|
|
Serial No.
|
Reg. No.
|
Reg.Date
|
Name
|
Address
|
Qualification
|
Place of practice
|
Name of practice
|
Practice fee
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
_________________________
|
FORM 7
|
(r. 10(c))
|
|
VETERINARY PARA-PROFESSIONALS (TECHNOLOGISTS DEGREE) REGISTER
|
|
Serial No.
|
Reg. No.
|
Reg. Date
|
Name
|
Address
|
Date of Qualification
|
College of graduation
|
Retention
|
Practice fee
|
Practice name
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
_________________________
|
FORM 8
|
(r. 10(d))
|
|
VETERINARY PARA-PROFESSIONALS (TECHNNOLOGISTS DIPLOMA) REGISTER
|
|
Serial No.
|
Reg. No.
|
Reg. Date
|
Name
|
Address
|
Date of Qualification
|
College of graduation
|
Retention
|
Practice fee
|
Practice name
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
_________________________
|
FORM 9
|
(r.10 (e))
|
|
VETERINARY PARA-PROFESSIONALS (TECHNICIANS) REGISTER
|
|
Serial No.
|
Reg. No.
|
Reg. Date
|
Name
|
Address
|
//Date ofQualification//
|
//College ofgraduation//
|
Retention
|
Practice fee
|
Practice name
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
_________________________
|
FORM 10
|
(r. 10 (f))
|
|
LICENCED VETERINARY PARA-PROFESSIONALS REGISTER
|
|
Serial No.
|
Reg. No.
|
Reg. Date
|
Name
|
Address
|
Date of Qualification
|
College of graduation
|
Retention
|
Practice fee
|
Practice name
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
_________________________
|
FORM 11
|
(r. 10 (e))
|
|
VETERINARY INTERNS REGISTER
|
|
Serial No.
|
Reg. No.
|
Reg. Date
|
Name
|
Address
|
Date of Qualification
|
College of graduation
|
Hosting Organization
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
_________________________
|
FORM 12
|
(r. 10 (h))
|
|
VETERINARY INTERNS REGISTER
|
|
Serial No.
|
Reg. No.
|
Reg. Date
|
Name
|
Address
|
Course offered
|
Location
|
Registration fee
|
Date of last inspection
|
Status
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
_________________________
|
FORM 13
|
(r. 10 (i))
|
|
REGISTER FOR LICENCED VETERINARY LABORATORIES
|
|
Serial No.
|
Reg. No.
|
Reg. Date
|
Name of Laboratory
|
Address
|
Location
|
Vet surgeon laboratory specialist
|
Date of last inspection
|
Status
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
_________________________
|
FORM 14
|
(r. 10 (j))
|
|
REGISTER FOR FACILITIES AND ORGANIZATIONS OFFERING ANIMAL HEALTH AND WELFARE SERVICES
|
|
Serial No.
|
Reg. No.
|
Reg. Date
|
Name of institution
|
Address
|
Location
|
Nature of operations
|
Veterinary surgeon in-charge
|
Date of last inspection
|
Status
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
__________________________________
|
FORM 15
|
(r. 10 (k))
|
|
VETERINARY SPECIALISTS REGISTER
|
|
Serial No.
|
Reg. No.
|
Reg. Date
|
Name
|
Address
|
Qualification
|
Date of Qualification
|
Specialization
|
Institution
|
Practice Institution
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
__________________________________
|
FORM 16
|
(r. 10 (1))
|
|
FOREIGN VETERINARY SURGEONS REGISTER
|
|
Serial No.
|
Reg. No.
|
Reg. Date
|
Name
|
Country
|
Address
|
Qualification
|
Year of graduation
|
College of graduation
|
Retention
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
__________________________________
|
FORM 17
|
(r. 10 (m))
|
|
VETERINARY STUDENT INDEXING REGISTER
|
|
Serial No.
|
Reg. No.
|
Reg. Date
|
Name
|
Address
|
Date of admission
|
College of admission
|
College of graduation
|
Continuity
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
__________________________________
|
FORM 18
|
(r. 10 (n))
|
|
REGISTER FOR ACCREDITED CPD PROVIDERS
|
|
Serial No.
|
Reg. No.
|
Reg. Date
|
Name
|
Address
|
Program offered
|
Duration of program
|
Status
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
__________________________________
|
FORM 19
|
(r. 10 (o))
|
|
REGISTER FOR APPROVED VETERINARY CLINICAL CENTRES/HOSPITALS/AMBULATORY SERVICES
|
|
Serial No.
|
Reg. No.
|
Reg. Date
|
Name of owner
|
Name of facility
|
Address
|
Location
|
Vet surgeon in-charge
|
Date of last inspection
|
Status
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
__________________________________
|
FORM 20
|
(r. 10 (p))
|
|
REGISTER OF BREEDING SERVICE PROVIDERS
|
Year............................................
|
Serial No.
|
Reg. No.
|
Reg. Date
|
Name
|
Address
|
Academic qualification
|
Breeding services Qualification
|
Year of certification
|
College of certification
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
__________________________________
|
FORM 21
|
(r. 11)
|
|
VETERINARY CLINICAL CENTRE/HOSPITAL/AMBULATORY SERVICES INSPECTION REPORT FORM
|
I, the undersigned of (postal address) ..................have today carried out an inspection of ..........................as required by section 6 (2) (m) of Veterinary Surgeons and Para-Professionals Act.
| A. |
Identification of premises;
| (1) |
Name of Veterinary Surgeon/Veterinary Para-Professional...............................
|
| (2) |
Physical location......................................
|
| (3) |
Address.................................................
|
| (4) |
Practicing license No. .......................................
|
| (5) |
Authorized Services Provided..........................
|
|
| 1. |
Veterinary Clinic Structure and Location ..........................
|
| 2. |
Diagnostic Facilities Status...............................
|
| 3. |
Surgical Facilities Status ..................................
|
| 4. |
Treatment Room Status............................................
|
| 5. |
Status of Sourcing, Handling, Storage of Drugs and Vaccines ...................................
|
| 6. |
Stationery & Records.....................................
|
| 7. |
Personal Protection Equipments .............................
|
| 8. |
Status of Ambulatory Facilities...........................
|
| 9. |
Records of Official Reporting of Notifiable Diseases...............................
|
| 10. |
Veterinary medicines and other inputs................................
|
| 11. |
Other comments .............................................
I.......................name and registration number................... (person in charge of the inspected veterinary clinic/hospital/ambulatory) confirm that my premises has been inspected.
Date ..............,.....................
FOR OFFICIAL USE
I have the following recommendations to make:
.........................................................
Signature..............................
Date .....................................
Chief Inspector
Comments ......................................................
Signature ...................... Date....................
The Chief Executive Officer, Kenya Veterinary Board
Veterinary clinic/hospital/ambulatory approved/rejected
Action taken ........................................
Signature .......................... Date..............................
|
__________________________________
|
FORM 22
|
(r. 14(2))
|
|
APPLICATION FOR REGISTRATION AS A VETERINARY SPECIALIST
|
The Registrar,
The Kenya Veterinary Board,
Kabete.
I, ................................................of ................................ hereby make an application for registration as a Veterinary Specialist/Consultant (delete as appropriate)
My qualifications are.......................................
I enclose:-
(1) The following certificates and testimonials ..................................
.....................................................................
.....................................................................
.....................................................................
Date: .............................. Signature: ....................
__________________________________
|
FORM 23
|
(r. 14(3))
|
|
CERTIFICATE OF REGISTRATION AS A VETERINARY SPECIALIST
|
(Name) ........................................ is hereby registered as a veterinary specialist in ...................... by the college of ................... as provided by section 6 (2)(j) of the Veterinary Surgeons and Veterinary Para-Professionals Act, 2011.
Given this ........ day of................20 ..............
This certificate is valid for 5 years from the date of issue.
....................................
Registrar, Kenya Veterinary Board
__________________________________
|
FORM 24
|
(r. 16(1))
|
|
APPLICATION FORM FOR TEMPORARY REGISTRATION OF A FOREIGN VETERINARY SURGEON
|
The Registrar,
The Kenya Veterinary Board,
Kabete.
Name of the organization .....................................
Address (physical)............................................
Proposed area of operation.......................................
Hereby make an application for registration on behalf of ..........................................
Name of the veterinary surgeon..................................
Address (physical).............................................
Registration body...............................................
Professional association membership................................
I hereby provide the following documents:
| 3. |
Professional association certificate
|
| 4. |
Letter of good professional conduct
|
| 5. |
ldentification document
........................ Date.......................
Signature of Head of organization and stamp
|
__________________________________
|
FORM 25
|
(r. 16(3))
|
|
CERTIFICATE OF REGISTRATION OF A FOREIGN VETERINARY SURGEON
|
(Name) .............................. is hereby registered as a Foreign Veterinary Surgeon under section 16 of the Veterinary Surgeons and Para-Professionals Act, 2011.
The terms and conditions of this temporary registration are as follows:
........................................................................
........................................................................
........................................................................
........................................................................
Given this .....................day of ......................20 ...........
............................
Registrar, Kenya Veterinary Board
__________________________________
|
FORM 26
|
(r. 16(4))
|
|
LICENCE TO PRACTISE AS A FOREIGN VETERINARY SURGEON
|
Name of Veterinary Surgeon ............................................
Organization name .................................................
Physical address: ........................P.O..Box......................
Telephone:..........................................................
Email: ..........................................................
Is hereby licensed to practice as a Foreign Veterinary Surgeon in accordance with the provisions of section 16 of the Veterinary Surgeons and Veterinary Para-Professionals Act, 2011
This license is subject to the conditions set out by the Board below:
....................................................................................
....................................................................................
....................................................................................
Dated this ......................day of ................. 20.............
.........................................
Registrar, Kenya Veterinary Board
__________________________________
|
FORM 27
|
(r. 17(5))
|
|
KVB CPD RECORD SHEET
|
Name: ............................... KVB Registration Number: ..........................
Postal Address: ............................... CPD Year: ...............................
Email Contact: ............................... Phone Contact:...........................
|
Activity Dates
|
CPD Activity
|
CPD Service Provider
|
CPD Points awarded
|
Certificate Attached
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total CPD Points for the Year
|
|
|
I declare that all the information provided on this form and on the attached certificates and/or course details are correct and valid to the best of my knowledge.
Signature ____________________ Date____________________
Note: This form MUST be submitted accompanied with copies of certificate of attendance or course details when paying your annual retention fees.
Kindly retain a copy of your completed CPD Record sheet, original certificates and course details as they are subject to Board audits.
__________________________________
|
FORM 28
|
(r. 18(1))
|
|
APPLICATION FOR APPROVAL AS A CONTINUOUS PROFESSIONAL DEVELOPMENT PROVIDER
|
Name of service provider .......................................
Full Address:.......................................................
Physical location: ................................................
Contact person:....................................................
Tel Number (Landline):..........................................
Tel Number (Mobile)................................................
Fax number............................................................
Email address: ........................................................
| 1. |
Proposed CPD Year _________________________________
|
| 2. |
Describe the Human and Institutional capacity
.......................................................................
.......................................................................
.......................................................................
|
| 3. |
Describe relevant experience in conducting similar activities.
.......................................................................
.......................................................................
.......................................................................
|
| 4. |
Referees: (Please provide names of two referees who are familiar with your organisation)
|
| (1) |
Name .......................................................................
Job titte .......................................................................
Physical Address.......................................................................
P.O. Box .....................................Tel No.............................
Email............................Fax No.....................................
(2) Name .......................................................................
Job titte .......................................................................
Physical Address.......................................................................
P.O. Box .....................................Tel No.............................
Email............................Fax No.....................................
DECLARATION:
The provider declares that all information provided herein is correct and agrees to comply with the criteria; and terms and conditions of authorization
Name.................................................
Signature.............................................
Job title............................................
Date ..................................................
Please return the duly completed application to:
The Kenya Veterinary Board
P.O. Box 513 - 00605
NAIROBI, or
Email : info@kenyavetboard.org
|
__________________________________
|
FORM 29
|
(r. 18(2))
|
|
CONFIRMATION OF ACCREDITATION AS A CPD PROVIDER BY THE KVB
|
The KVB hereby confirms that you have been accredited as a CPD provider with registration number.......................
Yours sincerely
Signature: ..................................
Date: .......................................
__________________________________
|
FORM 30
|
(r. 18(3))
|
|
LICENCE TO PRACTISE AS A FOREIGN VETERINARY SURGEON
|
APPLICATION FOR APPROVAL OF CONTINUOUS PROFESSIONAL DEVELOPMENT ACTIVITY
| 1. |
Name of CPD provider .....................................
Address ..........................................
Tel:.................... Fax: ..............E-mail address:........................
KVB Registration/Approval No. ...................................
Name of coordinator/contact person ...................................
Proposed Date(s): (attach schedule if it is repetitive)...................Duration (in hours) ..............
Venue .........................................................
Title of activity/program :..........................................
Congress ..................... Conference ...............
Symposium..................... Course .................
Workshop .................... Others (specify): ...........
|
| 2. |
Activity outline with expected knowledge, skills and attitudes to be imparted (attach program): ...................................
|
| 3. |
List of presenters/facilitators (Attach CVs)
|
Name
|
Address
|
Professional registration No.
|
Role
|
Specialty
|
Qualification
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| 8. |
Target audience: .....................................
|
| 9. |
Expected Number of participants (approximate) .........................
Signature of applicant:............................
Date:............................................
|
__________________________________
|
FORM 31
|
(r. 19(1))
|
|
FEES SUMMARY FORM
|
|
Purpose
|
Amount (Paid )
|
Frequency
|
|
Cost of applications
|
1,000 (one thousand)
|
Once
|
|
Fees to practice as a Veterinary Surgeon
|
8,000 (Eight thousand)
|
Annually
|
|
Inspection fees for registration of a veterinary clinical centre
|
50,000 (fifty thousand)
|
Once
|
|
Inspection fees for veterinary clinical centre
|
15,000 (fifteen thousand)
|
Annually
|
|
Inspection fees for registration for a veterinary hospital
|
100,000 (one hundred thousand)
|
Once
|
|
Licence fees for veterinary hospital
|
30,000 (thirty thousand)
|
Annually
|
|
Inspection fees for ambulatory facility
|
5,000 (five thousand)
|
Annually
|
|
Fees to practice as a Veterinary Specialist
|
20,000 (Twenty thousand
|
Every five years
|
|
Fees to practice as a Veterinary Para-Professional
|
3,000 (Three thousand)
|
Annually
|
|
Fees for registration/retention as a Veterinary Surgeon
|
4,000 (Four thousand)
|
Annually
|
|
Fees for registration/retention as aveterinary para-professional
|
|
|
|
1 Veterinary Technologist2. Veterinary Technician
|
2,000 (Two thousand)1,000 (One thousand)
|
Annually
|
|
Fees for temporary registration as a veterinary surgeon intern
|
1,000 (One thousand)
|
Once
|
|
Fees for temporary registration as a veterinary para-professional intern
|
500 (Five hundred)
|
Once
|
|
Fees for inspection for registration of veterinary laboratory
|
100,000 (One hundred thousand)
|
Once
|
|
Fees for licence of a Veterinary laboratory
|
10,000 (Ten thousand)
|
Annually
|
|
Fees for inspection of Animal Health Training Institutions1. University2.Other training institutions
|
750,000 (Seven hundred & fifty thousand)500,000 (Five hundred thousand)
|
Every five years
|
|
Fees for temporary registration andretention of a Veterinary Surgeon
|
1000 USD (One thousand USD)
|
Every two years
|
|
CPD activity Fees
|
10% of the gross proceeds
|
per activity
|
|
License for Non-GovernmentalAnimal Health and Welfare Providers
|
100,000 (one hundred thousand)
|
Once
|
|
Inspection fees for Non-GovernmentalAnimal Health and Welfare Providers
|
30,000 (Thirty thousand)
|
Annually
|
|
Application fees
|
1,000 (one thousand)
|
Per application
|
|
Board examination fees
|
9,500 (nine thousand five hundred)
|
Per examination
|
__________________________________
|
FORM 32
|
(r. 20 (1))
|
|
APPLICATION FOR APPROVAL FOR NON ANIMAL HEALTH GOVERNMENTAL PROVIDERS OF SERVICES
|
The Chief Executive Officer,
Kenya Veterinary Board, P. O. Box 513 - 00605,
Nairobi.
I/we........................................... wishing to offer animal health services do hereby apply for approval of our organization known as ..................... situated in the town of....................................
Our organization desires to offer animal health services in the following region of Kenya ......................................
The animal health operation of our organization will be under the responsibility of .................... a registered veterinary surgeon.
I hereby provide copies of registration documents of they organization and the veterinary surgeon.
Date. ..............................
Signature of applicant (in-charge of the organization)
__________________________________
|
FORM 33
|
(r. 20 (2))
|
|
GOVERNMENTAL PROVIDERS OF ANIMAL HEALTH SERVICES
|
Name ....................................................
Physical address:.............................................
P.O. Box Telephone:...........................................
Email: ................................... is hereby licensed to offer animal health services.
The organization shall offer animal health services at ........................
This license is subject to the conditions set out by the Veterinary Board below:
...........................................................................
...........................................................................
...........................................................................
...........................................................................
Dated this .................day of ...........20............
__________________________________
|
FORM 34
|
(a)(r. 21 (1))
|
|
APPLICATION FOR APPROVAL OF A VETERINARY CLINICAL CENTRE/HOSPITAL/AMBULATORY FACILITY
|
The Chief Executive Officer,
Kenya Veterinary Board,
P. O. Box 513 - 00605,
Nairobi.
I/we. ................................................... wishing to operate a veterinary clinic/hospital/ambulatory do hereby apply for registration of premises situated at........................in the town of........................................
The veterinary clinic/hospital/ambulatory will operate under the control of ...................... a veterinary surgeon registration No............in accordance with Part III of the Act.
Date. .................................
Signature of applicant.
__________________________________
|
FORM 34
|
(b)(r.21 (3))
|
|
LICENCE TO OPERATE A VETERINARY CLINICAL/VETERINARY/HOSPITAL/ AMBULATORY SERVICE
|
Name of licensee......................................
Name of veterinary clinic/ veterinary hospital/ambulatory service .................................
Physical address: ........................................
P.O. Box Telephone: ..................................................
Email: ................................. is hereby licensed to operate a veterinary clinic/ veterinary hospital/ambulatory service in accordance with the provisions of the Veterinary Surgeons and Veterinary Para-Professionals Act.
This license is subject to the conditions set out by the Veterinary Board below:
...........................................................................
...........................................................................
...........................................................................
...........................................................................
Dated this ..........................day of ...........20 ............
__________________________________
|
FORM 35
|
(r.22 (1))
|
|
APPLICATION FOR APPROVAL FOR A VETERINARY LABORATORY
|
The Chief Executive Officer,
Kenya Veterinary Board,
P. O. Box 513 - 00605,
Nairobi.
In accordance with the provisions of the Veterinary Surgeons and Veterinary Para-Professionals (Veterinary Clinic) Act.
I/we............................................ wishing to operate a veterinary laboratory hereby apply for registration of premises situated at .................:.................. in the town of .................................
The veterinary laboratory will operate under the contol of ..................... a veterinary surgeon registration number.......................
Date. ....................................................
Signature of applicant.
__________________________________
|
FORM 36
|
(r.22 (4))
|
|
LICENCE TO OPERATE A VETERINARY LABORATORY
|
Name of licensee.......................................... of
Laboratory name ............................................
Physical address:...........................................
P.O. Box ....................... Telephone:..............
Email: ..................................................
is hereby licensed to operate a Veterinary Laboratory in accordance with the provisions of the Veterinary Surgeons and Veterinary Para-Professionals Act.
This license is subject to the conditions set out by the Veterinary Board below:
..................................................................................................
..................................................................................................
................................................................................................
Dated this ..........................day of ...........20 ............
__________________________________
|
FORM 37
|
(r.31)
|
|
SUSPECTED NOTIFIABLE DISEASES REPORTING FORM
|
I Dr/Mr/Ms. ............................. of ........................ Telephone............ report the following disease as detailed below.
|
Name of owner
|
|
|
Date
|
|
|
Location
|
|
|
Species (No. affected)
|
|
|
History
|
|
|
Symptoms
|
|
|
Action taken (including treatment reporting etc)
|
|
|
Reported to
|
|
Date .............................
Signature ................................
Note:
Report must be done in three copies and disseminated as follows:
| (a) |
original, to the nearest Government officer;
|
| (b) |
second copy to the Director of Veterinary Services; and
|
| (c) |
third copy to be retained by the veterinary surgeon and veterinary para-professional reporting.
|
__________________________________
|
FORM 38
|
(a)(r. 32 (2))
|
|
APPLICATION FOR APPROVAL FOR TRAINING INSTITUTIONS OFFERING ANIMAL HEALTH COURSES
|
The Chief Executive Officer,
Kenya Veterinary Board,
P. O. Box 513 - 00605,
Nairobi.
In accordance with the provisions of section 6(2) (c) of thc Veterinary Surgeons and Veterinary Para-Professionals Act, 2011.
I/we................................................ wishing to offer animal health services do hereby apply for approval of our institution
known as .............................. situated in the town of...............................
Our institution desires to offer the following animal health courses:
.........................................................................................
The animal health courses will be under the responsibility of ..... .............., all registered veterinary surgeon.
I hereby provide copies of registration documents of the institution and the veterinary surgeon(s) responsible.
Date. ..................................................
Signature of applicant (in-charge of the organization)
__________________________________
|
FORM 38
|
(b)(r. 30 (1))
|
|
ACCREDITATION CERTIFICATE FOR ANIMAL HEALTH TRAINING INSTITUTIONS
|
|
KENYA VETERINARY BOARDAccreditation Certificate for Animal Health Training InstitutionsThis is to Certify that_________________________________________________________Has been accredited by the Board on this _________________day of ___________________to offer Training in Animal Health at____________________________Signed_____________________________KVB RegistrarSEALThis accreditation if valid for five (5) years
|
__________________________________
|
FORM 39
|
(a)(r. 36 (6))
|
|
APPLICATION OF INDEXING OF ANIMAL HEALTH STUDENTS
|
|
PASSPORTPHOTO
|
KVB OFFICIAL USE
|
|
Index number
|
|
|
Indexing Officer
|
|
IMPORTANT INFORMATION
This form MUST be completed in FULL and submitted to the Registrar, Kenya Veterinary Board within 30 days following commencement of the training.
All applications MUST be accompanied with copies of your National Identity (ID) Card/Passport, Certified KSCE Examination Results Slip, Secondary School Leaving Certificate, and 1 (one) coloured passport size photograph. Upon upgrading, applicants must attach copies of previous certificates/qualifications and practice license.
|
Application Information [PRINT]
|
|
|
|
Surname
|
|
ID/Passport No.
|
|
Email address
|
|
|
MiddleName
|
|
Citizenship
|
|
Personal Mobile phone
|
|
|
First Name
|
|
Home Address
|
|
Date of birth
|
|
|
Training information
|
|
Secondary schoolattended
|
|
Grade obtained
|
|
|
Certificate held
|
|
Training institution
|
|
|
Certificate no.
|
|
Date trainingcommencement
|
|
|
Training cadres [Clearly tick the course to be undertakenJ
|
|
Certificate in Animal Health
|
|
Bachelor of Veterinary Medicine
|
|
|
Diploma in Animal Health
|
|
Other (specify)
|
|
|
Declaration
|
|
I hereby declare that the foregoing information is true and correct to the best of my knowledge
|
|
Applicant signature:
|
Date:
|
|
I hereby declare that the applicant is suitable to be indexed by the Kenya Veterinary Board
|
|
Dean/Principal of Animal Health Training Institution [Please include official stamp]
|
Date:
|
|
Name
|
Signature:
|
|
__________________________________
|
FORM 39
|
(r. 36 (5))
|
|
KENYA VETERINARY BOARD INDEXING FORM
|
Institution name ......................................................................
Address..........................................................................
Telephone.................................................................
Presented by (Head of institution)...............................................
|
Name
|
ID No.
|
Address
|
Regstration No.
|
Course of study
|
Expected date of course beginning
|
Expected date of completion
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Attach ID card, copy of certificate and certified photo of all the students.
Signature.......................... Date..........................
__________________________________
|
FORM 40
|
(r. 38 (1))
|
|
APPLICATION FORM FOR REGISTRATION OF TNTERNS
|
The Registar,
The Kenya Veterinary Board,
Kabete.
I..............................of............................. hereby make an application for registration as a veterinary surgeon/veterinary para-professionals (mark as appropriate) intern.
My qualifications are:
..............................................................................................
..............................................................................................
Host organization ..................................................
Name of supervising veterinary surgeon
.......................................................................
I enclose the following:
| 1. |
Certificate/diploma/degree......................
|
| 2. |
Veterinary Surgeon's/Para-Professional's (mark as appropriate) oath as required by section 16 (3) of the Veterinary Surgeons and Veterinary Para-Professionals Act
Signature ......................Date ......................
|
__________________________________
|
FORM 41
|
(r. 38 (2))
|
|
CERTIFICATE OF REGISTRATION AS A VETERINARY INTERN
|
(Name).................................................
Registration No. ......................................
is hereby registered as a Veterinary Surgeon /Veterinary Para-Professional Intern (delete as appropriate).
Given this ..........................day of ........20...........
Valid up to .................................
Registrar, Kenya Veterinary Board
__________________________________
|
FORM 42
|
(r. 38 (6))
|
|
INTERNSHIP REPORT FORM DECLARATION BY SUPERVISOR
|
The Registrar
Kenya Veterinary Board
I (Full name in block letters) ...................................
Registration Number .................................
Hereby certify that (intern's full Name and refercnce intern registration).......................................
Has been undergoing internship training at .......................... (establishment) from.................to .................. (dates internship).
Being the internship Supervisor, I make the following observations:
.......................................................................
.......................................................................
.......................................................................
Recommended/not recommended
.......................................................................
Signature: .................... Official Stamp
Date:...................................
Signature:
Date:
Official Stamp
SECOND SCHEDULE [r.9(1)]
BOARD'S LOGO
THE VETERINARY SURGEONS AND VETERINARY PARA-PROFESSIONALS (CODE OF ETHICS) REGULATIONS
ARRANGEMENT OF REGULATIONS
| 3. |
Administration of Oath
|
| 5. |
Safeguarding welfare of animals
|
PART II – PROFESSIONALISM
| 6. |
Integrity, independence and impartiality
|
| 7. |
Update in relevant law and innovations
|
| 8. |
Professional indemnity
|
| 10. |
Competency to practice
|
| 11. |
Sourcing and handling of veterinary inputs
|
Conduct within the Profession
| 12. |
Collaboration with colleagues
|
| 13. |
Substitute veterinary surgeon or veterinary para-professionals
|
| 18. |
Relationship with client
|
| 20. |
Responsibility to client and para-professional
|
| 21. |
Responsibility of a veterinary para-professional to a client
|
PART III – HANDLING OF ANIMALS
| 22. |
Examination of patients
|
| 25. |
Use of animals in research and education
|
| 26. |
Transportation and processing of animals
|
PART IV – VETERINARY PREMISES AND PRACTICE
| 27. |
Professional premises
|
| 30. |
Ethics in prescribing, dispensing and administering, veterinary medicine
|
| 31. |
Control of unqualified persons
|
PART V – GENERAL PROVISIONS
| 33. |
Soliciting for professional work
|
| 35. |
Expert advice and report
|
| 36. |
Veterinary certificate
|
| 37. |
Recreational activities
|
| 39. |
Publicity and advertisement
|
| 42. |
Settling disputes within the profession
|
THE VETERINARY SURGEONS AND VETERINARY PARA-PROFESSIONALS (CODE OF ETHICS) REGULATIONS
| 1. |
Citation
This Code may be cited as the Code of Ethics for Veterinary Surgeons and Veterinary Para-professionals, 2015.
|
| 2. |
Interpretation
In this Code, unless the context otherwise requires—
"Act" means the Veterinary Surgeons and Veterinary Para-professionals Act, 2011 (No. 29 of 2011);
"area" means an area where a veterinary practice is licensed to operate and in which the veterinary surgeon or veterinary para-professional ordinarily operates;
"certification" means the issuance of certificate by a veterinary surgeon that attests to the health or condition of an animal or to a service rendered to an animal and "certificate" shall be construed accordingly;
"client" means—
| (a) |
the owner of an animal;
|
| (b) |
a person acting with the authority of the owner; or
|
| (c) |
a person with statutory or other legal authority over the animal;
|
"colleague" means a veterinary surgeon or a veterinary para-professional;
"direction" means instruction by a veterinary surgeon to a student, an intern or a veterinary para-professional as to a procedure, test or treatment to be administered on an animal, in the presence or absence of the veterinary surgeon;
"dope" means any medication administered to an animal for the purpose of enhancing performance of that animal in a competition;
"emergency" means a life threatening situation of an animal arising from illness, injury or other distress;
"ethical veterinary medicine" includes antibiotics, narcotics, hormones or vaccines;
"euthanasia" means the killing of an animal without subjecting the animal to pain, fear or discomfort in order to relieve that animal's suffering when is terminally ill, seriously injured or distressed;
"feral animal" means a domestic animal that has become wild and difficult to handle;
"fraud" includes using professional knowledge to grant an unfair advantage to the veterinary surgeon, veterinary para-professional or to deceive a client;
"international law" means any international convention or protocol which Kenya has ratified and includes the standards, Guidelines and recommendations of the World Health Organization.
"medical practitioner" has the same meaning as assigned to it under the Medical Practitioner and Dentists Act (Cap. 253);
"oath" means the veterinary surgeons or the veterinary para-professionals oath set out in the Third Schedule of the Veterinary Surgeons and Veterinary Paraprofessionals Act, 2011 (No. 29 of 2011);
"overdose" means using a veterinary medicine in a dose or frequency higher that the recommended level resulting to harm to an animal;
"patient" means an animal under the care of a veterinary surgeon or a veterinary para-professional;
"personal protection equipment" means any clothing or equipment used to protect the veterinary surgeon, veterinary para-professional or other staff in practice from possible contamination or injury while handling, examining or treating an animal;
"prescribed equipment" means the equipment approved by the Board for undertaking a procedure while treating or performing a surgical operation or used during veterinary training;
"raced" means an animal used for racing competition;
"referral" means where a veterinary surgeon sends a patient under his care to a second or subsequent referrals to a veterinary surgeon for further diagnosis and treatment with the objective of returning the patient to the original referring veterinary surgeon;
"specialized service" means a service provided by a veterinary specialist as defined in the Act;
"supervision" means instruction by a veterinary surgeon to a student, an intern or a veterinary para-professional, on a procedure, test or treatment while the veterinary surgeon is physically present during the administration of the procedure, test or treatment;
"veterinary-client-patient relationship" means a relationship arising from the rendering of an examination and preliminary diagnosis of an animal by a veterinary surgeon or veterinary para-professional, resulting in the veterinary surgeon or veterinary para-professional having sufficient knowledge of that animal;
"withholding period" means the period when an animal cannot be slaughtered due to drug residues in the body.
|
| 3. |
Administration of Oath
| (1) |
A veterinary surgeon or a veterinary para-professional shall before undertaking his profession take the Oath set out in the Third Schedule of the Act.
|
| (2) |
A veterinary surgeon or a veterinary para-professional shall uphold the Oath and this Code during his veterinary practice.
|
|
| 4. |
Guiding principles
The guiding principles for the ethical conduct of veterinary surgeons and veterinary para-professionals shall be to—
| (a) |
safeguard the welfare of the animal and alleviate animal suffering;
|
| (b) |
maintain professional conduct that promotes the reputation of the veterinary profession;
|
| (c) |
avoid any act that may in any way bring the image of the veterinary profession into disrepute;
|
| (d) |
handle clients and members of the public with respect, consideration and courtesy;
|
| (e) |
be conversant with, and abide by, all laws affecting the profession;
|
| (f) |
be well informed on veterinary matters keeping abreast of current knowledge and enhancing skills in order to ensure that expert knowledge, experience and judgment are available to the community;
|
| (g) |
report to the Board and the nearest law enforcement agency on any illegal or unauthorized veterinary practice;
|
| (h) |
promote the sale and use of registered veterinary medicines;
|
| (i) |
practice within one's area of competence and refer patients where necessary.
|
|
| 5. |
Safeguarding welfare of animals
It is the paramount duty of a veterinary surgeon and a veterinary para-professional to safeguard the welfare of an animal and alleviate animal suffering.
|
PART II – PROFESSIONALISM
General Conduct
| 6. |
Integrity, independence and impartiality
| (1) |
A veterinary surgeon or veterinary para-professional shall, after taking into consideration all relevant circumstances and without any application of personal interest or external influence, exercise professional and independent judgment.
|
| (2) |
A veterinary surgeon or a veterinary para-professional shall respect the need or requirement expressed by a client as long as the need or requirement does not conflict with this Code or any other law.
|
| (3) |
A veterinary surgeon or a veterinary para-professional shall not discriminate a client on grounds of race, age, ethnicity, gender, religion, political affiliation, disability or marital status.
|
|
| 7. |
Update in relevant law and innovations
A veterinary surgeon or veterinary para-professional shall ensure that he updates himself, on a regular basis, on the relevant local and international laws and any scientific and technological innovations related to the profession.
|
| 8. |
Professional indemnity
A veterinary surgeon may purchase a professional indemnity cover form an insurance company or undertake any other form of guarantee for purposes of safeguarding himself against any adverse effect resulting from errors or omissions made in the provision of a service.
|
| 9. |
Fraud
| (1) |
A veterinary surgeon or a veterinary para-professional shall not—
| (a) |
when engaged by either a seller or a buyer to examine an animal which is for sale or purchase, accept a fee from both the seller and the buyer;
|
| (b) |
perform surgical or medical procedures on any animal for the purpose of concealing genetic defects in an animal, which animal is to be displayed, raced, bred or sold;
|
| (c) |
give false information during the certification of an animal, or on the application or reporting of any test for disease in animals;
|
| (d) |
knowingly, make a false report of any contagious or infectious disease;
|
| (e) |
administer an expired veterinary medicine on an animal;
|
| (f) |
knowingly administer illegal veterinary medicine on an animal;
|
| (g) |
participate in the doping of an animal;
|
| (h) |
under-dose or over-dose an animal;
|
| (i) |
administer any unnecessary treatment, diagnostic or surgical procedures;
|
| (j) |
falsify or misrepresent a recommendation from a consultant or peer; or
|
| (k) |
commit or aid another to commit fraud, deceit or corruption in billing, payment or insurance reimbursement procedures.
|
|
| (2) |
A veterinary surgeon or a veterinary para-professional who violates the provisions of paragraph (1) commits an offence of fraud and in addition to any penalty imposed under any other law, shall be subject to disciplinary action under the Act.
|
|
| 10. |
Competency to practice
| (1) |
A veterinary surgeon or a veterinary para-professional shall, at the time of filling of an application for an annual practicing license, declare if they have in the last one year—
| (a) |
been charged with or found guilty of a criminal offence;
|
| (b) |
been suspended or dismissed, by employer who engaged him as a veterinary professional, on ethical grounds, negligence or incompetence;
|
| (c) |
resigned from employment as a veterinary professional for reasons relating to competency or ethics;
|
| (d) |
been subject to an adverse finding by the Kenya Veterinary Board.
|
|
| (2) |
A veterinary surgeon or a veterinary para-professional shall notify the Board of any issues specified under paragraph (1) at the time they arise.
|
| (3) |
A veterinary surgeon or veterinary para-professional shall immediately notify the Board of any health condition that may interfere with his professional work or endanger the health of an animal under his care.
|
| (4) |
A veterinary surgeon or a veterinary para-professional shall not practice veterinary medicine when his physical or mental ability is impaired—
| (b) |
by other physical, psychological or mental impediment.
|
|
| (5) |
It is the duty of a veterinary surgeon or veterinary paraprofessional to bring to the notice of the Board the impairment of a colleague in terms of paragraph (4), if in the opinion of that veterinary surgeon or veterinary para-professional the life of any animal or person is endangered due to the colleague's impairment.
|
| (6) |
The Board shall, after investigations and on receipt of a report by a medical practitioner have power to suspend or revoke the licence of a veterinary surgeon or veterinary para-professional who—
| (a) |
is addicted to alcohol or habit forming drugs;
|
| (b) |
is using controlled veterinary medicines on himself;
|
| (c) |
due to any physical or mental condition poses a danger to any person or animal;
|
| (d) |
has been subject to an adverse finding by a veterinary regulatory body;
|
| (e) |
has been charged with or found guilty of a criminal offence; or
|
| (f) |
has been suspended or dismissed on ethical grounds, negligence or incompetence.
|
|
| (7) |
A veterinary surgeon or a veterinary para-professional shall, on demand, divulge to the Board the means, methods, device, or equipment used in the treatment of a disease, injury, ailment or infirmity of an animal.
|
|
| 11. |
Sourcing and handling of veterinary inputs
| (1) |
A veterinary surgeon or veterinary para-professional shall—
| (a) |
source veterinary medicines, equipment and other relevant materials from reputable sources;
|
| (b) |
store a veterinary medicine as prescribed; and
|
| (c) |
not administer or dispense a veterinary medicine whose storage condition has been breached.
|
|
| (2) |
A veterinary surgeon or veterinary para-professional shall, at all times use prescribed equipment that is hygienically handled and stored.
|
| (3) |
A veterinary surgeon or veterinary para-professional practicing artificial insemination of animals shall ensure that prescribed standards are maintained in sourcing, handling, storage and transportation of semen.
|
| (4) |
Prior to carrying out any insemination under (a), a veterinary surgeon or veterinary para-professional shall conduct a thorough examination of the animal receiving the semen to ensure it is healthy, not pregnant and ready for insemination.
|
| (5) |
A veterinary surgeon conducting ova transplant or embryo transfer in any animal shall, in the sourcing, handling, storage and transportation of the ova or embryo, maintain the standards prescribed by the World Animal Health Organization.
|
|
Conduct within the Profession
| 12. |
Collaboration with colleagues
| (1) |
A veterinary surgeon or veterinary para-professional shall always seek to foster and maintain good understanding with and among colleagues and shall not—
| (a) |
by action or implication, ridicule a colleague;
|
| (b) |
slander or libel a colleague;
|
| (c) |
conduct or carry himself or act in a manner that is likely to bring a colleague and the veterinary profession into disrepute;
|
| (d) |
encourage dispute which may damage the reputation of the veterinary profession;
|
| (e) |
plagiarize the scholarly work of a colleague.
|
|
| (2) |
A veterinary surgeon or a veterinary para-professional who becomes aware of unprofessional or discreditable conduct or other misconduct by a colleague, shall attempt to counsel the colleague either directly or through the professional association.
|
| (3) |
If the behavior of the colleague specified under paragraph (2) does not change, the veterinary surgeon shall inform the Board.
|
|
| 13. |
Substitute veterinary surgeon or veterinary para-professionals
| (1) |
A veterinary surgeon or veterinary para-professional shall, where professional services may be required, at night, on a weekend, holiday or other off-duty hours, or by reason of, sickness or personal emergencies of any kind, make specific arrangements with a colleague to stand in on his behalf, for purposes of ensuring that emergency and first-aid services are available to clients.
|
| (2) |
Where first aid service has been given to an animal, a system for assessment and follow-up of cases shall be put in place at the veterinary practice.
|
| (3) |
A veterinary practice, a veterinary surgeon or veterinary para-professional shall, if called to stand in on behalf of a colleague, return to the care of that colleague on his resumption of practice, any animal attended to during his absence.
|
| (4) |
A veterinary surgeon or veterinary para-professional who stands in for a colleague shall not do anything that may be construed as an attempt to attract or entice a client, from the original veterinary surgeon or veterinary para-professional, to his veterinary practice.
|
| (5) |
An animal shall be considered to be under the immediate care of a veterinary surgeon or veterinary para-professional if—
| (a) |
the animal is under treatment by the veterinary surgeon or veterinary para-professional;
|
| (b) |
the veterinary surgeon or veterinary para-professional has been given and has accepted responsibility for the health of the animal; or
|
| (c) |
the ongoing and continuing care of the animal is recognized as a veterinary-client-patient relationship in terms of these Regulations.
|
|
|
| 14. |
Referrals
| (1) |
A veterinary surgeon shall, when a case or a treatment option is outside their area of competence, refer it to an expert whom they are satisfied is competent to carry out the investigation or treatment required.
|
| (2) |
A veterinary surgeon making a referral in accordance with (1), has a responsibility to ensure that the client is made aware of the recognized specialists or experts available in the area of competence in which the referral is made.
|
| (3) |
The veterinary surgeon referring a client and the expert to whom the client is referred to, shall ensure that the client understands the cost arising from the referral.
|
| (4) |
A veterinary surgeon referring a client as provided under paragraph (1) shall inform the client of all the experts who may be found within the client's area of residence and upon which the client may make an appointment with the expert of his choice.
|
| (5) |
A veterinary surgeon referring a client shall, on request, within one hour of the request, provide the expert with the case history of the animal and any further information that may be requested.
|
| (6) |
An expert or specialist to whom a case has been referred shall maintain professionalism when stating their opinion so it does not unduly or wrongly discredit the veterinary surgeon that has referred the client.
|
|
| 15. |
Second opinion
| (1) |
A veterinary surgeon or a client may request a second opinion on diagnosis or treatment of an animal from another veterinary surgeon.
|
| (2) |
A veterinary para-professional or his client may request a second opinion on the treatment and examination of an animal from a veterinary surgeon in the area of practice allowed in the Act.
|
| (3) |
A veterinary surgeon or veterinary para-professional shall, after seeking a second opinion in accordance with these Regulations, retain the continued responsibility of the animal.
|
| (4) |
A veterinary surgeon or a veterinary para-professional shall recognize and respect the right of a client to a second opinion and shall facilitate a client's request for a second opinion.
|
| (5) |
A veterinary surgeon shall, while providing a second opinion, state their opinion in a way which does not unduly or wrongly discredit the veterinary surgeon seeking that opinion.
|
|
| 16. |
Supersession
| (1) |
A veterinary surgeon who is called upon to attend to a patient by an owner in an ongoing case that has been under the care of another veterinary surgeon, shall, before proceeding with the treatment of the animal, endeavour to establish whether the first attending veterinary surgeon has retired from the case, or has been told by the owner that his services are no longer required.
|
| (2) |
The provisions of paragraph (1) shall not apply in case of—
| (ii) |
a second opinion or referral. |
|
| (3) |
A veterinary surgeon invited to attend to an animal under paragraph (1), shall liaise with the first attending veterinary surgeon for purposes of obtaining the history of the case.
|
| (4) |
Where a veterinary surgeon is invited to take over the whole of the veterinary work from another member of the veterinary profession, the provisions of paragraph (1) shall apply.
|
| (5) |
A veterinary surgeon is duty bound to accept and take up a case which has been interfered with by an unqualified person, provided that the client is made aware of the likelihood of poor clinical outcome for which the veterinary surgeon may not take full responsibility.
|
| (6) |
This regulation shall apply to a veterinary para-professional with respect to services allowed under the Act.
|
|
| 17. |
Specialized service
A veterinary surgeon providing specialized services shall contact the client's usual veterinary surgeon in advance and keep him fully informed, while making prior arrangements for continuation of the service.
|
Interaction with clients
| 18. |
Relationship with client
| (1) |
A veterinary surgeon or veterinary para-professional, providing veterinary services, shall create a contractual relationship between himself and the client.
|
| (2) |
A veterinary surgeon or veterinary para-professional shall—
| (a) |
display his current professional identification card to a client when called upon to offer a service;
|
| (b) |
maintain a professional relationship with a client at all times and avoid any conduct or relationship that may bring disrepute to the profession;
|
| (c) |
ensure that clear information is provided to the client about practice arrangements, the services available, initial costs, location of the out-of-hours emergency service, and information on the care of any in-patients;
|
| (d) |
maintain clear, accurate and comprehensive patients' records and accounts;
|
| (e) |
give reasonable fee estimates based on treatment options;
|
| (f) |
keep the client informed of the patient's progress, and of any escalation or reduction in costs once treatment has started;
|
| (g) |
ensure that the client is made aware of any procedures to be performed by a staff in the practice who is not a veterinary surgeon;
|
| (h) |
recognize that a client has the freedom to choose the veterinary surgeon who shall take care of his animal; and
|
| (i) |
uphold a client's right to receive impartial, independent, honest and objective advice.
|
|
| (3) |
A veterinary surgeon or veterinary para-professional shall as far as possible provide to the client copies of the records of an animal under his care.
|
| (4) |
A veterinary surgeon or veterinary para-professional shall not intentionally or recklessly cause physical or emotional harm to a client or patient.
|
| (5) |
A veterinary surgeon shall inform clients of any human health implications arising from the condition, care, tests or treatment of their animals, with special emphasis on those persons who may be more at risk.
|
|
| 19. |
Informed consent
| (1) |
A veterinary surgeon or veterinary para-professional shall acquire the informed consent of a client by explaining to the client the proposed treatment options, the risks and the significance to the patient and the fee estimates.
|
| (2) |
A veterinary surgeon or veterinary para-professional shall satisfy himself that the person giving consent under paragraph (1) has the authority to provide that consent.
|
| (3) |
A veterinary surgeon or veterinary para-professional, shall where the owner of an animal under treatment is not known, make every reasonable effort to locate the owner.
|
| (4) |
A veterinary surgeon or veterinary para-professional shall—
| (a) |
use the simple language to communicate with a client and explain any technical terminology that the client may not understand;
|
| (b) |
seek to ensure that he understands the client and that client also understands the veterinary surgeon or veterinary para-professional; and
|
| (c) |
encourage a client to take full part in any discussion related to the patient.
|
|
| (5) |
A veterinary surgeon or veterinary para-professional shall—
| (a) |
use consent forms to record the informed consent of a client obtained for the purpose of carrying out procedures of euthanasia, anesthesia and surgery; and
|
| (b) |
as far as possible, use consent forms in all other cases.
|
|
| (6) |
A consent form under this regulation shall include the following information—
| (b) |
identification of the animal;
|
| (c) |
treatment or procedure being undertaken;
|
| (d) |
name, signature and registration number of attending veterinary surgeon;
|
| (e) |
signed consent of the client; and
|
|
| (7) |
If a veterinary surgeon or veterinary para-professional is unable to obtain a client's consent in accordance with this provision, he shall indicate, in the report, the measures taken to try and obtain that consent and any action taken on the patient.
|
| (8) |
A copy of the duly filled and signed consent form shall be shall be provided to the client.
|
|
| 20. |
Responsibility to client and para-professional
| (1) |
A veterinary surgeon has a legal and moral responsibility to the client to—
| (a) |
personally examine an animal before a veterinary para-professional carries out any procedure, except in circumstances permitted under the Act;
|
| (b) |
direct, control and supervise a veterinary para-professional and ensure the work is conducted in the proper method and manner;
|
| (c) |
authorize or require that a veterinary para-professional to perform only the work as the veterinary surgeon knows that the veterinary para-professional is capable of performing.
|
|
| (2) |
A veterinary surgeon shall not staff a veterinary practice with veterinary para-professionals unless the necessary supervision or direction of a veterinary surgeon is also provided in that practice.
|
|
| 21. |
Responsibility of a veterinary para-professional to a client
A veterinary para-professional has a responsibility to the client to—
| (a) |
undertake only the treatment, procedure or other action as the Act authorizes or as directed by a veterinary surgeon;
|
| (b) |
perform his work only in the manner authorized under the Act or as directed by the veterinary surgeon;
|
| (c) |
employ the best care and skill in performing all work undertaken;
|
| (d) |
refrain from undertaking any work which he is not capable of performing satisfactorily; and
|
| (e) |
refrain from diagnosing, making a prognosis, prescribing, initiating treating or performing surgery other than that prescribed under the Act.
|
|
PART III – HANDLING OF ANIMALS
| 22. |
Examination of patients
| (1) |
A veterinary surgeon or veterinary para-professional shall at the time of making a provisional or other diagnosis physically examine a patient.
|
| (2) |
A veterinary surgeon shall physically examine an animal before giving a prescription of veterinary medicine.
|
|
| 23. |
Euthanasia
| (1) |
A veterinary surgeon, veterinary para-professional or veterinary practice may, in appropriate cases, carry out euthanasia to relieve the suffering of an animal.
|
| (2) |
Despite paragraph (1), a veterinary para-professional and a veterinary practice shall only carry out euthanasia under the supervision of a veterinary surgeon.
|
| (3) |
It is the responsibility of a veterinary professional under this regulation, to ensure the procedure and veterinary medicine used, are as guided by the standards, guidelines and recommendations of the World Health Organization.
|
| (4) |
All staff in a veterinary practice involved in euthanasia shall be fully trained and planned, rehearsed and coordinated approach shall be taken while carrying out the euthanasia.
|
| (5) |
A veterinary surgeon or a veterinary para-professional is not obligated to undertake euthanasia on a healthy animal unless required to do so under statutory powers as part of the conditions of employment.
|
| (6) |
A veterinary surgeon facing difficulties including but not limited to—
| (a) |
an owner who desires to have a perfectly healthy or treatable animal to be euthanized; or
|
| (b) |
an owner who wishes to keep an animal alive in circumstances where euthanasia would be the kindest course of action;
|
| (c) |
shall keep in mind that his primary obligation is to relieve the suffering of that animal.
|
|
| (7) |
Where the client is not in agreement with the advice given under paragraph (6), the veterinary surgeon shall make efforts to direct the client to alternative sources of advice, while recognizing the extreme sensitivity of the situation.
|
| (8) |
A veterinary surgeon shall, when euthanizing a feral or wild animal, ensure that the procedure is carried out as humanely as possible, with due consideration to the particular animal and to the group from which it is drawn.
|
| (9) |
A person who is of the opinion that a veterinary surgeon has not exhibited professional competence during euthanasia may make a complaint to the Board.
|
|
| 24. |
Ethics in teaching
| (1) |
A veterinary surgeon or veterinary para-professional engaged in teaching shall, by instruction and example—
| (a) |
emphasize the importance of humane treatment of all animals;
|
| (b) |
ensure that animals used for teaching purposes are handled humanely and are not subjected to excessive manipulation which may injure or distress the animal;
|
| (c) |
ensure that the highest possible standards of ethical conduct are promoted and maintained in all aspects of professional activity;
|
| (d) |
adhere to national and the World Organization for Animal Health guidelines on the use of animals in education.
|
|
| (2) |
A veterinary surgeon or veterinary para-professional shall teach only approved programmes in education facilities approved by the Board.
|
|
| 25. |
Use of animals in research and education
| (1) |
A veterinary surgeon or veterinary para-professional involved in research using animals shall—
| (a) |
adhere with the national legislation and the World Organization for Animal Health guidelines on use of animals in research; and
|
| (b) |
ensure that the highest standards of management, handling and analgesia are used at all times.
|
|
| (2) |
Without prejudice to paragraph (1) a veterinary surgeon or veterinary para-professional shall ensure that—
| (a) |
an animal is only used in cases where there are no alternative methods, including utilization of cells, tissues or organs of animals, may be used to obtain similar results;
|
| (b) |
care is taken to use fewer animals to obtain comparable levels of information or to obtain more information from the same number of animals; and
|
| (c) |
the methods used prevent, alleviate or minimize pain, suffering, distress or lasting harm and enhance welfare of the animal used.
|
|
| (3) |
A veterinary surgeon or veterinary para-professional shall undertake regular training to update themselves on the humane handling of animals.
|
| (4) |
A veterinary surgeon or veterinary para-professional shall actively discourage research work using animals where the research is unnecessarily repetitive or has ill-defined goals and shall, whenever possible, encourage the substitution of in-vivo with in-vitro techniques.
|
|
| 26. |
Transportation and processing of animals
| (1) |
A veterinary surgeon or veterinary para-professional shall be responsible for ensuring compliance with the welfare requirements, relevant national and international laws and the proper handling of an animal being transported.
|
| (2) |
A veterinary surgeon and veterinary para-professional shall ensure that an animal destined for an animal product processing industry is transported and slaughtered in a humane manner.
|
|
PART IV – VETERINARY PREMISES AND PRACTICE
| 27. |
Professional premises
A veterinary professional premises shall be located—
| (a) |
away from premises that may not reflect the good standing of the profession including gambling shops and places of public entertainment; or
|
| (b) |
in an area where animals being attended at the veterinary practice are not likely to pose any danger or discomfort to the general public and vice versa.
|
|
| 28. |
Environmental health
| (1) |
A veterinary surgeon or a veterinary para-professional shall work and dispose waste in the manner as to reduce pollution from disinfectants, medicinal products, chemicals and biological waste.
|
| (2) |
Waste produced at a veterinary practice shall be disposed in a manner that does not pose a danger to the environment or to any person or animal that may come into contact with the waste.
|
|
| 29. |
Employment
| (1) |
A veterinary surgeon or veterinary para-professional employed by a person or an institution, by virtue of their professional training and qualifications shall conduct himself or herself with professional dignity and decorum.
|
| (2) |
A practicing veterinary surgeon shall not, in matters of the veterinary profession, be subject to the direction or control of a veterinary para-professional or a lay person.
|
| (3) |
A veterinary surgeon responsible to another Board or committee shall regard it as a professional responsibility to advise the board or committee on all matters of veterinary professional ethics.
|
| (4) |
A veterinary surgeon or veterinary para-professional shall comply with the recommendations of this Code, and be careful to not to gain unfair advantage over any other colleague or veterinary practice because of the particular advantages the structure of the employing organization may allow.
|
| (5) |
Where an employing organization fails to follow professional advice resulting or which may result in a breach of the ethical requirements of the veterinary profession, the veterinary surgeon or veterinary para-professional shall immediately notify the Board.
|
| (6) |
A veterinary surgeon or a veterinary para-professional practicing in a food and feed plant shall ensure that he or she is conversant with the codes of practice relevant to the food or feed in question and shall only certify that the food or feed is fit for human and animal consumption based on scientific principles and the existing law.
|
| (7) |
A veterinary surgeon or a veterinary para-professional practicing in a veterinary laboratory shall update his knowledge of, and ensure observation of the law relating to the bio-safety and bio-containment.
|
| (8) |
A veterinary surgeon practicing in a veterinary laboratory shall only release results of tests he or she has conducted or supervised.
|
|
| 30. |
Ethics in prescribing, dispensing and administering, veterinary medicine
| (1) |
A veterinary surgeon prescribing a veterinary medicine to a patient shall do so in writing and shall indicate—
| (a) |
the correct method of use;
|
| (b) |
route or manner of administration;
|
| (d) |
special precautions relevant to that veterinary medicine.
|
|
| (2) |
A veterinary surgeon dispensing a veterinary medicine to a client or directing a veterinary para-professional on the use of a veterinary medicine shall ensure that the client or the veterinary para-professional is aware of the details specified under paragraph (1) (a), (b), (c) and (d).
|
| (3) |
A veterinary surgeon shall inform a client of the safety, efficacy and side effects of a veterinary medicine at the time of dispensing the medicine.
|
| (4) |
A veterinary surgeon who supplies a restricted veterinary medicine to a client to use on that client's animal or who directs a veterinary para-professional to use a restricted veterinary medicine bears full responsibility for the use of that medicine and any adverse reactions that may result.
|
| (5) |
A veterinary surgeon may only prescribe a veterinary medicine for a patient with whom there exists a veterinary-client-patient relationship.
|
| (6) |
A veterinary surgeon and a veterinary para-professional shall protect veterinary medicine from misuse.
|
| (7) |
Without prejudice to paragraph (5), a veterinary surgeon shall promote a veterinary practice that reduces the need to use antibiotics through—
| (a) |
maintaining the highest standards of hygiene in animal establishments;
|
| (b) |
use of vaccines instead of antibiotics;
|
| (c) |
where applicable, avoiding the use of antibiotics for preventive purposes;
|
| (d) |
encouraging the use of pro-biotics; and
|
| (e) |
conducting sensitivity tests, as much as possible, to inform on the most effective antibiotic for a particular infection.
|
|
|
| 31. |
Control of unqualified persons
| (1) |
A veterinary surgeon, whether or not physically present, is ultimately responsible for the outcome and the technical competence of anyone to whom he has delegated veterinary procedures or other veterinary duties.
|
| (2) |
A veterinary student gaining experience as part of his veterinary medicine or animal health course may examine animals and carry out diagnostic tests under the supervision of a veterinary surgeon.
|
| (3) |
A veterinary graduate on internship may—
| (a) |
examine animals, carry out diagnostic tests and administer treatment under the direction of a veterinary surgeon; or
|
| (b) |
perform surgical operations under the supervision of a veterinary surgeon.
|
|
| (4) |
A veterinary surgeon or a veterinary para-professional shall not engage in any act that aids or abets the unlicensed practice of veterinary medicine including the delegation of duties to unregistered staff or allowing unregistered staff to influence or engage in the practice of veterinary medicine.
|
| (5) |
A veterinary surgeon in-charge of a veterinary practice shall apply fair labour and employment practices as guided by the laws relating to labour.
|
|
PART V – GENERAL PROVISIONS
| 32. |
Conflict of interest
| (1) |
A veterinary surgeon or a veterinary para-professional shall refrain from being involved in any situation where conflict of interest may arise.
|
| (2) |
A veterinary surgeon or veterinary para-professional shall not undertake a business which may give rise to conflict of interest or call into question the integrity of the profession.
|
| (3) |
Without prejudice to the provisions of paragraph (2), a veterinary surgeon shall not provide certification or valuation of an animal or valuation where the veterinary surgeon is the owner or the breeder of that animal.
|
|
| 33. |
Soliciting for professional work
A veterinary surgeon or a veterinary para-professional who, with a view to obtaining professional work, makes or instigates an unsolicited approach to an organization, person or persons who are not bona fide clients, shall ensure that the approach—
| (a) |
does not in any way bring the veterinary profession into disrepute;
|
| (b) |
is in a manner which cannot be considered by the recipient to be intrusive, offensive or harassing;
|
| (c) |
provides information that is factual; and
|
| (d) |
does not include claims or implications of personal superiority over any other veterinary surgeon or veterinary para-professional.
|
|
| 34. |
Confidentiality
| (1) |
A veterinary surgeon or veterinary para-professional shall treat all information concerning a client or an animal, that arises out of his care, attention or treatment, as confidential and the information shall, unless there is a legal obligation to do so, not be divulged to any other person outside the practice without the owner's consent.
|
| (2) |
Despite paragraph (1), where members of the public or animals are endangered, and a veterinary surgeon or veterinary paraprofessional determines that there is an obligation to divulge any information which has come to his attention in the course of his professional work, the cooperation of the client shall, if possible, be obtained before taking action to divulge the information.
|
|
| 35. |
Expert advice and report
| (1) |
A veterinary surgeon shall give evidence impartially and in the interest of justice where—
| (a) |
required to write an expert witness statement, opinion or report; or
|
| (b) |
is summoned or called upon in a professional capacity to give expert evidence in a court of law.
|
|
| (2) |
A veterinary surgeon shall prepare and present expert advice, whether serving as an expert witness or in any other capacity, in a thorough and professional manner which shall include the—
| (a) |
identification of the animal or herd;
|
|
| (3) |
Without prejudice to the provisions of paragraph (2), where a veterinary surgeon is required to divulge information in circumstances where the disclosure—
| (a) |
concerns public safety, consumer health and welfare; or
|
| (b) |
is required by law or as a witness in court,
|
a veterinary surgeon shall divulge only that information which is relevant.
|
| (4) |
A veterinary surgeon shall take care to differentiate factual information and personal opinion.
|
|
| 36. |
Veterinary certificate
| (1) |
A certificate or other document issued by a veterinary surgeon in accordance with any legal requirement, shall—
| (a) |
be carefully compiled, completed and shall be accurate in every detail;
|
| (b) |
bear the name, signature and registration number of the veterinary surgeon;
|
| (c) |
ensure all necessary steps are taken to ascertain that the matters certified are true; and
|
| (d) |
include details relating to the animal which is the subject of the certificate or document including—
| (ii) |
details of ownership; |
| (iii) |
locality where examination was done; |
| (vii) |
color of the animal; and |
| (viii) |
tattoos, tags notches, microchip or other identification marks. |
|
|
| (2) |
A veterinary surgeon shall attach any other relevant documentation to the certificate or document issued under paragraph (1).
|
| (3) |
A veterinary surgeon shall not sign a vaccination certificate unless he has administered the vaccination personally or has supervised the administration of the vaccine by a veterinary para-professional.
|
|
| 37. |
Recreational activities
| (1) |
Subject to paragraph (1), a veterinary surgeon or a veterinary para-professional may engage in social and recreational activities including arts and sports.
|
| (2) |
Despite paragraph (1), a veterinary surgeon or a veterinary para-professional may engage in social and recreational activities including arts and sports where the activities do not adversely affect the dignity of the profession or interfere with the performance of his professional duties.
|
| (3) |
A veterinary surgeon or veterinary para-professional may make a useful contribution to the promotion of animal welfare and responsible pet ownership by taking part in public life, whether in national or local politics, community service, or involvement with the media and in so doing ensure he distinguishes between personal opinion, political belief and established facts.
|
|
| 38. |
Communication
| (1) |
A veterinary surgeon or veterinary para-professional shall advise the staff in his veterinary practice to communicate any concerns raised by a client or misunderstanding, to the management of the practice and shall ensure that the concerns are addressed and the client is kept fully informed.
|
| (2) |
A veterinary surgeon or veterinary para-professional who is part of a veterinary team or teams in different veterinary practices shall always endeavor to work collaboratively and harmoniously with team members, show respect and ensure effective communication with other members of the team.
|
|
| 39. |
Publicity and advertisement
| (1) |
For purposes of this regulations "to endorse" includes the wearing of a 'tee shirt' or other attire, writing a prescription on a headed pad, or other action that promotes directly or indirectly a particular medicine or drug manufacturer through the use of a trade mark or logo.
|
| (2) |
A veterinary surgeon or veterinary para-professional shall not endorse an ethical veterinary medicine or service but may endorse non-medical products and services provided the endorsement does not bring the profession into disrepute.
|
| (3) |
A veterinary surgeon or veterinary para-professional shall ensure that any publicity or advertising he undertakes—
| (a) |
is factual and informative to the clients and the general public;
|
| (b) |
does not exploit a client's lack of veterinary knowledge;
|
| (c) |
is not of a character likely to bring the profession into disrepute;
|
| (d) |
whether by telephone or visit, cannot be construed as being solicitation for clients.
|
|
| (4) |
A veterinary practice may, as a means of communication, release newsletters and circulars to clients and may include comments about animal remedy.
|
|
| 40. |
Ethics in the media
| (1) |
A veterinary surgeon or veterinary para-professional shall in the use of all forms of media, demonstrate high standards of sensitivity, etiquette and responsibility and shall not use language, pictures or other thing that may bring disrepute to a colleague or to the profession.
|
| (2) |
A veterinary surgeon or a veterinary para-professional shall, when communicating matters of a professional nature or of concern to the profession, through print or social media, identify himself by his name as registered by the Board.
|
|
| 41. |
Dress code
| (1) |
A veterinary surgeon or veterinary para-professional shall adopt an appropriate dress code and grooming that brings honour and dignity to the profession.
|
| (2) |
A veterinary practice or a veterinary institution shall provide uniform and other protective clothing for its staff.
|
| (3) |
A veterinary surgeon or a veterinary para-professional shall at all times use personal protection equipment or gear while at the work place.
|
|
| 42. |
Settling disputes within the profession
| (1) |
A veterinary surgeon or veterinary para-professional shall attempt by all means possible, to settle a professional dispute within the profession in the manner so as to avoid any recourse to publicity that may bring the profession into disrepute.
|
| (2) |
A veterinary surgeon or veterinary para-professional involved in a dispute with a colleague may approach the Board for advice.
|
| (3) |
A client or a member of the public who is dissatisfied with a decision or the conduct of a veterinary surgeon or veterinary para-professional may register the dissatisfaction with the Board and request appropriate action be taken against the veterinary surgeon or veterinary para-professional.
|
|
| 43. |
Discipline
A veterinary surgeon or veterinary para-professional in breach of this code is guilty of professional misconduct and shall be subjected to disciplinary proceedings in accordance with Part VI of the Act.
|
THE VETERINARY SURGEONS AND VETERINARY PARA-PROFESSIONALS (CODE OF ETHICS) REGULATIONS
| 1. |
Citation
This Code may be cited as the Code of Ethics for Veterinary Surgeons and Veterinary Para-Professionals.
|
| 2. |
Interpretation
In this Code, unless the context otherwise requires—
"Act" means the Veterinary Surgeons and Veterinary Para-Professionals Act (Cap. 366);
"area" means an area where a veterinary practice is licensed to operate and in which the veterinary surgeon or veterinary para-professional ordinarily operates;
"certification" means the issuance of certificate by a veterinary surgeon that attests to the health or condition of an animal or to a service rendered to an animal and "certificate" shall be construed accordingly;
"client" means—
| (b) |
a person acting with the authority of the owner; or
|
| (c) |
a person with statutory or other legal authority over the animal;
|
"colleague" means a veterinary surgeon or a veterinary para-professional;
"direction" means instruction by a veterinary surgeon to a student, an intern or a veterinary para-professional as to a procedure, test or treatment to be administered on an animal, in the presence or absence of the veterinary surgeon;
"dope" means any medication administered to an animal for the purpose of enhancing performance of that animal in a competition;
"emergency" means a life threatening situation of an animal arising from illness, injury or other distress;
"ethical veterinary medicine" includes antibiotics, narcotics, hormones or vaccines;
"euthanasia" means the killing of an animal without subjecting the animal to pain, fear or discomfort in order to relieve that animal's suffering when is terminally ill, seriously injured or distressed;
"feral animal" means a domestic animal that has become wild and difficult to handle;
"fraud" includes using professional knowledge to grant an unfair advantage to the veterinary surgeon, veterinary para-professional or to deceive a client;
"international law" means any international convention or protocol which Kenya has ratified and includes the standards, Guidelines and recommendations of the World Health Organization.
"medical practitioner" has the same meaning as assigned to it under the Medical Practitioner and Dentists Act (Cap. 253);
"oath" means the veterinary surgeons or the veterinary para-professionals oath set out in the Third Schedule of the Veterinary Surgeons and Veterinary Paraprofessionals Act (Cap. 366);
"overdose" means using a veterinary medicine in a dose or frequency higher that the recommended level resulting to harm to an animal;
"patient" means an animal under the care of a veterinary surgeon or a veterinary para-professional;
"personal protection equipment" means any clothing or equipment used to protect the veterinary surgeon, veterinary para-professional or other staff in practice from possible contamination or injury while handling, examining or treating an animal;
"prescribed equipment" means the equipment approved by the Board for undertaking a procedure while treating or performing a surgical operation or used during veterinary training;
"raced" means an animal used for racing competition;
"referral" means where a veterinary surgeon sends a patient under his care to a second or subsequent referrals to a veterinary surgeon for further diagnosis and treatment with the objective of returning the patient to the original referring veterinary surgeon;
"specialized service" means a service provided by a veterinary specialist as defined in the Act;
"supervision" means instruction by a veterinary surgeon to a student, an intern or a veterinary para-professional, on a procedure, test or treatment while the veterinary surgeon is physically present during the administration of the procedure, test or treatment;
"veterinary-client-patient relationship" means a relationship arising from the rendering of an examination and preliminary diagnosis of an animal by a veterinary surgeon or veterinary para-professional, resulting in the veterinary surgeon or veterinary para-professional having sufficient knowledge of that animal;
"withholding period" means the period when an animal cannot be slaughtered due to drug residues in the body.
|
| 3. |
Administration of Oath
| (1) |
A veterinary surgeon or a veterinary para-professional shall before undertaking his profession take the Oath set out in the Third Schedule of the Act.
|
| (2) |
A veterinary surgeon or a veterinary para-professional shall uphold the Oath and this Code during his veterinary practice.
|
|
| 4. |
Guiding principles
The guiding principles for the ethical conduct of veterinary surgeons and veterinary para-professionals shall be to—
| (a) |
safeguard the welfare of the animal and alleviate animal suffering;
|
| (b) |
maintain professional conduct that promotes the reputation of the veterinary profession;
|
| (c) |
avoid any act that may in any way bring the image of the veterinary profession into disrepute;
|
| (d) |
handle clients and members of the public with respect, consideration and courtesy;
|
| (e) |
be conversant with, and abide by, all laws affecting the profession;
|
| (f) |
be well informed on veterinary matters keeping abreast of current knowledge and enhancing skills in order to ensure that expert knowledge, experience and judgment are available to the community;
|
| (g) |
report to the Board and the nearest law enforcement agency on any illegal or unauthorized veterinary practice;
|
| (h) |
promote the sale and use of registered veterinary medicines;
|
| (i) |
practice within one's area of competence and refer patients where necessary.
|
|
| 5. |
Safeguarding welfare of animals
It is the paramount duty of a veterinary surgeon and a veterinary para-professional to safeguard the welfare of an animal and alleviate animal suffering.
|
PART II – PROFESSIONALISM
| 6. |
Integrity, independence and impartiality
| (1) |
A veterinary surgeon or veterinary para-professional shall, after taking into consideration all relevant circumstances and without any application of personal interest or external influence, exercise professional and independent judgment.
|
| (2) |
A veterinary surgeon or a veterinary para-professional shall respect the need or requirement expressed by a client as long as the need or requirement does not conflict with this Code or any other law.
|
| (3) |
A veterinary surgeon or a veterinary para-professional shall not discriminate a client on grounds of race, age, ethnicity, gender, religion, political affiliation, disability or marital status.
|
|
| 7. |
Update in relevant law and innovations
A veterinary surgeon or veterinary para-professional shall ensure that he updates himself, on a regular basis, on the relevant local and international laws and any scientific and technological innovations related to the profession.
|
| 8. |
Professional indemnity
A veterinary surgeon may purchase a professional indemnity cover form an insurance company or undertake any other form of guarantee for purposes of safeguarding himself against any adverse effect resulting from errors or omissions made in the provision of a service.
|
| 9. |
Fraud
| (1) |
A veterinary surgeon or a veterinary para-professional shall not—
| (a) |
when engaged by either a seller or a buyer to examine an animal which is for sale or purchase, accept a fee from both the seller and the buyer;
|
| (b) |
perform surgical or medical procedures on any animal for the purpose of concealing genetic defects in an animal, which animal is to be displayed, raced, bred or sold;
|
| (c) |
give false information during the certification of an animal, or on the application or reporting of any test for disease in animals;
|
| (d) |
knowingly, make a false report of any contagious or infectious disease;
|
| (e) |
administer an expired veterinary medicine on an animal;
|
| (f) |
knowingly administer illegal veterinary medicine on an animal;
|
| (g) |
participate in the doping of an animal;
|
| (h) |
under-dose or over-dose an animal;
|
| (i) |
administer any unnecessary treatment, diagnostic or surgical procedures;
|
| (j) |
falsify or misrepresent a recommendation from a consultant or peer; or
|
| (k) |
commit or aid another to commit fraud, deceit or corruption in billing, payment or insurance reimbursement procedures.
|
|
| (2) |
A veterinary surgeon or a veterinary para-professional who violates the provisions of paragraph (1) commits an offence of fraud and in addition to any penalty imposed under any other law, shall be subject to disciplinary action under the Act.
|
|
| 10. |
Competency to practice
| (1) |
A veterinary surgeon or a veterinary para-professional shall, at the time of filling of an application for an annual practicing license, declare if they have in the last one year—
| (a) |
been charged with or found guilty of a criminal offence;
|
| (b) |
been suspended or dismissed, by employer who engaged him as a veterinary professional, on ethical grounds, negligence or incompetence;
|
| (c) |
resigned from employment as a veterinary professional for reasons relating to competency or ethics;
|
| (d) |
been subject to an adverse finding by the Kenya Veterinary Board.
|
|
| (2) |
A veterinary surgeon or a veterinary para-professional shall notify the Board of any issues specified under paragraph (1) at the time they arise.
|
| (3) |
A veterinary surgeon or veterinary para-professional shall immediately notify the Board of any health condition that may interfere with his professional work or endanger the health of an animal under his care.
|
| (4) |
A veterinary surgeon or a veterinary para-professional shall not practice veterinary medicine when his physical or mental ability is impaired—
| (b) |
by other physical, psychological or mental impediment.
|
|
| (5) |
It is the duty of a veterinary surgeon or veterinary paraprofessional to bring to the notice of the Board the impairment of a colleague in terms of paragraph (4), if in the opinion of that veterinary surgeon or veterinary para-professional the life of any animal or person is endangered due to the colleague's impairment.
|
| (6) |
The Board shall, after investigations and on receipt of a report by a medical practitioner have power to suspend or revoke the licence of a veterinary surgeon or veterinary para-professional who—
| (a) |
is addicted to alcohol or habit forming drugs;
|
| (b) |
is using controlled veterinary medicines on himself;
|
| (c) |
due to any physical or mental condition poses a danger to any person or animal;
|
| (d) |
has been subject to an adverse finding by a veterinary regulatory body;
|
| (e) |
has been charged with or found guilty of a criminal offence; or
|
| (f) |
has been suspended or dismissed on ethical grounds, negligence or incompetence.
|
|
| (7) |
A veterinary surgeon or a veterinary para-professional shall, on demand, divulge to the Board the means, methods, device, or equipment used in the treatment of a disease, injury, ailment or infirmity of an animal.
|
|
| 11. |
Sourcing and handling of veterinary inputs
| (1) |
A veterinary surgeon or veterinary para-professional shall—
| (a) |
source veterinary medicines, equipment and other relevant materials from reputable sources;
|
| (b) |
store a veterinary medicine as prescribed; and
|
| (c) |
not administer or dispense a veterinary medicine whose storage condition has been breached.
|
|
| (2) |
A veterinary surgeon or veterinary para-professional shall, at all times use prescribed equipment that is hygienically handled and stored.
|
| (3) |
A veterinary surgeon or veterinary para-professional practicing artificial insemination of animals shall ensure that prescribed standards are maintained in sourcing, handling, storage and transportation of semen.
|
| (4) |
Prior to carrying out any insemination under (a), a veterinary surgeon or veterinary para-professional shall conduct a thorough examination of the animal receiving the semen to ensure it is healthy, not pregnant and ready for insemination.
|
| (5) |
A veterinary surgeon conducting ova transplant or embryo transfer in any animal shall, in the sourcing, handling, storage and transportation of the ova or embryo, maintain the standards prescribed by the World Animal Health Organization.
|
|
| 12. |
Collaboration with colleagues
| (1) |
A veterinary surgeon or veterinary para-professional shall always seek to foster and maintain good understanding with and among colleagues and shall not—
| (a) |
by action or implication, ridicule a colleague;
|
| (b) |
slander or libel a colleague;
|
| (c) |
conduct or carry himself or act in a manner that is likely to bring a colleague and the veterinary profession into disrepute;
|
| (d) |
encourage dispute which may damage the reputation of the veterinary profession;
|
| (e) |
plagiarize the scholarly work of a colleague.
|
|
| (2) |
A veterinary surgeon or a veterinary para-professional who becomes aware of unprofessional or discreditable conduct or other misconduct by a colleague, shall attempt to counsel the colleague either directly or through the professional association.
|
| (3) |
If the behavior of the colleague specified under paragraph (2) does not change, the veterinary surgeon shall inform the Board.
|
|
| 13. |
Substitute veterinary surgeon or veterinary para-professionals
| (1) |
A veterinary surgeon or veterinary para-professional shall, where professional services may be required, at night, on a weekend, holiday or other off-duty hours, or by reason of, sickness or personal emergencies of any kind, make specific arrangements with a colleague to stand in on his behalf, for purposes of ensuring that emergency and first-aid services are available to clients.
|
| (2) |
Where first aid service has been given to an animal, a system for assessment and follow-up of cases shall be put in place at the veterinary practice.
|
| (3) |
A veterinary practice, a veterinary surgeon or veterinary para-professional shall, if called to stand in on behalf of a colleague, return to the care of that colleague on his resumption of practice, any animal attended to during his absence.
|
| (4) |
A veterinary surgeon or veterinary para-professional who stands in for a colleague shall not do anything that may be construed as an attempt to attract or entice a client, from the original veterinary surgeon or veterinary para-professional, to his veterinary practice.
|
| (5) |
An animal shall be considered to be under the immediate care of a veterinary surgeon or veterinary para-professional if—
| (a) |
the animal is under treatment by the veterinary surgeon or veterinary para-professional;
|
| (b) |
the veterinary surgeon or veterinary para-professional has been given and has accepted responsibility for the health of the animal; or
|
| (c) |
the ongoing and continuing care of the animal is recognized as a veterinary-client-patient relationship in terms of these Regulations.
|
|
|
| 14. |
Referrals
| (1) |
A veterinary surgeon shall, when a case or a treatment option is outside their area of competence, refer it to an expert whom they are satisfied is competent to carry out the investigation or treatment required.
|
| (2) |
A veterinary surgeon making a referral in accordance with (1), has a responsibility to ensure that the client is made aware of the recognized specialists or experts available in the area of competence in which the referral is made.
|
| (3) |
The veterinary surgeon referring a client and the expert to whom the client is referred to, shall ensure that the client understands the cost arising from the referral.
|
| (4) |
A veterinary surgeon referring a client as provided under paragraph (1) shall inform the client of all the experts who may be found within the client's area of residence and upon which the client may make an appointment with the expert of his choice.
|
| (5) |
A veterinary surgeon referring a client shall, on request, within one hour of the request, provide the expert with the case history of the animal and any further information that may be requested.
|
| (6) |
An expert or specialist to whom a case has been referred shall maintain professionalism when stating their opinion so it does not unduly or wrongly discredit the veterinary surgeon that has referred the client.
|
|
| 15. |
Second opinion
| (1) |
A veterinary surgeon or a client may request a second opinion on diagnosis or treatment of an animal from another veterinary surgeon.
|
| (2) |
A veterinary para-professional or his client may request a second opinion on the treatment and examination of an animal from a veterinary surgeon in the area of practice allowed in the Act.
|
| (3) |
A veterinary surgeon or veterinary para-professional shall, after seeking a second opinion in accordance with these Regulations, retain the continued responsibility of the animal.
|
| (4) |
A veterinary surgeon or a veterinary para-professional shall recognize and respect the right of a client to a second opinion and shall facilitate a client's request for a second opinion.
|
| (5) |
A veterinary surgeon shall, while providing a second opinion, state their opinion in a way which does not unduly or wrongly discredit the veterinary surgeon seeking that opinion.
|
|
| 16. |
Supersession
| (1) |
A veterinary surgeon who is called upon to attend to a patient by an owner in an ongoing case that has been under the care of another veterinary surgeon, shall, before proceeding with the treatment of the animal, endeavour to establish whether the first attending veterinary surgeon has retired from the case, or has been told by the owner that his services are no longer required.
|
| (2) |
The provisions of paragraph (1) shall not apply in case of—
| (ii) |
a second opinion or referral. |
|
| (3) |
A veterinary surgeon invited to attend to an animal under paragraph (1), shall liaise with the first attending veterinary surgeon for purposes of obtaining the history of the case.
|
| (4) |
Where a veterinary surgeon is invited to take over the whole of the veterinary work from another member of the veterinary profession, the provisions of paragraph (1) shall apply.
|
| (5) |
A veterinary surgeon is duty bound to accept and take up a case which has been interfered with by an unqualified person, provided that the client is made aware of the likelihood of poor clinical outcome for which the veterinary surgeon may not take full responsibility.
|
| (6) |
This regulation shall apply to a veterinary para-professional with respect to services allowed under the Act.
|
|
| 17. |
Specialized service
A veterinary surgeon providing specialized services shall contact the client's usual veterinary surgeon in advance and keep him fully informed, while making prior arrangements for continuation of the service.
|
| 18. |
Relationship with client
| (1) |
A veterinary surgeon or veterinary para-professional, providing veterinary services, shall create a contractual relationship between himself and the client.
|
| (2) |
A veterinary surgeon or veterinary para-professional shall—
| (a) |
display his current professional identification card to a client when called upon to offer a service;
|
| (b) |
maintain a professional relationship with a client at all times and avoid any conduct or relationship that may bring disrepute to the profession;
|
| (c) |
ensure that clear information is provided to the client about practice arrangements, the services available, initial costs, location of the out-of-hours emergency service, and information on the care of any in-patients;
|
| (d) |
maintain clear, accurate and comprehensive patients' records and accounts;
|
| (e) |
give reasonable fee estimates based on treatment options;
|
| (f) |
keep the client informed of the patient's progress, and of any escalation or reduction in costs once treatment has started;
|
| (g) |
ensure that the client is made aware of any procedures to be performed by a staff in the practice who is not a veterinary surgeon;
|
| (h) |
recognize that a client has the freedom to choose the veterinary surgeon who shall take care of his animal; and
|
| (i) |
uphold a client's right to receive impartial, independent, honest and objective advice.
|
|
| (3) |
A veterinary surgeon or veterinary para-professional shall as far as possible provide to the client copies of the records of an animal under his care.
|
| (4) |
A veterinary surgeon or veterinary para-professional shall not intentionally or recklessly cause physical or emotional harm to a client or patient.
|
| (5) |
A veterinary surgeon shall inform clients of any human health implications arising from the condition, care, tests or treatment of their animals, with special emphasis on those persons who may be more at risk.
|
|
| 19. |
Informed consent
| (1) |
A veterinary surgeon or veterinary para-professional shall acquire the informed consent of a client by explaining to the client the proposed treatment options, the risks and the significance to the patient and the fee estimates.
|
| (2) |
A veterinary surgeon or veterinary para-professional shall satisfy himself that the person giving consent under paragraph (1) has the authority to provide that consent.
|
| (3) |
A veterinary surgeon or veterinary para-professional, shall where the owner of an animal under treatment is not known, make every reasonable effort to locate the owner.
|
| (4) |
A veterinary surgeon or veterinary para-professional shall—
| (a) |
use the simple language to communicate with a client and explain any technical terminology that the client may not understand;
|
| (b) |
seek to ensure that he understands the client and that client also understands the veterinary surgeon or veterinary para-professional; and
|
| (c) |
encourage a client to take full part in any discussion related to the patient.
|
|
| (5) |
A veterinary surgeon or veterinary para-professional shall—
| (a) |
use consent forms to record the informed consent of a client obtained for the purpose of carrying out procedures of euthanasia, anesthesia and surgery; and
|
| (b) |
as far as possible, use consent forms in all other cases.
|
|
| (6) |
A consent form under this regulation shall include the following information—
| (b) |
identification of the animal;
|
| (c) |
treatment or procedure being undertaken;
|
| (d) |
name, signature and registration number of attending veterinary surgeon;
|
| (e) |
signed consent of the client; and
|
|
| (7) |
If a veterinary surgeon or veterinary para-professional is unable to obtain a client's consent in accordance with this provision, he shall indicate, in the report, the measures taken to try and obtain that consent and any action taken on the patient.
|
| (8) |
A copy of the duly filled and signed consent form shall be shall be provided to the client.
|
|
| 20. |
Responsibility to client and para-professional
| (1) |
A veterinary surgeon has a legal and moral responsibility to the client to—
| (a) |
personally examine an animal before a veterinary para-professional carries out any procedure, except in circumstances permitted under the Act;
|
| (b) |
direct, control and supervise a veterinary para-professional and ensure the work is conducted in the proper method and manner;
|
| (c) |
authorize or require that a veterinary para-professional to perform only the work as the veterinary surgeon knows that the veterinary para-professional is capable of performing.
|
|
| (2) |
A veterinary surgeon shall not staff a veterinary practice with veterinary para-professionals unless the necessary supervision or direction of a veterinary surgeon is also provided in that practice.
|
|
| 21. |
Responsibility of a veterinary para-professional to a client
A veterinary para-professional has a responsibility to the client to—
| (a) |
undertake only the treatment, procedure or other action as the Act authorizes or as directed by a veterinary surgeon;
|
| (b) |
perform his work only in the manner authorized under the Act or as directed by the veterinary surgeon;
|
| (c) |
employ the best care and skill in performing all work undertaken;
|
| (d) |
refrain from undertaking any work which he is not capable of performing satisfactorily; and
|
| (e) |
refrain from diagnosing, making a prognosis, prescribing, initiating treating or performing surgery other than that prescribed under the Act.
|
|
PART III – HANDLING OF ANIMALS
| 22. |
Examination of patients
| (1) |
A veterinary surgeon or veterinary para-professional shall at the time of making a provisional or other diagnosis physically examine a patient.
|
| (2) |
A veterinary surgeon shall physically examine an animal before giving a prescription of veterinary medicine.
|
|
| 23. |
Euthanasia
| (1) |
A veterinary surgeon, veterinary para-professional or veterinary practice may, in appropriate cases, carry out euthanasia to relieve the suffering of an animal.
|
| (2) |
Despite paragraph (1), a veterinary para-professional and a veterinary practice shall only carry out euthanasia under the supervision of a veterinary surgeon.
|
| (3) |
It is the responsibility of a veterinary professional under this regulation, to ensure the procedure and veterinary medicine used, are as guided by the standards, guidelines and recommendations of the World Health Organization.
|
| (4) |
All staff in a veterinary practice involved in euthanasia shall be fully trained and planned, rehearsed and coordinated approach shall be taken while carrying out the euthanasia.
|
| (5) |
A veterinary surgeon or a veterinary para-professional is not obligated to undertake euthanasia on a healthy animal unless required to do so under statutory powers as part of the conditions of employment.
|
| (6) |
A veterinary surgeon facing difficulties including but not limited to—
| (a) |
an owner who desires to have a perfectly healthy or treatable animal to be euthanized; or
|
| (b) |
an owner who wishes to keep an animal alive in circumstances where euthanasia would be the kindest course of action;
|
| (c) |
shall keep in mind that his primary obligation is to relieve the suffering of that animal.
|
|
| (7) |
Where the client is not in agreement with the advice given under paragraph (6), the veterinary surgeon shall make efforts to direct the client to alternative sources of advice, while recognizing the extreme sensitivity of the situation.
|
| (8) |
A veterinary surgeon shall, when euthanizing a feral or wild animal, ensure that the procedure is carried out as humanely as possible, with due consideration to the particular animal and to the group from which it is drawn.
|
| (9) |
A person who is of the opinion that a veterinary surgeon has not exhibited professional competence during euthanasia may make a complaint to the Board.
|
|
| 24. |
Ethics in teaching
| (1) |
A veterinary surgeon or veterinary para-professional engaged in teaching shall, by instruction and example—
| (a) |
emphasize the importance of humane treatment of all animals;
|
| (b) |
ensure that animals used for teaching purposes are handled humanely and are not subjected to excessive manipulation which may injure or distress the animal;
|
| (c) |
ensure that the highest possible standards of ethical conduct are promoted and maintained in all aspects of professional activity;
|
| (d) |
adhere to national and the World Organization for Animal Health guidelines on the use of animals in education.
|
|
| (2) |
A veterinary surgeon or veterinary para-professional shall teach only approved programmes in education facilities approved by the Board.
|
|
| 25. |
Use of animals in research and education
| (1) |
A veterinary surgeon or veterinary para-professional involved in research using animals shall—
| (a) |
adhere with the national legislation and the World Organization for Animal Health guidelines on use of animals in research; and
|
| (b) |
ensure that the highest standards of management, handling and analgesia are used at all times.
|
|
| (2) |
Without prejudice to paragraph (1) a veterinary surgeon or veterinary para-professional shall ensure that—
| (a) |
an animal is only used in cases where there are no alternative methods, including utilization of cells, tissues or organs of animals, may be used to obtain similar results;
|
| (b) |
care is taken to use fewer animals to obtain comparable levels of information or to obtain more information from the same number of animals; and
|
| (c) |
the methods used prevent, alleviate or minimize pain, suffering, distress or lasting harm and enhance welfare of the animal used.
|
|
| (3) |
A veterinary surgeon or veterinary para-professional shall undertake regular training to update themselves on the humane handling of animals.
|
| (4) |
A veterinary surgeon or veterinary para-professional shall actively discourage research work using animals where the research is unnecessarily repetitive or has ill-defined goals and shall, whenever possible, encourage the substitution of in-vivo with in-vitro techniques.
|
|
| 26. |
Transportation and processing of animals
| (1) |
A veterinary surgeon or veterinary para-professional shall be responsible for ensuring compliance with the welfare requirements, relevant national and international laws and the proper handling of an animal being transported.
|
| (2) |
A veterinary surgeon and veterinary para-professional shall ensure that an animal destined for an animal product processing industry is transported and slaughtered in a humane manner.
|
|
PART IV – VETERINARY PREMISES AND PRACTICE
| 27. |
Professional premises
A veterinary professional premises shall be located—
| (a) |
away from premises that may not reflect the good standing of the profession including gambling shops and places of public entertainment; or
|
| (b) |
in an area where animals being attended at the veterinary practice are not likely to pose any danger or discomfort to the general public and vice versa.
|
|
| 28. |
Environmental health
| (1) |
A veterinary surgeon or a veterinary para-professional shall work and dispose waste in the manner as to reduce pollution from disinfectants, medicinal products, chemicals and biological waste.
|
| (2) |
Waste produced at a veterinary practice shall be disposed in a manner that does not pose a danger to the environment or to any person or animal that may come into contact with the waste.
|
|
| 29. |
Employment
| (1) |
A veterinary surgeon or veterinary para-professional employed by a person or an institution, by virtue of their professional training and qualifications shall conduct himself or herself with professional dignity and decorum.
|
| (2) |
A practicing veterinary surgeon shall not, in matters of the veterinary profession, be subject to the direction or control of a veterinary para-professional or a lay person.
|
| (3) |
A veterinary surgeon responsible to another Board or committee shall regard it as a professional responsibility to advise the board or committee on all matters of veterinary professional ethics.
|
| (4) |
A veterinary surgeon or veterinary para-professional shall comply with the recommendations of this Code, and be careful to not to gain unfair advantage over any other colleague or veterinary practice because of the particular advantages the structure of the employing organization may allow.
|
| (5) |
Where an employing organization fails to follow professional advice resulting or which may result in a breach of the ethical requirements of the veterinary profession, the veterinary surgeon or veterinary para-professional shall immediately notify the Board.
|
| (6) |
A veterinary surgeon or a veterinary para-professional practicing in a food and feed plant shall ensure that he or she is conversant with the codes of practice relevant to the food or feed in question and shall only certify that the food or feed is fit for human and animal consumption based on scientific principles and the existing law.
|
| (7) |
A veterinary surgeon or a veterinary para-professional practicing in a veterinary laboratory shall update his knowledge of, and ensure observation of the law relating to the bio-safety and bio-containment.
|
| (8) |
A veterinary surgeon practicing in a veterinary laboratory shall only release results of tests he or she has conducted or supervised.
|
|
| 30. |
Ethics in prescribing, dispensing and administering, veterinary medicine
| (1) |
A veterinary surgeon prescribing a veterinary medicine to a patient shall do so in writing and shall indicate—
| (a) |
the correct method of use;
|
| (b) |
route or manner of administration;
|
| (d) |
special precautions relevant to that veterinary medicine.
|
|
| (2) |
A veterinary surgeon dispensing a veterinary medicine to a client or directing a veterinary para-professional on the use of a veterinary medicine shall ensure that the client or the veterinary para-professional is aware of the details specified under paragraph (1) (a), (b), (c) and (d).
|
| (3) |
A veterinary surgeon shall inform a client of the safety, efficacy and side effects of a veterinary medicine at the time of dispensing the medicine.
|
| (4) |
A veterinary surgeon who supplies a restricted veterinary medicine to a client to use on that client's animal or who directs a veterinary para-professional to use a restricted veterinary medicine bears full responsibility for the use of that medicine and any adverse reactions that may result.
|
| (5) |
A veterinary surgeon may only prescribe a veterinary medicine for a patient with whom there exists a veterinary-client-patient relationship.
|
| (6) |
A veterinary surgeon and a veterinary para-professional shall protect veterinary medicine from misuse.
|
| (7) |
Without prejudice to paragraph (5), a veterinary surgeon shall promote a veterinary practice that reduces the need to use antibiotics through—
| (a) |
maintaining the highest standards of hygiene in animal establishments;
|
| (b) |
use of vaccines instead of antibiotics;
|
| (c) |
where applicable, avoiding the use of antibiotics for preventive purposes;
|
| (d) |
encouraging the use of pro-biotics; and
|
| (e) |
conducting sensitivity tests, as much as possible, to inform on the most effective antibiotic for a particular infection.
|
|
|
| 31. |
Control of unqualified persons
| (1) |
A veterinary surgeon, whether or not physically present, is ultimately responsible for the outcome and the technical competence of anyone to whom he has delegated veterinary procedures or other veterinary duties.
|
| (2) |
A veterinary student gaining experience as part of his veterinary medicine or animal health course may examine animals and carry out diagnostic tests under the supervision of a veterinary surgeon.
|
| (3) |
A veterinary graduate on internship may—
| (a) |
examine animals, carry out diagnostic tests and administer treatment under the direction of a veterinary surgeon; or
|
| (b) |
perform surgical operations under the supervision of a veterinary surgeon.
|
|
| (4) |
A veterinary surgeon or a veterinary para-professional shall not engage in any act that aids or abets the unlicensed practice of veterinary medicine including the delegation of duties to unregistered staff or allowing unregistered staff to influence or engage in the practice of veterinary medicine.
|
| (5) |
A veterinary surgeon in-charge of a veterinary practice shall apply fair labour and employment practices as guided by the laws relating to labour.
|
|
PART V – GENERAL PROVISIONS
| 32. |
Conflict of interest
| (1) |
A veterinary surgeon or a veterinary para-professional shall refrain from being involved in any situation where conflict of interest may arise.
|
| (2) |
A veterinary surgeon or veterinary para-professional shall not undertake a business which may give rise to conflict of interest or call into question the integrity of the profession.
|
| (3) |
Without prejudice to the provisions of paragraph (2), a veterinary surgeon shall not provide certification or valuation of an animal or valuation where the veterinary surgeon is the owner or the breeder of that animal.
|
|
| 33. |
Soliciting for professional work
A veterinary surgeon or a veterinary para-professional who, with a view to obtaining professional work, makes or instigates an unsolicited approach to an organization, person or persons who are not bona fide clients, shall ensure that the approach—
| (a) |
does not in any way bring the veterinary profession into disrepute;
|
| (b) |
is in a manner which cannot be considered by the recipient to be intrusive, offensive or harassing;
|
| (c) |
provides information that is factual; and
|
| (d) |
does not include claims or implications of personal superiority over any other veterinary surgeon or veterinary para-professional.
|
|
| 34. |
Confidentiality
| (1) |
A veterinary surgeon or veterinary para-professional shall treat all information concerning a client or an animal, that arises out of his care, attention or treatment, as confidential and the information shall, unless there is a legal obligation to do so, not be divulged to any other person outside the practice without the owner's consent.
|
| (2) |
Despite paragraph (1), where members of the public or animals are endangered, and a veterinary surgeon or veterinary paraprofessional determines that there is an obligation to divulge any information which has come to his attention in the course of his professional work, the cooperation of the client shall, if possible, be obtained before taking action to divulge the information.
|
|
| 35. |
Expert advice and report
| (1) |
A veterinary surgeon shall give evidence impartially and in the interest of justice where—
| (a) |
required to write an expert witness statement, opinion or report; or
|
| (b) |
is summoned or called upon in a professional capacity to give expert evidence in a court of law.
|
|
| (2) |
A veterinary surgeon shall prepare and present expert advice, whether serving as an expert witness or in any other capacity, in a thorough and professional manner which shall include the—
| (a) |
identification of the animal or herd;
|
|
| (3) |
Without prejudice to the provisions of paragraph (2), where a veterinary surgeon is required to divulge information in circumstances where the disclosure—
| (a) |
concerns public safety, consumer health and welfare; or
|
| (b) |
is required by law or as a witness in court,
|
a veterinary surgeon shall divulge only that information which is relevant.
|
| (4) |
A veterinary surgeon shall take care to differentiate factual information and personal opinion.
|
|
| 36. |
Veterinary certificate
| (1) |
A certificate or other document issued by a veterinary surgeon in accordance with any legal requirement, shall—
| (a) |
be carefully compiled, completed and shall be accurate in every detail;
|
| (b) |
bear the name, signature and registration number of the veterinary surgeon;
|
| (c) |
ensure all necessary steps are taken to ascertain that the matters certified are true; and
|
| (d) |
include details relating to the animal which is the subject of the certificate or document including—
| (ii) |
details of ownership; |
| (iii) |
locality where examination was done; |
| (vii) |
color of the animal; and |
| (viii) |
tattoos, tags notches, microchip or other identification marks. |
|
|
| (2) |
A veterinary surgeon shall attach any other relevant documentation to the certificate or document issued under paragraph (1).
|
| (3) |
A veterinary surgeon shall not sign a vaccination certificate unless he has administered the vaccination personally or has supervised the administration of the vaccine by a veterinary para-professional.
|
|
| 37. |
Recreational activities
| (1) |
Subject to paragraph (1), a veterinary surgeon or a veterinary para-professional may engage in social and recreational activities including arts and sports.
|
| (2) |
Despite paragraph (1), a veterinary surgeon or a veterinary para-professional may engage in social and recreational activities including arts and sports where the activities do not adversely affect the dignity of the profession or interfere with the performance of his professional duties.
|
| (3) |
A veterinary surgeon or veterinary para-professional may make a useful contribution to the promotion of animal welfare and responsible pet ownership by taking part in public life, whether in national or local politics, community service, or involvement with the media and in so doing ensure he distinguishes between personal opinion, political belief and established facts.
|
|
| 38. |
Communication
| (1) |
A veterinary surgeon or veterinary para-professional shall advise the staff in his veterinary practice to communicate any concerns raised by a client or misunderstanding, to the management of the practice and shall ensure that the concerns are addressed and the client is kept fully informed.
|
| (2) |
A veterinary surgeon or veterinary para-professional who is part of a veterinary team or teams in different veterinary practices shall always endeavor to work collaboratively and harmoniously with team members, show respect and ensure effective communication with other members of the team.
|
|
| 39. |
Publicity and advertisement
| (1) |
For purposes of this regulations "to endorse" includes the wearing of a 'tee shirt' or other attire, writing a prescription on a headed pad, or other action that promotes directly or indirectly a particular medicine or drug manufacturer through the use of a trade mark or logo.
|
| (2) |
A veterinary surgeon or veterinary para-professional shall not endorse an ethical veterinary medicine or service but may endorse non-medical products and services provided the endorsement does not bring the profession into disrepute.
|
| (3) |
A veterinary surgeon or veterinary para-professional shall ensure that any publicity or advertising he undertakes—
| (a) |
is factual and informative to the clients and the general public;
|
| (b) |
does not exploit a client's lack of veterinary knowledge;
|
| (c) |
is not of a character likely to bring the profession into disrepute;
|
| (d) |
whether by telephone or visit, cannot be construed as being solicitation for clients.
|
|
| (4) |
A veterinary practice may, as a means of communication, release newsletters and circulars to clients and may include comments about animal remedy.
|
|
| 40. |
Ethics in the media
| (1) |
A veterinary surgeon or veterinary para-professional shall in the use of all forms of media, demonstrate high standards of sensitivity, etiquette and responsibility and shall not use language, pictures or other thing that may bring disrepute to a colleague or to the profession.
|
| (2) |
A veterinary surgeon or a veterinary para-professional shall, when communicating matters of a professional nature or of concern to the profession, through print or social media, identify himself by his name as registered by the Board.
|
|
| 41. |
Dress code
| (1) |
A veterinary surgeon or veterinary para-professional shall adopt an appropriate dress code and grooming that brings honour and dignity to the profession.
|
| (2) |
A veterinary practice or a veterinary institution shall provide uniform and other protective clothing for its staff.
|
| (3) |
A veterinary surgeon or a veterinary para-professional shall at all times use personal protection equipment or gear while at the work place.
|
|
| 42. |
Settling disputes within the profession
| (1) |
A veterinary surgeon or veterinary para-professional shall attempt by all means possible, to settle a professional dispute within the profession in the manner so as to avoid any recourse to publicity that may bring the profession into disrepute.
|
| (2) |
A veterinary surgeon or veterinary para-professional involved in a dispute with a colleague may approach the Board for advice.
|
| (3) |
A client or a member of the public who is dissatisfied with a decision or the conduct of a veterinary surgeon or veterinary para-professional may register the dissatisfaction with the Board and request appropriate action be taken against the veterinary surgeon or veterinary para-professional.
|
|
| 43. |
Discipline
A veterinary surgeon or veterinary para-professional in breach of this code is guilty of professional misconduct and shall be subjected to disciplinary proceedings in accordance with Part VI of the Act.
|
THE VETERINARY SURGEONS AND VETERINARY PARA-PROFESSIONALS (VETERINARY MEDICINES DIRECTORATE) REGULATIONS
ARRANGEMENT OF REGULATIONS
PART I – PRELIMINARY
| 3. |
Objectives and purpose of the Regulations
|
PART II – THE VETERINARY MEDICINES DIRECTORATE
| 5. |
Establishment of the Directorate
|
| 6. |
Functions of the Directorate
|
| 8. |
Composition and appointment of the Council
|
| 9. |
Co-option of specialized persons
|
| 10. |
Meetings of the Council
|
| 13. |
Chief Executive Officer
|
| 14. |
Removal of the Chief Executive Officer
|
| 15. |
Veterinary medicines inspectors
|
| 18. |
Protection from personal liability
|
PART III – MANUFACTURING, IMPORTATION AND REGISTRATION OF VETERINARY MEDICINES
| 19. |
Veterinary medicines to be registered
|
| 21. |
Manufacturing of veterinary medicines
|
| 22. |
Powers of inspectors with regard manufacturing plants
|
| 23. |
Application for registration of a veterinary medicine
|
| 24. |
Renewal of registration of veterinary medicine
|
| 25. |
Clinical and non-clinical trials and toxicity testing
|
| 26. |
Conditions for registration of veterinary medicines and pesticides
|
| 27. |
Veterinary Medicines Registration Committee
|
| 28. |
Rejection of application for registration of a veterinary medicine
|
| 29. |
Registration of veterinary medicine researched outside Kenya
|
| 30. |
Suspension and revocation of certificate of registration
|
| 31. |
Period for renewal of registration and retention of veterinary medicine
|
PART IV – CLASSIFICATION AND CATEGORIZATION OF VETERINARY MEDICINES
| 32. |
Classification of Veterinary Medicines
|
| 33. |
Categorization of Veterinary Medicines
|
| 34. |
Use of veterinary pesticides
|
PART V – VETERINARY PHARMACY
| 35. |
Standards of Veterinary Pharmacy
|
| 36. |
Qualifications to operate a veterinary pharmacy
|
| 37. |
Council to approve veterinary pharmacy businesses
|
| 39. |
Practice permit to be displayed
|
PART VI – VETERINARY PHARMACY AND COMPLEMENTARY BUSINESSES
| 42. |
Complementary businesses
|
| 43. |
Stock, supply and distribution of veterinary medicine
|
PART VII – MARKET AUTHORIZATION
| 44. |
Authorization to import or export veterinary medicine
|
PART VIII – VETERINARY MEDICINE INSPECTORS
| 45. |
Appointment of veterinary medicine inspectors
|
| 46. |
Identification of veterinary medicines inspector
|
| 47. |
Powers of veterinary medicines inspectors
|
PART IX – FINANCIAL PROVISIONS
| 48. |
Funds of the Directorate
|
PART X – OFFENCES
| 54. |
Officers of corporations
|
| 55. |
Passing confidential information without authorization
|
PART XI – GENERAL PROVISIONS
| 57. |
Advertising veterinary standards for medicines
|
| 58. |
Exemptions to Regulations
|
| 59. |
Discretion of Council
|
| 60. |
Copy of certificate or authorization
|
| 63. |
Transition and validity of licences and permits
|
SCHEDULES
| FIRST SCHEDULE [r. 8(6)] — |
CONDUCT OF THE AFFAIRS OF THE COUNCIL
|
| SECOND SCHEDULE [rr. 32(1) & 33(1).] — |
CONDUCT OF THE AFFAIRS OF THE COUNCIL
|
| THIRD SCHEDULE [rr. 23(1) & 38(6).] — |
FORMS
|
| FOURTH SCHEDULE [r. 17.] — |
CODE OF CONDUCT FOR MEMBERS OF AND EMPLOYEES OF THE DIRECTORATE
|
THE VETERINARY SURGEONS AND VETERINARY PARA-PROFESSIONALS (VETERINARY MEDICINES DIRECTORATE) REGULATIONS
PART I – PRELIMINARY
| 1. |
Citation
These Regulations may be cited as the Veterinary Surgeons and Veterinary Paraprofessionals (Veterinary Medicines Directorate) Regulations, 2015.
|
| 2. |
Interpretation
In these Regulations, unless the context otherwise requires—
"accredited laboratory" means a laboratory recognized as an accredited laboratory by the Directorate;
"advertisement" means any written or visual notice, circular, label, or wrapper, or other descriptive matter, verbal statement or reference appearing in any newspaper, television, film or mass media or brought to the attention of the public in any other form, which is intended to promote the sale of a veterinary medicine;
"alternative medicine" means the unrefined plant, animal and mineral substances commonly used in traditional animal treatments;
"Cabinet Secretary" means the Cabinet Secretary responsible for matters relating to veterinary service;
"Chief Executive Officer" means the Chief Executive Officer of the Directorate appointed under regulation 13;
"Committee" means the Veterinary Medicines Registration Committee established under regulation 27;
"controlled veterinary medicine" means a veterinary medicine specified in the Fourth Schedule as a Category I or Category II of veterinary medicine;
"Conventional medicines" means the regular and standardized veterinary medicines;
"Council" means the Council of the Directorate comprised under regulation 8 (1);
"crude drug" means an unrefined medicine of biological origin;
"Directorate" means the Veterinary Medicines Directorate established under regulation 5 and as envisaged under section 39(2)(a) of the Act;
"dispense" means the sale or supply of a veterinary medicine by a veterinary surgeon or other person authorised in accordance with these Regulations;
"inspector" means a person appointed as an inspector under regulation 45;
"manufacture" means any stage in the manufacturing of a veterinary medicine until the finished product is ready for sale in its final form as specified in the marketing authorization, and includes repackaging, repacking or labeling of a veterinary medicine in an authorized facility but does not include the breaking open of the package of a veterinary medicine by retailers;
"market authorization" means registration of a veterinary medicine by the Council and the issuance of a registration certificate under regulation 23;
"orphan veterinary medicine" means a veterinary medicine that is not economical to trade in but is required for specific medical use;
"pharmaco-vigilance" means the routine inspections and surveys carried out in the veterinary medicines market to safeguard general animal and human health and trade;
"quality assurance standards" means the good manufacturing practice, good laboratory practice, good veterinary practice or any other standard developed by an international standardization body, the East African Community Standards Committee or the Kenya Bureau of Standards which the Cabinet Secretary, on the advice of the Council, shall recognize through the Gazette as a quality assurance standard for the purposes of these Regulations;
"register" means a register maintained by the Council containing the details of—
| (a) |
premises which have been issued with a permit in accordance with these Regulations; or
|
| (b) |
veterinary medicines registered in the country;
|
"retailer" means a veterinary pharmacy registered by the Council for the sale of veterinary medicines to the end users;
"specialized committees" means persons who are not Council members;
"veterinary medicines inspector" means an inspector appointed by the Council pursuant to regulation 45;
"veterinary pesticide" means a veterinary medicine used as a pest control product on animals or the animals' environment;
"veterinary pharmaceutical" means a chemical substance formulated or compounded as a single active ingredient or in any combination of the chemical substances, for veterinary curative use;
"veterinary pharmacy" means a business authorized by the Council to stock, dispense or distribute veterinary medicines for sale;
"veterinary pharmacy assistant" means a veterinary para-professional trading in veterinary medicines listed under Category III of the Fourth Schedule;
"veterinary pharmacy practice" means the business of veterinary pharmacy carried out by veterinary surgeons, veterinary para-professionals or any other person permitted by the Council to carry out veterinary pharmacy practice;
"veterinary product" means veterinary medicines and veterinary pesticides;
"wholesale" means a manufacturer or veterinary pharmacy approved by the Council to trade in bulk in the supply of veterinary medicines to wholesalers or retailers in the original outer pack-sizes.
|
| 3. |
Objectives and purpose of the Regulations
The object and purpose of these Regulations is to—
| (a) |
regulate the manufacture, importation, exportation, registration, distribution, prescription and dispensing of veterinary medicines and the practice of veterinary pharmacy in Kenya; and
|
| (b) |
advise the Kenya Veterinary Board in relation to all aspects listed under paragraph (a).
|
|
| 4. |
Application
Subject to regulation 58, these Regulations shall apply to all conventional and alternate veterinary medicine.
|
PART II – THE VETERINARY MEDICINES DIRECTORATE
| 5. |
Establishment of the Directorate
| (1) |
There is hereby established a Directorate to be known as the Veterinary Medicines Directorate whose management shall vest on a Council appointed under regulation 8.
|
| (2) |
The Directorate shall be a body corporate with perpetual succession and a common seal and shall in its corporate name be capable of—
| (b) |
taking, purchasing, or otherwise acquiring, holding, charging or disposing of movable and immovable property;
|
| (c) |
borrowing and lending money; and
|
| (d) |
doing all such other things or acts as may lawfully be done by a body corporate.
|
|
|
| 6. |
Functions of the Directorate
The functions of the Directorate are to —
| (a) |
formulate and enforce quality assurance standards in the in the manufacture, distribution and use of veterinary medicines to safeguard human and animal health and the environment;
|
| (b) |
in consultation with the Director of Veterinary Services, regulate the use of veterinary medicine for the treatment of animals under the Animal Diseases Act (Cap. 364);
|
| (c) |
consider applications for approval for market authorization of veterinary medicines;
|
| (d) |
set quality assurance standards for training in the management of veterinary medicines as directed by the Kenya Veterinary Board;
|
| (e) |
collaborate with the Kenya Veterinary Board in regulating training in the management of veterinary medicines;
|
| (f) |
inspect and approve premises in which the manufacture, sale or supply of veterinary medicine is conducted;
|
| (g) |
appoint and gazette veterinary medicine inspectors;
|
| (h) |
establish the standard operating procedures for veterinary medicine inspectors;
|
| (i) |
regulate veterinary pharmacy practices;
|
| (j) |
categorize veterinary medicines and the qualification of persons authorized to trade in each category and review the categories every five years;
|
| (k) |
regulate clinical and non-clinical trials of veterinary medicines by individuals and institutions to be involved in the trials;
|
| (l) |
regulate the manufacture, importation, exportation, handling, advertisement, labeling, sale and disposal of veterinary medicines;
|
| (m) |
register all veterinary medicines manufactured or imported for use in the country or exported from the country;
|
| (n) |
monitor the market for and take measures necessary for the elimination of trade in illegal and counterfeit veterinary medicines;
|
| (o) |
establish systems of pharmaco-vigilance and conduct pharmaco-vigilance of veterinary medicines through regular inspections and surveys;
|
| (p) |
enforce good manufacturing practice for veterinary medicines as approved by the Council;
|
| (q) |
develop, apply and from time to time review guidelines to be used in the inspection and ensuring compliance with good manufacturing practice;
|
| (r) |
ensure that the promotion and marketing of veterinary medicine is in accordance with the approved product information;
|
| (s) |
publish, on an annual basis, a notice in the Kenya Gazette inviting the public to note and inspect the register of veterinary medicines and the list of approved veterinary pharmacy practices within such period and at such place as may be specified in the notice;
|
| (t) |
consider, grant, issue or revoke authorizations and certificates in accordance with these Regulations;
|
| (u) |
collaborate with other regulatory agencies including the Public Health (Standards) Board in section 27 of the Food, Drugs and Chemical Substances Act (Cap. 254), the Pest Control Products Board established under section 5 of the Pest Control Products Act (Cap. 346) and the Central Board of Health established under section 3 of the Public Health Act (Cap. 242) to carry out its mandate; and
|
| (v) |
undertake any other thing necessary for the effective carrying out of its mandate under this or any other Act.
|
|
| 7. |
Powers of the Council
The Council shall have all the power necessary or expedient for the effective discharge of its functions under these Regulations and any other law, and in particular the power to—
| (a) |
control, supervise and manage the assets and liabilities of the Directorate;
|
| (b) |
determine the provision to be made for capital and recurrent expenditure and for the reserves of the Directorate;
|
| (c) |
seek and receive any grants or donations and make legitimate disbursements from such grants and donations for its purposes;
|
| (d) |
levy fees and charges for its services as provided in these Regulations;
|
| (e) |
enter into association with other bodies within or outside Kenya which the Council may consider desirable or appropriate;
|
| (f) |
invest funds of the Directorate not immediately required in securities in which trustees are empowered to invest under the Trustee Act (Cap. 167), and in other securities which may be approved for the purpose, by the Cabinet Secretary for the time being responsible for Finance;
|
| (g) |
establish and support investment and trust funds for the benefit of employees or ex-employees of the Directorate or dependants of such persons, to grant pension, benefits and allowances and to make such payments towards insurance as required under the relevant laws;
|
| (h) |
open and operate such accounts as are necessary for the funds of the Directorate, with a bank or financial institution licensed to conduct banking business under the Banking Act (Cap. 488);
|
| (i) |
recruit, support, discipline or dismiss the staff and inspectors of the Directorate;
|
| (j) |
in consultation with Salaries Remuneration Commission, determine the terms and conditions of employment of the staff and inspectors of the Directorate;
|
| (k) |
establish branch offices of the Directorate, to the extent that is practicable, to ensure accessibility of its services by all Kenyans;
|
| (l) |
superintend, regulate and assist branch offices, auxiliaries, committees and other forms of organizations established to advance the interest of the Directorate;
|
| (m) |
perform all things necessary or incidental to attain the objectives of the these Regulations or any other written law.
|
|
| 8. |
Composition and appointment of the Council
| (1) |
The Council shall be appointed by the Cabinet Secretary and shall consist of—
| (a) |
the Director of Veterinary Services who shall be the Chairperson;
|
| (b) |
the Registrar of the Pharmacy and Poisons Board;
|
| (c) |
the Chief Executive Officer of the Kenya Veterinary Board;
|
| (d) |
the Principal Secretary for the time being responsible for Finance;
|
| (e) |
the Principal Secretary for the time being responsible for animal health matters;
|
| (f) |
three veterinary surgeons nominated by the Kenya Veterinary Board;
|
| (g) |
one veterinary technologist from the veterinary pharmaceutical industry nominated by the Kenya Veterinary Board; and
|
| (h) |
the Chief Executive Officer of the Directorate who shall be the Secretary to the Council and shall be an ex-officio member.
|
|
| (2) |
A person appointed under paragraph (1)(f) shall be nominated by the Kenya Veterinary Board from a list of five names drawn from the veterinary pharmaceutical industry including a trainer in veterinary pharmacology submitted by a registered professional association representing the interests of veterinary surgeons countrywide.
|
| (3) |
A person appointed under paragraph (1)(g) shall be nominated by the Kenya Veterinary Board from a list of three names from the veterinary pharmaceutical industry submitted by the registered association representing the interests of veterinary paraprofessionals.
|
| (4) |
The nominating bodies referred to under paragraphs (2) and (3) shall observe Constitutional principles relating to gender, youth, persons with disability and minorities in identifying the persons whose names shall be submitted for nomination by the Kenya Veterinary Board for appointment to the Council.
|
| (5) |
The Cabinet Secretary shall appoint the members of the Council by notice in the Kenya Gazette.
|
| (6) |
The Council shall conduct its affairs in the manner set out in the First Schedule.
|
| (7) |
The members of the Council shall hold office for a term of three years and shall be eligible for re-appointment for one further term.
|
| (8) |
The members appointed under paragraph (1)(b), (d) and (e) may, in writing, designate representatives to attend the meetings of the Council on their behalf.
|
| (9) |
The members of the Council in paragraphs (f), (g) and (h) may—
| (a) |
at any time resign from office by notice in writing to the Cabinet Secretary;
|
| (b) |
be removed from office by the Cabinet Secretary, on the advice of the Council if the member —
| (i) |
is declared bankrupt; |
| (ii) |
is absent from three consecutive meetings of the Council, without the permission of the Chairperson; |
| (iii) |
is convicted of a criminal offence and, sentenced to a term of imprisonment of more than six months; |
| (iv) |
is unable or unfit, due to physical or mental illness, to perform the functions of his office; or |
| (v) |
has failed to comply with the provisions of Chapter Six of the Constitution. |
|
|
| (10) |
The Cabinet Secretary shall, on the recommendation of the Council, appoint a relevant person to serve in the place of any member of the Council in the case of death, resignation, absence from Kenya for six consecutive months without authorization or more or removal from office under paragraph (9)(b) and the person appointed under this sub-Regulation shall serve until the end of the term of the Council.
|
| (11) |
Where a vacancy arises under paragraph (10), the Cabinet Secretary shall undertake a fresh recruitment process to fill the vacancy.
|
|
| 9. |
Co-option of specialized persons
Pursuant to section 10 of the Act, the Council may for the effective discharge of its functions co-opt into the committees such persons with technical expertise or knowledge for the better carrying out of the functions of the Directorate.
|
| 10. |
Meetings of the Council
The conduct and regulation of the business and affairs of the Council shall be as provided in the First Schedule but subject thereto, the Council shall regulate its own procedure.
|
| 11. |
Conflict of interest
| (1) |
If any person is present at a meeting of the Council or any committee at which any matter is the subject of consideration and in which matter that person or that person's spouse or relative is directly or indirectly interested in a private capacity, that person shall as soon as is practicable after the commencement of the meeting declare such interest and shall not, unless the Council or committee otherwise directs, take part in any consideration or discussion of, or vote on any question connected to such matter.
|
| (2) |
The disclosure of interest shall be recorded in the minutes of the meeting at which it is made.
|
| (3) |
A member or employee of the Council shall not carry out an business or trade with the Council.
|
| (4) |
A member or staff of the Council who contravenes this Regulation commits an offence and is liable, on conviction, to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months or to both.
|
|
| 12. |
Common seal and logo
| (1) |
The Directorate shall have a common seal which shall be kept by the Chief Executive Officer.
|
| (2) |
The affixing of the seal shall be authenticated by signature of the chairperson and the Chief Executive Officer and in his absence, the signature of any other member authorized by resolution of the Council.
|
|
| 13. |
Chief Executive Officer
| (1) |
There shall be a Chief Executive Officer who shall be appointed by the Council through a competitive process and whose terms and conditions of service shall be determined by the Council in the instrument of appointment.
|
| (2) |
A person shall be qualified for appointment as a Chief Executive officer, if that person—
| (b) |
holds a degree in veterinary medicine from a university recognized by the Kenya Veterinary Board;
|
| (c) |
has at least ten years professional experience in veterinary medicine regulation, of which at least five years are at senior management level; and
|
| (d) |
satisfies the requirements of Chapter Six of the Constitution.
|
|
| (3) |
The Chief Executive Officer shall be an ex-officio member of the Council but shall have no right to vote at any meeting of the Council.
|
| (4) |
The Chief Executive Officer shall hold office for a term of five years and shall be eligible for reappointment for one further term.
|
| (5) |
The Chief Executive Officers shall, in the performance of the functions and duties of office. be responsible to the Council.
|
| (6) |
Without prejudice to the provisions of paragraph (5), the Chief Executive Officer shall—
| (a) |
| (i) |
carrying into effect the decisions of the Council; |
| (ii) |
day-to-day administration and management of the affairs of the Directorate; |
| (iii) |
supervision of the staff of the Directorate; |
|
| (b) |
be the Registrar of veterinary medicines; and
|
| (c) |
perform such other duties as may be assigned by the Council.
|
|
|
| 14. |
Removal of the Chief Executive Officer
| (1) |
The Chief Executive Officer may be removed from office by the Council in accordance with the terms and condition of service, for—
| (a) |
inability to perform functions of the office arising out of physical or mental infirmity;
|
| (b) |
gross misconduct or misbehavior;
|
| (c) |
incompetence or negligence of duty;
|
| (d) |
violation of the Constitution and any other written law; or
|
| (e) |
any other grounds specified in the terms and conditions of service.
|
|
| (2) |
Where the question of the removal of the Chief Executive Officer under paragraph (1) arises, the Council shall —
| (a) |
inform the Chief Executive Officer in writing of the reasons for the intended removal; and
|
| (b) |
provide the Chief Executive Officer with the opportunity to be heard in accordance with the principles of fair administrative action safeguarded under Article 47 of the Constitution.
|
|
|
| 15. |
Veterinary medicines inspectors
| (1) |
The Council shall employ such veterinary medicines inspectors as it deems necessary.
|
| (2) |
A person shall be qualified for appointment as a veterinary medicines inspector if that person —
| (a) |
is a veterinary surgeon or technologist; or
|
| (b) |
holds at least a diploma in animal health or other qualification recognized by the Council; and
|
| (c) |
has at least five years post qualification experience in a relevant field.
|
|
|
| 16. |
Staff
The Council may employ such staff as it deems appropriate for the effective performance of its functions.
|
| 17. |
Code of conduct
The members and the employees of the Council shall subscribe to the leadership and integrity code set out in the Fourth Schedule.
|
| 18. |
Protection from personal liability
Nothing done by a member of the Council or by any person working under the instructions of the Council shall, if done in good faith for the purpose of executing the powers, functions or duties of the Council under these Regulations render such member or officer personally liable for any action, claim or demand.
|
PART III – MANUFACTURING, IMPORTATION AND REGISTRATION OF VETERINARY MEDICINES
| 19. |
Veterinary medicines to be registered
| (1) |
A person shall not import, manufacture sell, transport or distribute any veterinary medicine in Kenya unless that veterinary medicine has been registered in accordance with the provisions of these Regulations.
|
| (2) |
Every person transporting a veterinary medicine in transit shall declare the veterinary medicine at the port of entry and exit.
|
|
| 20. |
Manufacturing permit
A person who intends to manufacture veterinary medicine shall make an application to the Council for a manufacturing permit.
|
| 21. |
Manufacturing of veterinary medicines
| (1) |
The Council shall, on receipt of an application made under regulation 20, before issuing a certificate of registration, cause the premises in which the manufacturing of the veterinary medicine is proposed to be conducted be inspected in order to ensure compliance to good manufacturing practice.
|
| (2) |
The inspector instructed under paragraph (1) shall submit the results of the inspection to the Council and the applicant in writing.
|
| (3) |
A manufacturer shall ensure that every stage in the manufacture of a veterinary medicine is carried out based on good manufacturing practices.
|
| (4) |
A manufacturer shall not manufacture a veterinary medicine unless the manufacturing plant has been inspected and licensed in accordance with this Regulation.
|
| (5) |
A person shall not use any premises, for the purposes of manufacturing. formulating, packaging, selling or storing veterinary medicine unless that person is in possession of a licence issued under these Regulations in respect of that premises.
|
| (6) |
Where an inspector has approved the compliance by the manufacturer, the Council shall on payment of the prescribed fee, issue a licence as set out in Form 1.3 set out in the a Third Schedule.
|
| (7) |
A person in executive authority of manufacturing company which manufactures veterinary medicines without a license commits an offence and shall be liable, on conviction, to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months or to both.
|
|
| 22. |
Powers of inspectors with regard manufacturing plants
| (1) |
An inspector appointed under these Regulations shall have power with regard to manufacturing plants to —
| (a) |
enter the premises and inspect the plant and the process of manufacture intended to be employed in the manufacturing of the veterinary medicine and shall make a report to the Council; or
|
| (b) |
order the immediate closure of a manufacturing plant where its continued operation appears to pose a serious threat to life and safety.
|
|
| (2) |
An inspector who has ordered the closure of a plant under paragraph (1)(b) shall within twenty-four hours of the closure notify the Council of the closure and shall provide reasons for the closure.
|
| (3) |
On receipt of the notification under paragraph (2), the Council shall direct the owner of the plant to take such measures as the Council may direct to ensure conformity with these Regulations.
|
| (4) |
A manufacturer who fails, within fourteen days of receipt of the directive made by the Council under paragraph (3), to take the measures indicated in the directive shall have its licence revoked.
|
| (5) |
The Council shall publish in at least one newspaper of wide national circulation the name of a manufacturing plant closed and whose licence has been revoked under this Regulation and shall place a notice of the closure at the entrance of such plant.
|
| (6) |
The protection of right to property under Article 40 of the Constitution shall be limited for purposes of maintenance of veterinary medicine standards for the protection and safety of the public.
|
|
| 23. |
Application for registration of a veterinary medicine
| (1) |
An application for registration of —
| (a) |
a veterinary medicine shall be in accordance with Form A set out in the Third Schedule; and
|
| (b) |
a veterinary medicine used as pesticides shall be Form B set out in the Third Schedule.
|
|
| (2) |
An applicant shall, in addition to the information required to be paragraph (1), furnish such further information and material as may be required by the Council for the proper evaluation of the veterinary medicine in respect of which the application is made.
|
| (3) |
The Council shall, on approval of an application made under this Regulation and on payment of the prescribed fees, register the veterinary medicine or veterinary pesticide and issue a certificate in Form D1 or D2, respectively set out in the Third Schedule.
|
| (4) |
A certificate of registration issued under these Regulations shall, unless suspended or revoked, be in force for a period of five years.
|
| (5) |
The Council shall not approve an application made under paragraph (1) unless satisfied that the applicant has attained the prescribed standards and satisfied all requirements for registration.
|
|
| 24. |
Renewal of registration of veterinary medicine
| (1) |
A person may make an application to the Council, for renewal of registration of a veterinary medicine in Form E set out in the Third Schedule.
|
| (2) |
The Council shall, before renewing registration under this Regulation conduct a pharmaco-vigilance study on the manufacturer.
|
| (3) |
An applicant shall on receipt of approval for renewal of registration from the Council, make payment of the fees as prescribed in the Fifth Schedule.
|
|
| 25. |
Clinical and non-clinical trials and toxicity testing
| (1) |
The Council may require that a clinical and non-clinical trial and toxicity testing be done before the registration of a veterinary medicine.
|
| (2) |
Despite paragraph (1) a veterinary pesticide shall not be registered unless an efficacy and an acute toxicological study is carried out by an accredited laboratory.
|
| (3) |
A person who or institution which intends to apply for accreditation under this Regulation shall, before making such application, obtain approval from the Council.
|
| (4) |
A person who or institution which desires to conduct clinical and non-clinical trials and toxicity testing shall apply to the Council for accreditation.
|
| (5) |
The cost of any clinical trial or toxicity testing shall be borne by the applicant.
|
|
| 26. |
Conditions for registration of veterinary medicines and pesticides
| (1) |
A veterinary medicine and pesticide shall not be registered unless a laboratory analysis has been carried out by an accredited laboratory.
|
| (2) |
A person who intends to register a veterinary medicine shall submit to the Council a certificate of analysis from an accredited laboratory together with the application for registration of the veterinary medicine
|
|
| 27. |
Veterinary Medicines Registration Committee
| (1) |
There is hereby established a standing committee of the Council to be known as the Veterinary Medicines Registration Committee.
|
| (2) |
The Committee shall be responsible for matters relating to registration of veterinary medicines under these Regulations and in particular shall be responsible for —
| (a) |
evaluating applications for veterinary medicines registration and shall make recommendations to the Council; and
|
| (b) |
issuing provisional approval pending the issuance of the veterinary medicines registration certificate by the Council.
|
|
| (3) |
A provisional certificate issued under paragraph (2)(b) shall be valid for a period of not more than three months.
|
| (4) |
The Council shall consider an application made under this Regulation, and if satisfied of the safety, efficacy and quality of the veterinary medicine or veterinary pesticide, shall register the veterinary medicine in accordance with Form C1 and C2 set out in the Third Schedule.
|
| (5) |
Upon registration of a veterinary medicine under paragraph (4), the applicant shall be issued with a certificate of registration in Form D1 or D2 set out in the Third Schedule.
|
| (6) |
The Council may, while considering a veterinary medicine for registration under paragraph (4), approve the details as supplied by the applicant or approve it with such modifications as it may consider appropriate in respect of the following particulars—
| (a) |
the name under which the veterinary medicine may be sold;
|
| (c) |
the statement of the representations to be made for the promotion of the veterinary medicine in respect of the —
| (i) |
claim to be made for the veterinary medicine; |
| (ii) |
route of administration; |
| (iv) |
contra-indications, side effects and precautions, if any; |
| (vi) |
withdrawal period for food producing animals; |
| (vii) |
proposed category; and |
|
|
|
| 28. |
Rejection of application for registration of a veterinary medicine
The Council shall, if it is not satisfied as to the safety, efficacy or quality of the veterinary medicine, reject the application for the registration of the veterinary medicine and inform the applicant, in writing, the reasons for rejection.
|
| 29. |
Registration of veterinary medicine researched outside Kenya
| (1) |
The Council may, before registering a new veterinary medicine for which the research work has been conducted in another country and its efficacy, safety, and quality established in that country, require an investigation on the pharmaceutical, pharmacological and other aspects of the veterinary medicine to be conducted and clinical trials to be carried out as necessary to establish its safety, efficacy, quality and where applicable the biological availability to be established under local conditions.
|
| (2) |
Despite paragraph (1), the Council may register a new veterinary medicine and require the investigations and clinical trials specified therein to be conducted after its registration.
|
|
| 30. |
Suspension and revocation of certificate of registration
| (1) |
The Council may suspend or revoke a certificate of registration issued under these Regulations for such period as the Council may determine.
|
| (2) |
The Council shall not revoke or cancel the certificate of registration unless —
| (a) |
where matters stated in the application on which the certificate of registration was granted were false or incomplete in any material particular;
|
| (b) |
where provision of the certificate of registration has to a material extent been contravened by the holder of the certificate;
|
| (c) |
where the premises on which, or on part of which, a veterinary medicine is manufactured, assembled or stored by or on behalf of the holder of the certificate of registration are unsuitable for the manufacturing, assembling or storage of veterinary medicines; or
|
| (d) |
where new information has been discovered by the Council which renders the veterinary medicines unsafe or dangerous.
|
|
| (3) |
Where a certificate has been revoked under paragraph (2)(d), the Council shall on delivery of the information to the manufacturer —
| (a) |
inspect the plant to ensure all the medicine or the relevant batch has been destroyed;
|
| (b) |
disseminate the information to the public through a newspaper advertisement in at least two daily newspapers of nationwide circulation; and
|
| (c) |
recall any medicine which has been distributed.
|
|
|
| 31. |
Period for renewal of registration and retention of veterinary medicine
| (1) |
The Council may grant the renewal of registration as provided under regulation 24 for a period not exceeding five years.
|
| (2) |
On the expiry of five years a veterinary medicine may be retained in the register upon an application made by the certificate holder which shall —
| (a) |
include a declaration that the requirements met by the veterinary medicine during registration has not changed; and
|
| (b) |
include payment of the requisite fees.
|
|
|
PART IV – CLASSIFICATION AND CATEGORIZATION OF VETERINARY MEDICINES
| 32. |
Classification of Veterinary Medicines
| (1) |
The Cabinet Secretary shall classify veterinary medicines in the classes set out in Part I of the Second Schedule.
|
| (2) |
The Cabinet Secretary may, on the advice of the Council, vary the classification of the various veterinary medicines depending on the therapeutic use and current information for the medicine.
|
|
| 33. |
Categorization of Veterinary Medicines
| (1) |
The Cabinet Secretary shall categorize veterinary medicines as set out in Part II of the Second Schedule.
|
| (2) |
The Cabinet Secretary shall be guided by the Council on the categorization of veterinary medicines and shall place the veterinary medicines in the following categories —
| (a) |
Category I relating to prescription only medicine—Veterinary Surgeon which shall comprise two sub-categories as follows —
| (i) |
Category IA relating to Controlled Veterinary Medicines includes products which contain narcotic or psychotropic substances or other substances very dangerous at small quantities; |
| (ii) |
Category IB relating to other prescription only medicine, comprising products intended for administration following a diagnosis or clinical assessment by a veterinary surgeon or dispensed by a veterinary surgeon or a person with equivalent qualification; |
|
| (b) |
Category II relating to prescription only medicine veterinary surgeon, pharmacist, and veterinary technologist who has served for at least five years in a veterinary pharmacy which contains —
| (i) |
veterinary medicines for use in food-producing animals; |
| (ii) |
veterinary medicines in respect of which special precautions shall be taken in order to avoid any unnecessary risk to the target species, the person administering the products to the animal and the environment; Provided that the requirement in this paragraph shall not apply if all the following criteria are met— |
|
| (c) |
Category III relating to authorised veterinary medicine general sales list which may be traded by veterinary surgeons and all categories of veterinary paraprofessionals and includes—
| (i) |
pest control veterinary medicines provided that the medicine are sold in the original package without repackaging and with an intact label; |
| (ii) |
endoparasiticide veterinary medicines considered safe for use in food animals; |
| (iii) |
endoparasiticide veterinary medicines for use in non-food animals; or |
| (iv) |
any other product the Directorate may prescribe. |
|
| (d) |
Category IV which includes alternative veterinary medicine general sales list.
|
|
|
| 34. |
Use of veterinary pesticides
| (1) |
A veterinary pesticide shall be—
| (a) |
sold only in the original sealed and labeled package as registered by the Council;
|
| (b) |
stored in accordance with the instructions on the label on the original container; and
|
| (c) |
used according to the instructions on the label.
|
|
| (2) |
The empty container of a veterinary pesticide shall be disposed in accordance with the instructions given on the label.
|
| (3) |
A person who removes or defaces a label on a veterinary pesticide or package commits an offence.
|
| (4) |
A person who sells, dispenses or otherwise gives out a veterinary pesticide in contravention of this Regulation commits an offence and shall be liable, on conviction, to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months or to both.
|
|
PART V – VETERINARY PHARMACY
| 35. |
Standards of Veterinary Pharmacy
| (1) |
A veterinary pharmacy shall satisfy the following minimum standards—
| (a) |
be located away from known fire hazards;
|
| (b) |
be a separate entity from other veterinary operations such as veterinary clinic, ambulatory and laboratory services;
|
| (c) |
be separated from non-complementary businesses;
|
| (d) |
have restricted access by personnel to Category I and II veterinary medicine; and
|
| (e) |
be vermin-proofed, including protection against insects and rodents.
|
|
| (2) |
A veterinary pharmacy shall—
| (a) |
demonstrate adequate security for the safety of veterinary medicine;
|
| (b) |
demonstrate appropriate storage conditions that shall maintain the temperature, lighting and humidity requirements as prescribed in the manufacturers' specifications of veterinary medicines;
|
| (c) |
have the floor and the wall of the building constructed from materials that are easy to clean, impervious and resistant to corrosion by chemicals;
|
| (d) |
have lockable safety cabinets to protect the controlled veterinary medicines;
|
| (e) |
provide the personnel with protective clothing for use only within the premises;
|
| (f) |
be of appropriate size and have sufficient space for carrying out of all necessary operations provided for the orderly receipt, warehousing and dispatch of the various classes of veterinary medicines and, in particular, a quarantine area for isolation when necessary, including isolation of faulty packs and recalled or expired veterinary products;
|
| (g) |
provide a disposal system, acceptable to the Council, for safe disposal of expired veterinary medicines;
|
| (h) |
be operated by competent staff as provided for in the permit;
|
| (i) |
display warning notices indicating hazardous areas;
|
| (j) |
label the various sections in the business;
|
| (k) |
store veterinary medicines only in the designated areas, which shall be labeled;
|
| (l) |
have emergency lighting, fire-fighting equipment and fully equipped first aid kit;
|
| (m) |
display emergency protocols within the veterinary pharmacy advising personnel on procedures to follow in case of emergencies;
|
| (n) |
display standard operating procedures;
|
| (o) |
retain records of the movement of all veterinary medicines for at least five years; and
|
| (p) |
provide separate sanitary facilities for each gender.
|
|
|
| 36. |
Qualifications to operate a veterinary pharmacy
A person who is a veterinary surgeon, or holds an equivalent qualification in matters of pertaining to veterinary medicines as determined by the Council, may apply to the Council for a permit to undertake a veterinary pharmacy.
|
| 37. |
Council to approve veterinary pharmacy businesses
| (1) |
The Council shall have power to approve any of the following types of veterinary pharmacy businesses—
| (a) |
manufacturing of veterinary medicines;
|
| (b) |
wholesaling of veterinary medicines; and
|
| (c) |
retailing of veterinary medicines.
|
|
| (2) |
Upon approval of the veterinary business specified under paragraph (1), the Council shall register the veterinary pharmacy premises in Form J set out in the Third Schedule.
|
|
| 38. |
Practicing permit
| (1) |
A person shall not practice the business of veterinary pharmacy unless he holds a valid practicing permit issued by the Council.
|
| (2) |
A person who intends to practice the business of veterinary pharmacy shall make an application in Forms H1, H2 and H2, respectively of the Third Schedule.
|
| (3) |
The Council shall issue a practicing permit if the premises in which the applicant intends to operate has been approved by the Council in accordance with these Regulations.
|
| (4) |
An application for a practicing permit shall be in —
| (a) |
Form I.1 for veterinary pharmacy practitioner;
|
| (b) |
Form I.2 for a wholesaler dealer permit; or
|
| (c) |
Form I.3 for manufacturers' permit.
|
|
| (5) |
An applicant under paragraph (1) shall, on approval of the application, by the Council, pay the fee prescribed in the Fifth Schedule.
|
| (6) |
The Council, upon confirming that the applicant has satisfied the conditions of a permit, and upon payment of the fee under paragraph (4) issue an applicant with a practicing permit as set out in Form A set out in the Third Schedule for veterinary pharmacy practices and Form B for wholesale pharmacy practice.
|
| (7) |
The practice permit shall be valid for a period of one year commencing on the 1st of January and ending on the 31st of December of each year.
|
| (8) |
The Council shall by the 31st March of every year publish, in the Kenya Gazette or in a newspaper of wide national circulation, the registered veterinary pharmacy businesses.
|
| (9) |
A person who conducts the business of veterinary pharmacy in premises not registered by the Council under these Regulations commits an offence, and shall be liable, on conviction, to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months or to both.
|
|
| 39. |
Practice permit to be displayed
| (1) |
A person who carries on the business of a veterinary pharmacy shall display his practice permit on a conspicuous place within the premises in which the business is being carried on.
|
| (2) |
A person who contravenes the provisions of this Regulation commits an offence and shall be liable, on conviction, to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months or to both.
|
|
| 40. |
Wholesale permit
A person who intends to trade in veterinary medicines, in bulk , shall make an application to the Council for a wholesale permit and support the application with the following—
| (a) |
a certified copy of the certificate of registration or incorporation of the business name or body corporate and the memorandum and articles of association;
|
| (b) |
provide evidence that the business is under the management of a supervisor who—
| (i) |
is a registered veterinary surgeon permitted to carry out the business of veterinary pharmacy; and |
| (ii) |
is a member of the Board of directors of the body corporate, and who is not acting in a similar capacity for any other body corporate. |
|
|
| 41. |
Retail permit
| (1) |
A person who intends to trade in veterinary medicines as a retailer shall make an application to the Council for a retail permit in Form H set out in the Third Schedule.
|
| (2) |
This regulation shall be applicable to a manufacturer and a wholesaler of veterinary medicines who desires to carry out retail trade.
|
|
PART VI – VETERINARY PHARMACY AND COMPLEMENTARY BUSINESSES
| 42. |
Complementary businesses
| (1) |
A veterinary pharmacy may be conducted alongside complementary businesses including the sale of human medicines, horticultural chemicals, agro-forestry chemicals and fertilizer, animal foodstuffs, seeds and agricultural equipment.
|
| (2) |
The building or premises of a veterinary pharmacy stocking or trading in complementary businesses referred to in paragraph (1), shall be built or adapted in such manner as to provide a separate and distinct partition for the veterinary products from any other products.
|
| (3) |
In addition to the provisions of paragraph (2), the premises shall be built or adapted in such manner as to provide separate and distinct partitions for veterinary medicines and veterinary pesticides such that no mixing of the two classes of products is permissible.
|
|
| 43. |
Stock, supply and distribution of veterinary medicine
| (1) |
A veterinary medicine shall be supplied or distributed only through a wholesale or retail business registered as a veterinary pharmacy business.
|
| (2) |
A holder of wholesale or veterinary pharmacy permit issued by the Council may sell veterinary medicines in bulk to registered retail dealers.
|
| (3) |
A holder of a retail dealer permit, issued by the Council, may sell veterinary medicines to the public.
|
| (4) |
A person shall not supply authorised human medicinal product for administration to an animal other than a product supplied by a veterinary surgeon or in accordance with a written prescription from a veterinary surgeon.
|
| (5) |
A veterinary pharmacy practitioner who dispenses a veterinary medicine belonging to Category I or II to any member of the public without a prescription, commits an offence and shall be liable, on conviction, to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months or to both.
|
|
PART VII – MARKET AUTHORIZATION
| 44. |
Authorization to import or export veterinary medicine
| (1) |
A person who intends to import or export a veterinary medicine shall apply to the Council for a permit in Form K set out in the Third Schedule.
|
| (2) |
The Council shall in determining the application under paragraph (1) consider whether the applicant has met the conditions for importation and exportation in these Regulations, and if so, shall on payment of the prescribed fee, issue the applicant with an import permit in Form L set out in the Third Schedule.
|
| (3) |
A holder of a market authorization may apply to the Council for a wholesale permit to authorize him to import the veterinary medicine specified in the authorization.
|
| (4) |
An authorised wholesale dealer may import a veterinary medicine if —
| (a) |
the authorization covers the veterinary product; and
|
| (b) |
the wholesale dealer has acquired the written consent of the holder of the market authorisation in writing before importation.
|
|
| (5) |
A veterinary surgeon may, with approval of the Council, import an orphan veterinary medicine.
|
|
PART VIII – VETERINARY MEDICINE INSPECTORS
| 45. |
Appointment of veterinary medicine inspectors
The Council shall appoint duly qualified persons on such terms and conditions of service as it may deem appropriate, to serve as veterinary medicine inspectors.
|
| 46. |
Identification of veterinary medicines inspector
| (1) |
The Council shall publish in the Kenya Gazette every inspector appointed under regulation 45 and issue each inspector with an official identity card which shall have a passport size photo of the inspector, duly stamped and signed by the Chief Executive Officer or his authorized agent.
|
| (2) |
Every inspector shall, in conducting inspections under these Regulations, identify himself using the identity card issued under paragraph (1).
|
| (3) |
A person who ceases to be an inspector shall surrender his identify card to the Chief Executive Officer or his authorized agent.
|
|
| 47. |
Powers of veterinary medicines inspectors
| (1) |
An inspector shall have the power, at all reasonable times, to —
| (a) |
enter upon the premises of any manufacturer, distributor or veterinary pharmacy and to inspect any books, papers, records or writings, veterinary medicines, whether patent or otherwise, or any article stored or offered for sale or used in the business; or
|
| (b) |
enter any premises in which he has reasonable cause to suspect that a breach of the law has been or is being committed, and to make such examination and inquiry and to do such other activities, including impounding and seizing suspect veterinary products, closing suspect premises and the taking of samples, as may be necessary for the purpose of ascertaining whether the provisions of these Regulations are being complied with.
|
|
| (2) |
| (a) |
for the purpose of inspecting a veterinary medicine use Form F set out in the Third Schedule; and
|
| (b) |
for any impounding of suspect veterinary medicines, use Form G set out in the Third Schedule.
|
|
| (3) |
An inspector shall observe confidentiality in the findings of his inspection.
|
| (4) |
An inspector shall be liable for any act of negligence he may commit in the performance of his duties.
|
|
PART IX – FINANCIAL PROVISIONS
| 48. |
Funds of the Directorate
| (1) |
The funds of the Directorate shall comprise of —
| (a) |
such monies as may be appropriated by Parliament for the purposes of the Directorate;
|
| (b) |
such monies as may accrue to or vest in the Directorate in the course of the exercise of its functions under these Regulations; and
|
| (c) |
monies from any other source provided for, donated or lent to the Directorate.
|
|
|
| 49. |
Financial year
The financial year of the Directorate shall be the period of twelve months from first July to thirtieth of June.
|
| 50. |
Annual estimates
| (1) |
Three months before the commencement of each financial year, the Council shall cause to be prepared estimates of revenue and expenditure of the Directorate for that year.
|
| (2) |
The annual estimates shall make provision for all the estimated expenditure of the Directorate for the financial year concerned, and in particular shall provide for—
| (a) |
the payment of salaries, allowances and other charges in respect of the staff of the Directorate;
|
| (b) |
the payment of pensions, gratuities and other charges in respect of retirement benefits which are payable out of the funds of the Directorate;
|
| (c) |
the proper maintenance of buildings and grounds of the Directorate;
|
| (d) |
the acquisition, maintenance, repair and replacement of the equipment and other movable property of the Directorate; and
|
| (e) |
the creation of such reserve funds to meet future or contingent liabilities in respect of retirement benefits, insurance or replacement of buildings or equipment or in respect of such other matter as the Directorate may find appropriate.
|
|
| (3) |
The annual estimates shall be approved by the Council before the commencement of the financial year to which they relate and shall be submitted to the Cabinet Secretary for approval.
|
| (4) |
The Directorate shall not increase any sum provided in the estimates without the consent of the Cabinet Secretary.
|
|
| 51. |
Investment of funds
The Council may invest any of the funds of the Directorate in a manner approved by the Treasury for the investment of trust funds.
|
| 52. |
Accounts and Audit
| (1) |
The Council shall cause to be kept all proper books and records of accounts of the income, expenditure, assets and liabilities of the Directorate.
|
| (2) |
The accounts of the Directorate shall be audited in accordance with the Public Finance Management Act, 2012 (No. 18 of 2012).
|
|
PART X – OFFENCES
| 53. |
Offences
A person commits an offence if that person —
| (a) |
imports, exports, manufactures, stores, distributes, sells or otherwise handles a veterinary medicine that has not been registered under these Regulations;
|
| (b) |
imports a veterinary medicine without a permit issued under these Regulations;
|
| (c) |
manufactures, stores, distributes or sells a veterinary medicine in premises which have not been registered under these Regulations;
|
| (d) |
presents for sale or distribution, expired, adulterated, Counterfeit or unlabeled veterinary medicines;
|
| (e) |
sells a veterinary medicine in any form other than the original sealed and labeled package, as registered with the Directorate;
|
| (f) |
sells or distributes a veterinary medicine in any other area other than a registered premises;
|
| (g) |
uses a veterinary medicine for purposes other than that for which it was registered, prescribed or dispensed;
|
| (h) |
supplies human medicinal product for administration to an animal in contravention of these Regulations;
|
| (i) |
is in possession of a Category I or Category II veterinary medicine other than in accordance with these Regulations;
|
| (j) |
advertises a veterinary medicine in contravention of these Regulations;
|
| (k) |
buys a veterinary medicine for resale in bulk while not being in possession of a wholesale dealer permit issued under these Regulations;
|
| (l) |
sells a veterinary medicine to the public while not being in possession of a retail dealer's permit issued in accordance with these Regulations;
|
| (m) |
undertakes research on imported and unregistered veterinary medicine without authorization from the Council;
|
| (n) |
obstructs or fails to assist the Council or veterinary medicine inspector in the performance of their lawful duties under these Regulations;
|
| (o) |
provides the Council or inspector with false or misleading information;
|
| (p) |
operates a veterinary pharmacy after it has been closed by an inspector; or
|
| (q) |
contravenes any provision of these Regulations.
|
|
| 54. |
Officers of corporations
If an institution commits an offence under these Regulations, any officer, director or agent of the corporation who directed, authorized, assented to, acquiesced in, or participated in the commission of the offence shall be a party to and shall be considered to have committed the offence and shall be liable on conviction to the punishment provided for the offence, whether or not the corporation has been prosecuted or convicted.
|
| 55. |
Passing confidential information without authorization
A member of the Council or staff of the Directorate, who —
| (a) |
being in possession of confidential information, however obtained without authorization of the Council —
| (ii) |
attempts, offers or threatens to divulge it, |
|
other than in accordance with these Regulations or other written law; or
| (b) |
willfully obtains or seeks to obtain confidential information to which he is not entitled, commits an offence.
|
|
| 56. |
Penalties
A person who contravenes these Regulations where no Penalties. penalty has been prescribed, shall on conviction be liable to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months, or to both.
|
PART XI – GENERAL PROVISIONS
| 57. |
Advertising veterinary standards for medicines
| (1) |
A person who intends to advertise a veterinary medicine, shall make an application to the Council for authority to advertise.
|
| (2) |
The Council may, on payment of the requisite fee and subject to such conditions as the Council or these Regulations may impose, grant the authorization applied for under paragraph (1).
|
| (3) |
An advertisement for a veterinary medicine shall not be misleading or contain any medicinal claim that is not in the summary of the product characteristics registered with the Council.
|
| (4) |
A veterinary medicine listed under Category I and II shall not be advertised unless —
| (a) |
in the case of a veterinary medicine listed under Category I, the advertisement is aimed at veterinary surgeons; and
|
| (b) |
in the case of a veterinary medicine listed under Category II, the advertisement is aimed at veterinary surgeons or suitably qualified persons as recognized by the Council.
|
|
| (5) |
A veterinary medicine listed in Category III and IV may be advertised to general members of the public.
|
|
| 58. |
Exemptions to Regulations
These Regulations do not apply to —
| (a) |
inactivated autogenous vaccine that is manufactured, on the instructions of a veterinary surgeon, from pathogens or antigens obtained from an animal and used for the treatment of that animal or —
| (i) |
other animals on the same site; |
| (ii) |
animals intended to be sent to those premises; or |
| (iii) |
animals on a site that receives animals from those premises; or |
|
| (b) |
animal foodstuff supplements with no therapeutic claim.
|
|
| 59. |
Discretion of Council
The Council shall have the discretion to —
| (a) |
approve the importation and use of any veterinary medicine for handling emergency situations;
|
| (b) |
cancel the registration of any veterinary medicine that is considered to be harmful to animal and human health and the environment; or
|
| (c) |
restrict the use of any specified veterinary medicine.
|
|
| 60. |
Copy of certificate or authorization
| (1) |
A person who has been issued with a certificate or authorization under these Regulations may, if the certificate or authorization is defaced, damaged or lost, on application to the Council and on payment of the prescribed fee, be issued with a copy of the certificate.
|
| (2) |
The copy of a certificate or authorization issued under paragraph (1) shall —
| (i) |
be issued only where the document is in force at the time the application is made; |
| (ii) |
be valid for the same period as the original document; and |
| (iii) |
bear the words "DUPLICATE COPY". |
|
|
| 61. |
Fees
Any fees payable under this Act is as set in the Fifth Schedule.
|
| 62. |
Supercession
These Regulations shall supersede any other Regulations on matters concerning veterinary medicines.
|
| 63. |
Transition and validity of licences and permits
Upon the commencement of these Regulations any certificate in force shall be deemed to have been issued under these Regulations and shall remain in force until its expiry.
|
FIRST SCHEDULE
CONDUCT OF THE AFFAIRS OF THE COUNCIL
| 1. |
Meetings of the Council.
The Council shall meet at least four times in a year and not more than eight times a year, except in case of an emergency, for the transaction of its business, and such meetings shall be held at such places and times and on such days as the Council may determine.
|
| 2. |
Chairperson and vice-charperson
| (1) |
The Council shall at its first meeting elect its vice-chairperson from among the three members appointed under regulation 8(3)(f).
|
| (2) |
The Chairperson shall preside at all meetings of the Council at which he is present, and in case of his absence the vice-chairperson shall preside, but in the absence of both the chairperson and vice-chairperson, members present and constituting a quorum shall elect one among their number to be the chairperson for purposes of the meeting.
|
|
| 3. |
Quorum
The quorum of a meeting of the Council shall be five, at least three of whom shall be veterinary surgeons.
|
| 4. |
Decisions of Council
The decisions of the Council shall be by a simple majority of the votes of the members present, but in the case of an equality of votes, the Chairperson or person presiding shall have a casting vote.
|
| 5. |
Minutes
| (1) |
The Minutes of each meeting shall be kept in the minute book, after they have been confirmed by the Council and signed by the Chairperson at a subsequent meeting of the Council.
|
| (2) |
The deliberations and minutes of meetings of the Council shall be confidential.
|
|
| 6. |
Vacancy not to invalidate proceedings
No proceedings of the Council shall be invalid by reason only of a vacancy among the members thereof.
|
| 7. |
Council to determine own procedure
Subject to this Schedule, the Council may determine its own procedure.
|
| 8. |
Invitation of other persons
The Council may invite any person to participate in its deliberations but a person who has been invited shall have no right to vote.
|
SECOND SCHEDULE
CONDUCT OF THE AFFAIRS OF THE COUNCIL
PART I – CLASSES OF VETERINARY MEDICINES
| 1. |
List of veterinary pharmaceuticals.
| (iii) |
Analgesics and anti-inflammatories |
| (iv) |
Drugs acting on the nervous system |
| (vi) |
Other systemic therapeutic agents |
| (vii) |
Local therapeutic agents |
|
| 2. |
| (v) |
Probiotics and enzymes. |
|
| 4. |
| (ii) |
Veterinary medicine administration devices. |
| (iii) |
Any other material and equipment of veterinary relevance. |
|
| 5. |
List of alternative medicines.
| (iii) |
Performance enhancers. |
|
| 6. |
| (v) |
Any other pesticides of veterinary relevance |
| (vi) |
And other ecto-parasiticides |
| (vii) |
substances used for euthanasia |
|
PART II – CATEGORIES OF VETERINARY MEDICINES
(a) Prescription Only Medicine—Veterinary surgeon (abbreviated to POM-V);
Category I A POM-V Controlled Veterinary Medicine
| 11. |
Morphine and its derivatives.
|
| 13. |
Methadone (amidone); its salts.
|
| 16. |
Phenoperidine; its salts.
|
| 19. |
Any other veterinary medicine relevant in the category.
|
Category IB — Other POM - V
CATEGORY II —PRESCRIPTION ONLY MEDICINE (POM-VT)
Prescription Only Medicine—Veterinary surgeon
CATEGORY III — Authorized Veterinary Medicines —General Sales List (AVM-GSL)
CATEGORY IV- Alternative Veterinary Medicine — General Sales List (AItVM-GSL)
THIRD SCHEDULE
FORM A
APPLICATION FOR REGISTRATION OF A VETERINARY MEDICINE
(to be submitted as one original hard-copy and one electronic copy in MS-Word)
The Registrar
Veterinary Medicines Directorate
KABETE




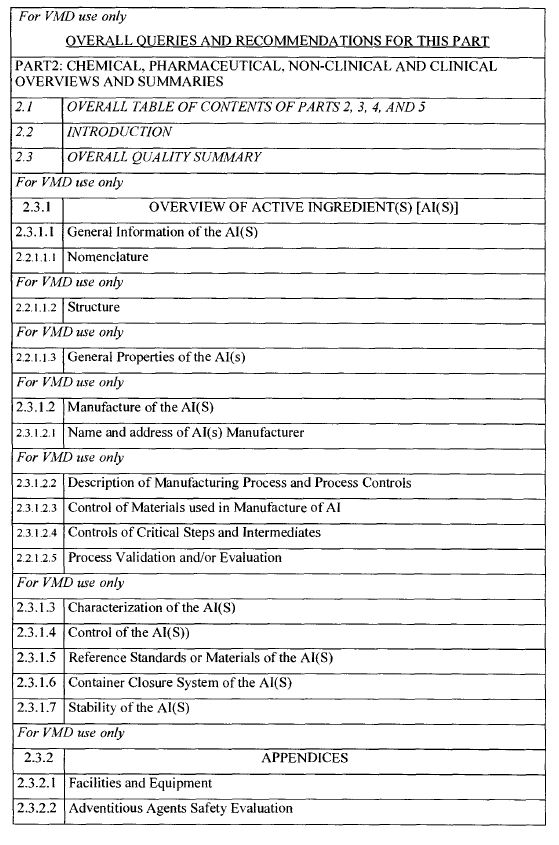



APPLICATION FOR THE REGISTRATION OF VETERINARY PESTICIDES
[to be submitted as one original hard-copy and one electronic copy (in MS-Word)]
The Director
Veterinary Medicines Directorate
KABETE
Information for Applicants
| 1. |
The application form must be completed by a duly authorized person.
|
| 2. |
Every application must be accompanied by 3 copies of the draft label as per VMD requirements.
|
| 3. |
The applicant may be required to submit:-
| (a) |
a sample of the veterinary pesticide;
|
| (b) |
a sample of the technical grade of its active ingredient;
|
| (c) |
a sample of the laboratory standard of its active ingredient;
|
| (d) |
any other sample as may be required by the Council.
|
|
| 4. |
The application must be accompanied by a technical dossier as per VMD data Requirements.
|
| 5. |
An applicant who is not a resident in Kenya shall appoint as an agent a person who is permanently resident in Kenya and duly recognized by the Veterinary Medicines Directorate.








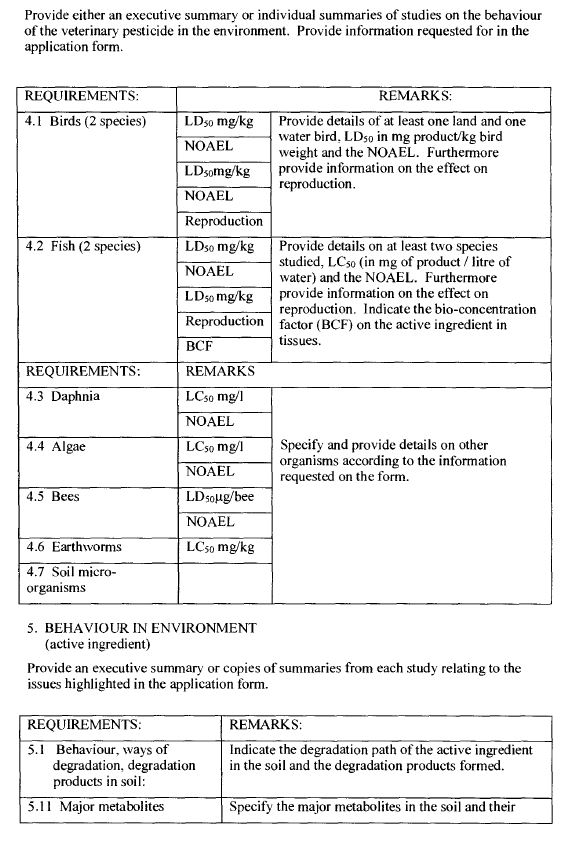
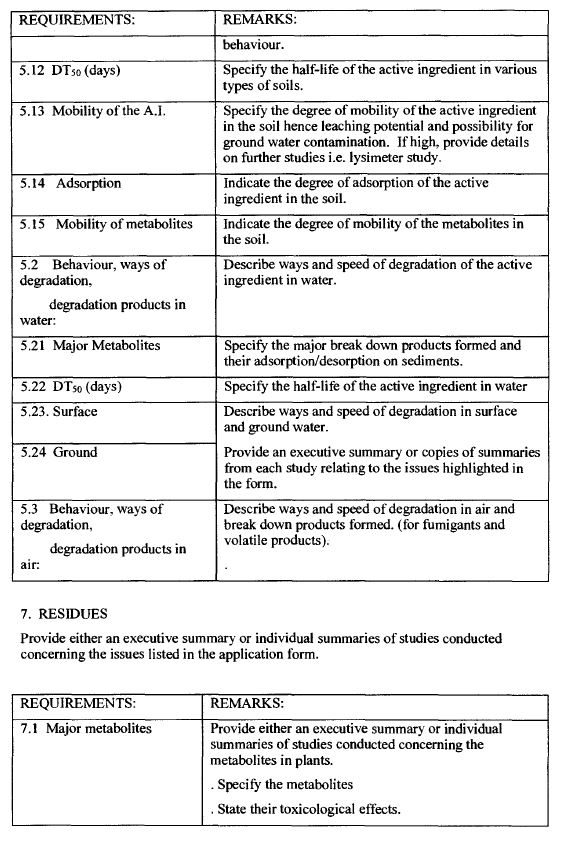
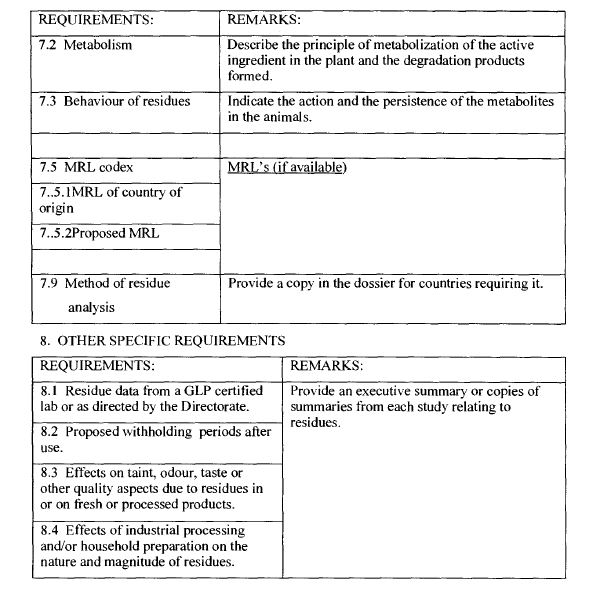
|
SUMMARY OF THE DATA SUBMITTED TO THE VMD FOR REGISTRATION OF A VETERINARY PESTICIDES
FOURTH SCHEDULE
CODE OF CONDUCT FOR MEMBERS OF AND EMPLOYEES OF THE
| 1. |
Impartiality and independence of members
| (1) |
Every member and employee of the Directorate shall impartially and independently perform the functions of a member in good faith and without fear, favour or prejudice, and without influence from—
| (a) |
the National or County Government;
|
| (d) |
any candidate participating in an election; or
|
| (e) |
any other person or authority.
|
|
|
| 2. |
Independence from political or public office
| (1) |
A member or employee of the Directorate shall not, during tenure of office, be eligible for—
| (a) |
appointment or nomination to a political office; or
|
| (b) |
appointment to another public office.
|
|
| (2) |
A member of the Directorate may not—
| (a) |
by their membership, association, statement, conduct or in any other manner place in jeopardy the perceived independence of the member, or in any other manner harm the credibility, impartiality, independence or integrity of the Directorate;
|
| (b) |
make private use of or profit from any confidential information gained as a result of being a member of the Directorate; or
|
| (c) |
divulge any information to any third party, save in the of official duty.
|
|
|
| 3. |
Disclosure of conflicting interests
| (1) |
If a member or an employee is directly or indirectly interested in any contract, proposed contract or other matter before the Council and is present at any meeting of the Council at which the contract, proposed contract or other matter is the subject of consideration, the member or employee shall, at the meeting and as soon as practicable after the commencement thereof, disclose the fact and shall not take part in the consideration or discussion of, or vote on, any questions with respect to the contract or other matter or be counted in the quorum of the meeting during consideration of the matter.
|
| (2) |
A member or employee whose personal interest conflicts with their official duties shall—
| (a) |
declare the personal interests to their supervisor or other appropriate person or body in writing and comply with any directions given to avoid the conflict; and
|
| (b) |
refrain from participating in any deliberations with respect to the matter.
|
|
| (3) |
No member of staff of the Directorate shall transact business with the Directorate directly or indirectly.
|
|
| 4. |
Professionalism
A member or employee of the Directorate shall—
| (a) |
perform their duties in a manner that promotes and maintains public confidence in the Directorate;
|
| (b) |
treat the public and colleagues with courtesy and respect;
|
| (c) |
discharge all their duties in a professional, timely and efficient manner and in line with the rule of law; and
|
| (d) |
respect the rights and freedom of all persons that he may interact with.
|
|
| 5. |
Improper enrichment
A member or employee of the Directorate shall not—
| (a) |
use their office or organization to improperly enrich themselves or others;
|
| (b) |
accept or request gifts or personal favours from any person who may have a commercial interest with the Directorate or any other interest that may be affected by the normal business of the Service; or
|
| (c) |
use information that is acquired during the course of their duties or connected to their duties for their benefit or for the benefit of others
|
|
| 6. |
Integrity in private affairs
A member or employee shall conduct their private affairs in manner that maintains public confidence in the integrity of their office and the Directorate as a whole and shall—
| (b) |
not neglect their financial obligations;
|
| (c) |
submit an annual declaration of their income, assets and liabilities to the Commission responsible for such declarations from public officers;
|
| (d) |
not engage in political activity that may compromise or be seen to compromise the neutrality of their office, or the Directorate; and
|
| (e) |
not preside over or play a central role in the organization of a fundraising activity.
|
|
| 7. |
Sexual harassment
| (1) |
A member or employee shall not sexually harass a member of the public or colleague.
|
| (2) |
Sexual harassment includes —
| (a) |
making a request or exerting pressure for sexual activity or favours;
|
| (b) |
making intentional or careless physical contact that is sexual in nature; or
|
| (c) |
making gestures, jokes or comments, including innuendoes regarding another person's sexuality.
|
|
|
| 8. |
Nepotism
A member or employee shall not practice favouritism on the grounds of tribe, race, kin, culture, sex or acquaintance or otherwise in performance of their duties.
|
| 9. |
Application of the Public Officers Ethics Act
This Code is in addition to the provisions of the Public Officers Ethics Act and where there is a conflict between the Code and these Regulations, the provisions of the Act shall prevail.
|
| 10. |
Breach of code
Any breach of the Code by a member or officer of the Service shall be treated as gross misconduct.
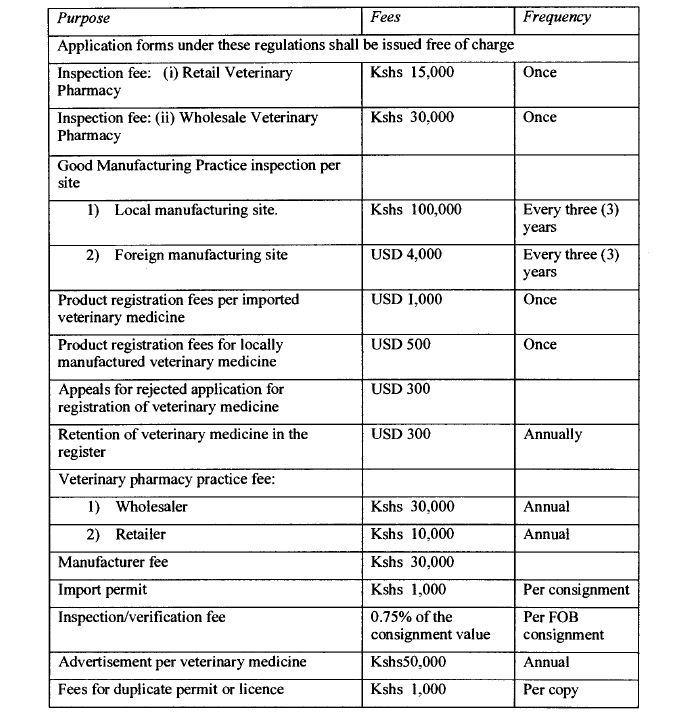
|
THE VETERINARY SURGEONS AND VETERINARY PARA-PROFESSIONALS (VETERINARY MEDICINES DIRECTORATE) REGULATIONS
IN EXERCISE of the powers conferred by section 39 (1) and 45 (1) of the Veterinary Surgeons and Veterinary Para-professionals Act, 2011, the Cabinet Secretary for Agriculture, Livestock and Fisheries makes the following regulations:—
[L.N. 293/2017, r. 2.]
PART I – PRELIMINARY
| 1. |
Citation
These Regulations may be cited as the Veterinary Surgeons and Veterinary Paraprofessionals (Veterinary Medicines Directorate) Regulations, 2015.
|
| 2. |
Interpretation
In these Regulations, unless the context otherwise requires—
"accredited laboratory" means a laboratory recognized as an accredited laboratory by the Directorate;
"advertisement" means any written or visual notice, circular, label, or wrapper, or other descriptive matter, verbal statement or reference appearing in any newspaper, television, film or mass media or brought to the attention of the public in any other form, which is intended to promote the sale of a veterinary medicine;
"alternative medicine" means the unrefined plant, animal and mineral substances commonly used in traditional animal treatments;
"Cabinet Secretary" means the Cabinet Secretary responsible for matters relating to veterinary service;
"Chief Executive Officer" means the Chief Executive Officer of the Directorate appointed under regulation 13;
"Committee" means the Veterinary Medicines Registration Committee established under regulation 27;
"controlled veterinary medicine" means a veterinary medicine specified in the Fourth Schedule as a Category I or Category II of veterinary medicine;
"Conventional medicines" means the regular and standardized veterinary medicines;
"Council" means the Council of the Directorate comprised under regulation 8 (1);
"crude drug" means an unrefined medicine of biological origin;
"Directorate" means the Veterinary Medicines Directorate established under regulation 5 and as envisaged under section 39(2)(a) of the Act;
"dispense" means the sale or supply of a veterinary medicine by a veterinary surgeon or other person authorised in accordance with these Regulations;
"inspector" means a person appointed as an inspector under regulation 45;
"manufacture" means any stage in the manufacturing of a veterinary medicine until the finished product is ready for sale in its final form as specified in the marketing authorization, and includes repackaging, repacking or labeling of a veterinary medicine in an authorized facility but does not include the breaking open of the package of a veterinary medicine by retailers;
"market authorization" means registration of a veterinary medicine by the Council and the issuance of a registration certificate under regulation 23;
"orphan veterinary medicine" means a veterinary medicine that is not economical to trade in but is required for specific medical use;
"pharmaco-vigilance" means the routine inspections and surveys carried out in the veterinary medicines market to safeguard general animal and human health and trade;
"quality assurance standards" means the good manufacturing practice, good laboratory practice, good veterinary practice or any other standard developed by an international standardization body, the East African Community Standards Committee or the Kenya Bureau of Standards which the Cabinet Secretary, on the advice of the Council, shall recognize through the Gazette as a quality assurance standard for the purposes of these Regulations;
"register" means a register maintained by the Council containing the details of—
| (a) |
premises which have been issued with a permit in accordance with these Regulations; or
|
| (b) |
veterinary medicines registered in the country;
|
"retailer" means a veterinary pharmacy registered by the Council for the sale of veterinary medicines to the end users;
"specialized committees" means persons who are not Council members;
"veterinary medicines inspector" means an inspector appointed by the Council pursuant to regulation 45;
"veterinary pesticide" means a veterinary medicine used as a pest control product on animals or the animals' environment;
"veterinary pharmaceutical" means a chemical substance formulated or compounded as a single active ingredient or in any combination of the chemical substances, for veterinary curative use;
"veterinary pharmacy" means a business authorized by the Council to stock, dispense or distribute veterinary medicines for sale;
"veterinary pharmacy assistant" means a veterinary para-professional trading in veterinary medicines listed under Category III of the Fourth Schedule;
"veterinary pharmacy practice" means the business of veterinary pharmacy carried out by veterinary surgeons, veterinary para-professionals or any other person permitted by the Council to carry out veterinary pharmacy practice;
"veterinary product" means veterinary medicines and veterinary pesticides;
"wholesale" means a manufacturer or veterinary pharmacy approved by the Council to trade in bulk in the supply of veterinary medicines to wholesalers or retailers in the original outer pack-sizes.
|
| 3. |
Objectives and purpose of the Regulations
The object and purpose of these Regulations is to—
| (a) |
regulate the manufacture, importation, exportation, registration, distribution, prescription and dispensing of veterinary medicines and the practice of veterinary pharmacy in Kenya; and
|
| (b) |
advise the Kenya Veterinary Board in relation to all aspects listed under paragraph (a).
|
|
| 4. |
Application
Subject to regulation 58, these Regulations shall apply to all conventional and alternate veterinary medicine.
|
PART II – THE VETERINARY MEDICINES DIRECTORATE
| 5. |
Establishment of the Directorate
| (1) |
There is hereby established a Directorate to be known as the Veterinary Medicines Directorate whose management shall vest on a Council appointed under regulation 8.
|
| (2) |
The Directorate shall be a body corporate with perpetual succession and a common seal and shall in its corporate name be capable of—
| (b) |
taking, purchasing, or otherwise acquiring, holding, charging or disposing of movable and immovable property;
|
| (c) |
borrowing and lending money; and
|
| (d) |
doing all such other things or acts as may lawfully be done by a body corporate.
|
|
|
| 6. |
Functions of the Directorate
The functions of the Directorate are to —
| (a) |
formulate and enforce quality assurance standards in the in the manufacture, distribution and use of veterinary medicines to safeguard human and animal health and the environment;
|
| (b) |
in consultation with the Director of Veterinary Services, regulate the use of veterinary medicine for the treatment of animals under the Animal Diseases Act (Cap. 364);
|
| (c) |
consider applications for approval for market authorization of veterinary medicines;
|
| (d) |
set quality assurance standards for training in the management of veterinary medicines as directed by the Kenya Veterinary Board;
|
| (e) |
collaborate with the Kenya Veterinary Board in regulating training in the management of veterinary medicines;
|
| (f) |
inspect and approve premises in which the manufacture, sale or supply of veterinary medicine is conducted;
|
| (g) |
appoint and gazette veterinary medicine inspectors;
|
| (h) |
establish the standard operating procedures for veterinary medicine inspectors;
|
| (i) |
regulate veterinary pharmacy practices;
|
| (j) |
categorize veterinary medicines and the qualification of persons authorized to trade in each category and review the categories every five years;
|
| (k) |
regulate clinical and non-clinical trials of veterinary medicines by individuals and institutions to be involved in the trials;
|
| (l) |
regulate the manufacture, importation, exportation, handling, advertisement, labeling, sale and disposal of veterinary medicines;
|
| (m) |
register all veterinary medicines manufactured or imported for use in the country or exported from the country;
|
| (n) |
monitor the market for and take measures necessary for the elimination of trade in illegal and counterfeit veterinary medicines;
|
| (o) |
establish systems of pharmaco-vigilance and conduct pharmaco-vigilance of veterinary medicines through regular inspections and surveys;
|
| (p) |
enforce good manufacturing practice for veterinary medicines as approved by the Council;
|
| (q) |
develop, apply and from time to time review guidelines to be used in the inspection and ensuring compliance with good manufacturing practice;
|
| (r) |
ensure that the promotion and marketing of veterinary medicine is in accordance with the approved product information;
|
| (s) |
publish, on an annual basis, a notice in the Kenya Gazette inviting the public to note and inspect the register of veterinary medicines and the list of approved veterinary pharmacy practices within such period and at such place as may be specified in the notice;
|
| (t) |
consider, grant, issue or revoke authorizations and certificates in accordance with these Regulations;
|
| (u) |
collaborate with other regulatory agencies including the Public Health (Standards) Board in section 27 of the Food, Drugs and Chemical Substances Act (Cap. 254), the Pest Control Products Board established under section 5 of the Pest Control Products Act (Cap. 346) and the Central Board of Health established under section 3 of the Public Health Act (Cap. 242) to carry out its mandate; and
|
| (v) |
undertake any other thing necessary for the effective carrying out of its mandate under this or any other Act.
|
|
| 7. |
Powers of the Council
The Council shall have all the power necessary or expedient for the effective discharge of its functions under these Regulations and any other law, and in particular the power to—
| (a) |
control, supervise and manage the assets and liabilities of the Directorate;
|
| (b) |
determine the provision to be made for capital and recurrent expenditure and for the reserves of the Directorate;
|
| (c) |
seek and receive any grants or donations and make legitimate disbursements from such grants and donations for its purposes;
|
| (d) |
levy fees and charges for its services as provided in these Regulations;
|
| (e) |
enter into association with other bodies within or outside Kenya which the Council may consider desirable or appropriate;
|
| (f) |
invest funds of the Directorate not immediately required in securities in which trustees are empowered to invest under the Trustee Act (Cap. 167), and in other securities which may be approved for the purpose, by the Cabinet Secretary for the time being responsible for Finance;
|
| (g) |
establish and support investment and trust funds for the benefit of employees or ex-employees of the Directorate or dependants of such persons, to grant pension, benefits and allowances and to make such payments towards insurance as required under the relevant laws;
|
| (h) |
open and operate such accounts as are necessary for the funds of the Directorate, with a bank or financial institution licensed to conduct banking business under the Banking Act (Cap. 488);
|
| (i) |
recruit, support, discipline or dismiss the staff and inspectors of the Directorate;
|
| (j) |
in consultation with Salaries Remuneration Commission, determine the terms and conditions of employment of the staff and inspectors of the Directorate;
|
| (k) |
establish branch offices of the Directorate, to the extent that is practicable, to ensure accessibility of its services by all Kenyans;
|
| (l) |
superintend, regulate and assist branch offices, auxiliaries, committees and other forms of organizations established to advance the interest of the Directorate;
|
| (m) |
perform all things necessary or incidental to attain the objectives of the these Regulations or any other written law.
|
|
| 8. |
Composition and appointment of the Council
| (1) |
The Council shall be appointed by the Cabinet Secretary and shall consist of—
| (a) |
the Director of Veterinary Services who shall be the Chairperson;
|
| (b) |
the Registrar of the Pharmacy and Poisons Board;
|
| (c) |
the Chief Executive Officer of the Kenya Veterinary Board;
|
| (d) |
the Principal Secretary for the time being responsible for Finance;
|
| (e) |
the Principal Secretary for the time being responsible for animal health matters;
|
| (f) |
three veterinary surgeons nominated by the Kenya Veterinary Board;
|
| (g) |
one veterinary technologist from the veterinary pharmaceutical industry nominated by the Kenya Veterinary Board; and
|
| (h) |
the Chief Executive Officer of the Directorate who shall be the Secretary to the Council and shall be an ex-officio member.
|
|
| (2) |
A person appointed under paragraph (1)(f) shall be nominated by the Kenya Veterinary Board from a list of five names drawn from the veterinary pharmaceutical industry including a trainer in veterinary pharmacology submitted by a registered professional association representing the interests of veterinary surgeons countrywide.
|
| (3) |
A person appointed under paragraph (1)(g) shall be nominated by the Kenya Veterinary Board from a list of three names from the veterinary pharmaceutical industry submitted by the registered association representing the interests of veterinary paraprofessionals.
|
| (4) |
The nominating bodies referred to under paragraphs (2) and (3) shall observe Constitutional principles relating to gender, youth, persons with disability and minorities in identifying the persons whose names shall be submitted for nomination by the Kenya Veterinary Board for appointment to the Council.
|
| (5) |
The Cabinet Secretary shall appoint the members of the Council by notice in the Kenya Gazette.
|
| (6) |
The Council shall conduct its affairs in the manner set out in the First Schedule.
|
| (7) |
The members of the Council shall hold office for a term of three years and shall be eligible for re-appointment for one further term.
|
| (8) |
The members appointed under paragraph (1)(b), (d) and (e) may, in writing, designate representatives to attend the meetings of the Council on their behalf.
|
| (9) |
The members of the Council in paragraphs (f), (g) and (h) may—
| (a) |
at any time resign from office by notice in writing to the Cabinet Secretary;
|
| (b) |
be removed from office by the Cabinet Secretary, on the advice of the Council if the member —
| (i) |
is declared bankrupt; |
| (ii) |
is absent from three consecutive meetings of the Council, without the permission of the Chairperson; |
| (iii) |
is convicted of a criminal offence and, sentenced to a term of imprisonment of more than six months; |
| (iv) |
is unable or unfit, due to physical or mental illness, to perform the functions of his office; or |
| (v) |
has failed to comply with the provisions of Chapter Six of the Constitution. |
|
|
| (10) |
The Cabinet Secretary shall, on the recommendation of the Council, appoint a relevant person to serve in the place of any member of the Council in the case of death, resignation, absence from Kenya for six consecutive months without authorization or more or removal from office under paragraph (9)(b) and the person appointed under this sub-Regulation shall serve until the end of the term of the Council.
|
| (11) |
Where a vacancy arises under paragraph (10), the Cabinet Secretary shall undertake a fresh recruitment process to fill the vacancy.
|
|
| 9. |
Co-option of specialized persons
Pursuant to section 10 of the Act, the Council may for the effective discharge of its functions co-opt into the committees such persons with technical expertise or knowledge for the better carrying out of the functions of the Directorate.
|
| 10. |
Meetings of the Council
The conduct and regulation of the business and affairs of the Council shall be as provided in the First Schedule but subject thereto, the Council shall regulate its own procedure.
|
| 11. |
Conflict of interest
| (1) |
If any person is present at a meeting of the Council or any committee at which any matter is the subject of consideration and in which matter that person or that person's spouse or relative is directly or indirectly interested in a private capacity, that person shall as soon as is practicable after the commencement of the meeting declare such interest and shall not, unless the Council or committee otherwise directs, take part in any consideration or discussion of, or vote on any question connected to such matter.
|
| (2) |
The disclosure of interest shall be recorded in the minutes of the meeting at which it is made.
|
| (3) |
A member or employee of the Council shall not carry out an business or trade with the Council.
|
| (4) |
A member or staff of the Council who contravenes this Regulation commits an offence and is liable, on conviction, to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months or to both.
|
|
| 12. |
Common seal and logo
| (1) |
The Directorate shall have a common seal which shall be kept by the Chief Executive Officer.
|
| (2) |
The affixing of the seal shall be authenticated by signature of the chairperson and the Chief Executive Officer and in his absence, the signature of any other member authorized by resolution of the Council.
|
|
| 13. |
Chief Executive Officer
| (1) |
There shall be a Chief Executive Officer who shall be appointed by the Council through a competitive process and whose terms and conditions of service shall be determined by the Council in the instrument of appointment.
|
| (2) |
A person shall be qualified for appointment as a Chief Executive officer, if that person—
| (b) |
holds a degree in veterinary medicine from a university recognized by the Kenya Veterinary Board;
|
| (c) |
has at least ten years professional experience in veterinary medicine regulation, of which at least five years are at senior management level; and
|
| (d) |
satisfies the requirements of Chapter Six of the Constitution.
|
|
| (3) |
The Chief Executive Officer shall be an ex-officio member of the Council but shall have no right to vote at any meeting of the Council.
|
| (4) |
The Chief Executive Officer shall hold office for a term of five years and shall be eligible for reappointment for one further term.
|
| (5) |
The Chief Executive Officers shall, in the performance of the functions and duties of office. be responsible to the Council.
|
| (6) |
Without prejudice to the provisions of paragraph (5), the Chief Executive Officer shall—
| (a) |
| (i) |
carrying into effect the decisions of the Council; |
| (ii) |
day-to-day administration and management of the affairs of the Directorate; |
| (iii) |
supervision of the staff of the Directorate; |
|
| (b) |
be the Registrar of veterinary medicines; and
|
| (c) |
perform such other duties as may be assigned by the Council.
|
|
|
| 14. |
Removal of the Chief Executive Officer
| (1) |
The Chief Executive Officer may be removed from office by the Council in accordance with the terms and condition of service, for—
| (a) |
inability to perform functions of the office arising out of physical or mental infirmity;
|
| (b) |
gross misconduct or misbehavior;
|
| (c) |
incompetence or negligence of duty;
|
| (d) |
violation of the Constitution and any other written law; or
|
| (e) |
any other grounds specified in the terms and conditions of service.
|
|
| (2) |
Where the question of the removal of the Chief Executive Officer under paragraph (1) arises, the Council shall —
| (a) |
inform the Chief Executive Officer in writing of the reasons for the intended removal; and
|
| (b) |
provide the Chief Executive Officer with the opportunity to be heard in accordance with the principles of fair administrative action safeguarded under Article 47 of the Constitution.
|
|
|
| 15. |
Veterinary medicines inspectors
| (1) |
The Council shall employ such veterinary medicines inspectors as it deems necessary.
|
| (2) |
A person shall be qualified for appointment as a veterinary medicines inspector if that person —
| (a) |
is a veterinary surgeon or technologist; or
|
| (b) |
holds at least a diploma in animal health or other qualification recognized by the Council; and
|
| (c) |
has at least five years post qualification experience in a relevant field.
|
|
|
| 16. |
Staff
The Council may employ such staff as it deems appropriate for the effective performance of its functions.
|
| 17. |
Code of conduct
The members and the employees of the Council shall subscribe to the leadership and integrity code set out in the Fourth Schedule.
|
| 18. |
Protection from personal liability
Nothing done by a member of the Council or by any person working under the instructions of the Council shall, if done in good faith for the purpose of executing the powers, functions or duties of the Council under these Regulations render such member or officer personally liable for any action, claim or demand.
|
PART III – MANUFACTURING, IMPORTATION AND REGISTRATION OF VETERINARY MEDICINES
| 19. |
Veterinary medicines to be registered
| (1) |
A person shall not import, manufacture sell, transport or distribute any veterinary medicine in Kenya unless that veterinary medicine has been registered in accordance with the provisions of these Regulations.
|
| (2) |
Every person transporting a veterinary medicine in transit shall declare the veterinary medicine at the port of entry and exit.
|
|
| 20. |
Manufacturing permit
A person who intends to manufacture veterinary medicine shall make an application to the Council for a manufacturing permit.
|
| 21. |
Manufacturing of veterinary medicines
| (1) |
The Council shall, on receipt of an application made under regulation 20, before issuing a certificate of registration, cause the premises in which the manufacturing of the veterinary medicine is proposed to be conducted be inspected in order to ensure compliance to good manufacturing practice.
|
| (2) |
The inspector instructed under paragraph (1) shall submit the results of the inspection to the Council and the applicant in writing.
|
| (3) |
A manufacturer shall ensure that every stage in the manufacture of a veterinary medicine is carried out based on good manufacturing practices.
|
| (4) |
A manufacturer shall not manufacture a veterinary medicine unless the manufacturing plant has been inspected and licensed in accordance with this Regulation.
|
| (5) |
A person shall not use any premises, for the purposes of manufacturing. formulating, packaging, selling or storing veterinary medicine unless that person is in possession of a licence issued under these Regulations in respect of that premises.
|
| (6) |
Where an inspector has approved the compliance by the manufacturer, the Council shall on payment of the prescribed fee, issue a licence as set out in Form 1.3 set out in the a Third Schedule.
|
| (7) |
A person in executive authority of manufacturing company which manufactures veterinary medicines without a license commits an offence and shall be liable, on conviction, to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months or to both.
|
|
| 22. |
Powers of inspectors with regard manufacturing plants
| (1) |
An inspector appointed under these Regulations shall have power with regard to manufacturing plants to —
| (a) |
enter the premises and inspect the plant and the process of manufacture intended to be employed in the manufacturing of the veterinary medicine and shall make a report to the Council; or
|
| (b) |
order the immediate closure of a manufacturing plant where its continued operation appears to pose a serious threat to life and safety.
|
|
| (2) |
An inspector who has ordered the closure of a plant under paragraph (1)(b) shall within twenty-four hours of the closure notify the Council of the closure and shall provide reasons for the closure.
|
| (3) |
On receipt of the notification under paragraph (2), the Council shall direct the owner of the plant to take such measures as the Council may direct to ensure conformity with these Regulations.
|
| (4) |
A manufacturer who fails, within fourteen days of receipt of the directive made by the Council under paragraph (3), to take the measures indicated in the directive shall have its licence revoked.
|
| (5) |
The Council shall publish in at least one newspaper of wide national circulation the name of a manufacturing plant closed and whose licence has been revoked under this Regulation and shall place a notice of the closure at the entrance of such plant.
|
| (6) |
The protection of right to property under Article 40 of the Constitution shall be limited for purposes of maintenance of veterinary medicine standards for the protection and safety of the public.
|
|
| 23. |
Application for registration of a veterinary medicine
| (1) |
An application for registration of —
| (a) |
a veterinary medicine shall be in accordance with Form A set out in the Third Schedule; and
|
| (b) |
a veterinary medicine used as pesticides shall be Form B set out in the Third Schedule.
|
|
| (2) |
An applicant shall, in addition to the information required to be paragraph (1), furnish such further information and material as may be required by the Council for the proper evaluation of the veterinary medicine in respect of which the application is made.
|
| (3) |
The Council shall, on approval of an application made under this Regulation and on payment of the prescribed fees, register the veterinary medicine or veterinary pesticide and issue a certificate in Form D1 or D2, respectively set out in the Third Schedule.
|
| (4) |
A certificate of registration issued under these Regulations shall, unless suspended or revoked, be in force for a period of five years.
|
| (5) |
The Council shall not approve an application made under paragraph (1) unless satisfied that the applicant has attained the prescribed standards and satisfied all requirements for registration.
|
|
| 24. |
Renewal of registration of veterinary medicine
| (1) |
A person may make an application to the Council, for renewal of registration of a veterinary medicine in Form E set out in the Third Schedule.
|
| (2) |
The Council shall, before renewing registration under this Regulation conduct a pharmaco-vigilance study on the manufacturer.
|
| (3) |
An applicant shall on receipt of approval for renewal of registration from the Council, make payment of the fees as prescribed in the Fifth Schedule.
|
|
| 25. |
Clinical and non-clinical trials and toxicity testing
| (1) |
The Council may require that a clinical and non-clinical trial and toxicity testing be done before the registration of a veterinary medicine.
|
| (2) |
Despite paragraph (1) a veterinary pesticide shall not be registered unless an efficacy and an acute toxicological study is carried out by an accredited laboratory.
|
| (3) |
A person who or institution which intends to apply for accreditation under this Regulation shall, before making such application, obtain approval from the Council.
|
| (4) |
A person who or institution which desires to conduct clinical and non-clinical trials and toxicity testing shall apply to the Council for accreditation.
|
| (5) |
The cost of any clinical trial or toxicity testing shall be borne by the applicant.
|
|
| 26. |
Conditions for registration of veterinary medicines and pesticides
| (1) |
A veterinary medicine and pesticide shall not be registered unless a laboratory analysis has been carried out by an accredited laboratory.
|
| (2) |
A person who intends to register a veterinary medicine shall submit to the Council a certificate of analysis from an accredited laboratory together with the application for registration of the veterinary medicine
|
|
| 27. |
Veterinary Medicines Registration Committee
| (1) |
There is hereby established a standing committee of the Council to be known as the Veterinary Medicines Registration Committee.
|
| (2) |
The Committee shall be responsible for matters relating to registration of veterinary medicines under these Regulations and in particular shall be responsible for —
| (a) |
evaluating applications for veterinary medicines registration and shall make recommendations to the Council; and
|
| (b) |
issuing provisional approval pending the issuance of the veterinary medicines registration certificate by the Council.
|
|
| (3) |
A provisional certificate issued under paragraph (2)(b) shall be valid for a period of not more than three months.
|
| (4) |
The Council shall consider an application made under this Regulation, and if satisfied of the safety, efficacy and quality of the veterinary medicine or veterinary pesticide, shall register the veterinary medicine in accordance with Form C1 and C2 set out in the Third Schedule.
|
| (5) |
Upon registration of a veterinary medicine under paragraph (4), the applicant shall be issued with a certificate of registration in Form D1 or D2 set out in the Third Schedule.
|
| (6) |
The Council may, while considering a veterinary medicine for registration under paragraph (4), approve the details as supplied by the applicant or approve it with such modifications as it may consider appropriate in respect of the following particulars—
| (a) |
the name under which the veterinary medicine may be sold;
|
| (c) |
the statement of the representations to be made for the promotion of the veterinary medicine in respect of the —
| (i) |
claim to be made for the veterinary medicine; |
| (ii) |
route of administration; |
| (iv) |
contra-indications, side effects and precautions, if any; |
| (vi) |
withdrawal period for food producing animals; |
| (vii) |
proposed category; and |
|
|
|
| 28. |
Rejection of application for registration of a veterinary medicine
The Council shall, if it is not satisfied as to the safety, efficacy or quality of the veterinary medicine, reject the application for the registration of the veterinary medicine and inform the applicant, in writing, the reasons for rejection.
|
| 29. |
Registration of veterinary medicine researched outside Kenya
| (1) |
The Council may, before registering a new veterinary medicine for which the research work has been conducted in another country and its efficacy, safety, and quality established in that country, require an investigation on the pharmaceutical, pharmacological and other aspects of the veterinary medicine to be conducted and clinical trials to be carried out as necessary to establish its safety, efficacy, quality and where applicable the biological availability to be established under local conditions.
|
| (2) |
Despite paragraph (1), the Council may register a new veterinary medicine and require the investigations and clinical trials specified therein to be conducted after its registration.
|
|
| 30. |
Suspension and revocation of certificate of registration
| (1) |
The Council may suspend or revoke a certificate of registration issued under these Regulations for such period as the Council may determine.
|
| (2) |
The Council shall not revoke or cancel the certificate of registration unless —
| (a) |
where matters stated in the application on which the certificate of registration was granted were false or incomplete in any material particular;
|
| (b) |
where provision of the certificate of registration has to a material extent been contravened by the holder of the certificate;
|
| (c) |
where the premises on which, or on part of which, a veterinary medicine is manufactured, assembled or stored by or on behalf of the holder of the certificate of registration are unsuitable for the manufacturing, assembling or storage of veterinary medicines; or
|
| (d) |
where new information has been discovered by the Council which renders the veterinary medicines unsafe or dangerous.
|
|
| (3) |
Where a certificate has been revoked under paragraph (2)(d), the Council shall on delivery of the information to the manufacturer —
| (a) |
inspect the plant to ensure all the medicine or the relevant batch has been destroyed;
|
| (b) |
disseminate the information to the public through a newspaper advertisement in at least two daily newspapers of nationwide circulation; and
|
| (c) |
recall any medicine which has been distributed.
|
|
|
| 31. |
Period for renewal of registration and retention of veterinary medicine
| (1) |
The Council may grant the renewal of registration as provided under regulation 24 for a period not exceeding five years.
|
| (2) |
On the expiry of five years a veterinary medicine may be retained in the register upon an application made by the certificate holder which shall —
| (a) |
include a declaration that the requirements met by the veterinary medicine during registration has not changed; and
|
| (b) |
include payment of the requisite fees.
|
|
|
PART IV – CLASSIFICATION AND CATEGORIZATION OF VETERINARY MEDICINES
| 32. |
Classification of Veterinary Medicines
| (1) |
The Cabinet Secretary shall classify veterinary medicines in the classes set out in Part I of the Second Schedule.
|
| (2) |
The Cabinet Secretary may, on the advice of the Council, vary the classification of the various veterinary medicines depending on the therapeutic use and current information for the medicine.
|
|
| 33. |
Categorization of Veterinary Medicines
| (1) |
The Cabinet Secretary shall categorize veterinary medicines as set out in Part II of the Second Schedule.
|
| (2) |
The Cabinet Secretary shall be guided by the Council on the categorization of veterinary medicines and shall place the veterinary medicines in the following categories —
| (a) |
Category I relating to prescription only medicine—Veterinary Surgeon which shall comprise two sub-categories as follows —
| (i) |
Category IA relating to Controlled Veterinary Medicines includes products which contain narcotic or psychotropic substances or other substances very dangerous at small quantities; |
| (ii) |
Category IB relating to other prescription only medicine, comprising products intended for administration following a diagnosis or clinical assessment by a veterinary surgeon or dispensed by a veterinary surgeon or a person with equivalent qualification; |
|
| (b) |
Category II relating to prescription only medicine veterinary surgeon, pharmacist, and veterinary technologist who has served for at least five years in a veterinary pharmacy which contains —
| (i) |
veterinary medicines for use in food-producing animals; |
| (ii) |
veterinary medicines in respect of which special precautions shall be taken in order to avoid any unnecessary risk to the target species, the person administering the products to the animal and the environment; Provided that the requirement in this paragraph shall not apply if all the following criteria are met— |
|
| (c) |
Category III relating to authorised veterinary medicine general sales list which may be traded by veterinary surgeons and all categories of veterinary paraprofessionals and includes—
| (i) |
pest control veterinary medicines provided that the medicine are sold in the original package without repackaging and with an intact label; |
| (ii) |
endoparasiticide veterinary medicines considered safe for use in food animals; |
| (iii) |
endoparasiticide veterinary medicines for use in non-food animals; or |
| (iv) |
any other product the Directorate may prescribe. |
|
| (d) |
Category IV which includes alternative veterinary medicine general sales list.
|
|
|
| 34. |
Use of veterinary pesticides
| (1) |
A veterinary pesticide shall be—
| (a) |
sold only in the original sealed and labeled package as registered by the Council;
|
| (b) |
stored in accordance with the instructions on the label on the original container; and
|
| (c) |
used according to the instructions on the label.
|
|
| (2) |
The empty container of a veterinary pesticide shall be disposed in accordance with the instructions given on the label.
|
| (3) |
A person who removes or defaces a label on a veterinary pesticide or package commits an offence.
|
| (4) |
A person who sells, dispenses or otherwise gives out a veterinary pesticide in contravention of this Regulation commits an offence and shall be liable, on conviction, to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months or to both.
|
|
PART V – VETERINARY PHARMACY
| 35. |
Standards of Veterinary Pharmacy
| (1) |
A veterinary pharmacy shall satisfy the following minimum standards—
| (a) |
be located away from known fire hazards;
|
| (b) |
be a separate entity from other veterinary operations such as veterinary clinic, ambulatory and laboratory services;
|
| (c) |
be separated from non-complementary businesses;
|
| (d) |
have restricted access by personnel to Category I and II veterinary medicine; and
|
| (e) |
be vermin-proofed, including protection against insects and rodents.
|
|
| (2) |
A veterinary pharmacy shall—
| (a) |
demonstrate adequate security for the safety of veterinary medicine;
|
| (b) |
demonstrate appropriate storage conditions that shall maintain the temperature, lighting and humidity requirements as prescribed in the manufacturers' specifications of veterinary medicines;
|
| (c) |
have the floor and the wall of the building constructed from materials that are easy to clean, impervious and resistant to corrosion by chemicals;
|
| (d) |
have lockable safety cabinets to protect the controlled veterinary medicines;
|
| (e) |
provide the personnel with protective clothing for use only within the premises;
|
| (f) |
be of appropriate size and have sufficient space for carrying out of all necessary operations provided for the orderly receipt, warehousing and dispatch of the various classes of veterinary medicines and, in particular, a quarantine area for isolation when necessary, including isolation of faulty packs and recalled or expired veterinary products;
|
| (g) |
provide a disposal system, acceptable to the Council, for safe disposal of expired veterinary medicines;
|
| (h) |
be operated by competent staff as provided for in the permit;
|
| (i) |
display warning notices indicating hazardous areas;
|
| (j) |
label the various sections in the business;
|
| (k) |
store veterinary medicines only in the designated areas, which shall be labeled;
|
| (l) |
have emergency lighting, fire-fighting equipment and fully equipped first aid kit;
|
| (m) |
display emergency protocols within the veterinary pharmacy advising personnel on procedures to follow in case of emergencies;
|
| (n) |
display standard operating procedures;
|
| (o) |
retain records of the movement of all veterinary medicines for at least five years; and
|
| (p) |
provide separate sanitary facilities for each gender.
|
|
|
| 36. |
Qualifications to operate a veterinary pharmacy
A person who is a veterinary surgeon, or holds an equivalent qualification in matters of pertaining to veterinary medicines as determined by the Council, may apply to the Council for a permit to undertake a veterinary pharmacy.
|
| 37. |
Council to approve veterinary pharmacy businesses
| (1) |
The Council shall have power to approve any of the following types of veterinary pharmacy businesses—
| (a) |
manufacturing of veterinary medicines;
|
| (b) |
wholesaling of veterinary medicines; and
|
| (c) |
retailing of veterinary medicines.
|
|
| (2) |
Upon approval of the veterinary business specified under paragraph (1), the Council shall register the veterinary pharmacy premises in Form J set out in the Third Schedule.
|
|
| 38. |
Practicing permit
| (1) |
A person shall not practice the business of veterinary pharmacy unless he holds a valid practicing permit issued by the Council.
|
| (2) |
A person who intends to practice the business of veterinary pharmacy shall make an application in Forms H1, H2 and H2, respectively of the Third Schedule.
|
| (3) |
The Council shall issue a practicing permit if the premises in which the applicant intends to operate has been approved by the Council in accordance with these Regulations.
|
| (4) |
An application for a practicing permit shall be in —
| (a) |
Form I.1 for veterinary pharmacy practitioner;
|
| (b) |
Form I.2 for a wholesaler dealer permit; or
|
| (c) |
Form I.3 for manufacturers' permit.
|
|
| (5) |
An applicant under paragraph (1) shall, on approval of the application, by the Council, pay the fee prescribed in the Fifth Schedule.
|
| (6) |
The Council, upon confirming that the applicant has satisfied the conditions of a permit, and upon payment of the fee under paragraph (4) issue an applicant with a practicing permit as set out in Form A set out in the Third Schedule for veterinary pharmacy practices and Form B for wholesale pharmacy practice.
|
| (7) |
The practice permit shall be valid for a period of one year commencing on the 1st of January and ending on the 31st of December of each year.
|
| (8) |
The Council shall by the 31st March of every year publish, in the Kenya Gazette or in a newspaper of wide national circulation, the registered veterinary pharmacy businesses.
|
| (9) |
A person who conducts the business of veterinary pharmacy in premises not registered by the Council under these Regulations commits an offence, and shall be liable, on conviction, to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months or to both.
|
|
| 39. |
Practice permit to be displayed
| (1) |
A person who carries on the business of a veterinary pharmacy shall display his practice permit on a conspicuous place within the premises in which the business is being carried on.
|
| (2) |
A person who contravenes the provisions of this Regulation commits an offence and shall be liable, on conviction, to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months or to both.
|
|
| 40. |
Wholesale permit
A person who intends to trade in veterinary medicines, in bulk , shall make an application to the Council for a wholesale permit and support the application with the following—
| (a) |
a certified copy of the certificate of registration or incorporation of the business name or body corporate and the memorandum and articles of association;
|
| (b) |
provide evidence that the business is under the management of a supervisor who—
| (i) |
is a registered veterinary surgeon permitted to carry out the business of veterinary pharmacy; and |
| (ii) |
is a member of the Board of directors of the body corporate, and who is not acting in a similar capacity for any other body corporate. |
|
|
| 41. |
Retail permit
| (1) |
A person who intends to trade in veterinary medicines as a retailer shall make an application to the Council for a retail permit in Form H set out in the Third Schedule.
|
| (2) |
This regulation shall be applicable to a manufacturer and a wholesaler of veterinary medicines who desires to carry out retail trade.
|
|
PART VI – VETERINARY PHARMACY AND COMPLEMENTARY BUSINESSES
| 42. |
Complementary businesses
| (1) |
A veterinary pharmacy may be conducted alongside complementary businesses including the sale of human medicines, horticultural chemicals, agro-forestry chemicals and fertilizer, animal foodstuffs, seeds and agricultural equipment.
|
| (2) |
The building or premises of a veterinary pharmacy stocking or trading in complementary businesses referred to in paragraph (1), shall be built or adapted in such manner as to provide a separate and distinct partition for the veterinary products from any other products.
|
| (3) |
In addition to the provisions of paragraph (2), the premises shall be built or adapted in such manner as to provide separate and distinct partitions for veterinary medicines and veterinary pesticides such that no mixing of the two classes of products is permissible.
|
|
| 43. |
Stock, supply and distribution of veterinary medicine
| (1) |
A veterinary medicine shall be supplied or distributed only through a wholesale or retail business registered as a veterinary pharmacy business.
|
| (2) |
A holder of wholesale or veterinary pharmacy permit issued by the Council may sell veterinary medicines in bulk to registered retail dealers.
|
| (3) |
A holder of a retail dealer permit, issued by the Council, may sell veterinary medicines to the public.
|
| (4) |
A person shall not supply authorised human medicinal product for administration to an animal other than a product supplied by a veterinary surgeon or in accordance with a written prescription from a veterinary surgeon.
|
| (5) |
A veterinary pharmacy practitioner who dispenses a veterinary medicine belonging to Category I or II to any member of the public without a prescription, commits an offence and shall be liable, on conviction, to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months or to both.
|
|
PART VII – MARKET AUTHORIZATION
| 44. |
Authorization to import or export veterinary medicine
| (1) |
A person who intends to import or export a veterinary medicine shall apply to the Council for a permit in Form K set out in the Third Schedule.
|
| (2) |
The Council shall in determining the application under paragraph (1) consider whether the applicant has met the conditions for importation and exportation in these Regulations, and if so, shall on payment of the prescribed fee, issue the applicant with an import permit in Form L set out in the Third Schedule.
|
| (3) |
A holder of a market authorization may apply to the Council for a wholesale permit to authorize him to import the veterinary medicine specified in the authorization.
|
| (4) |
An authorised wholesale dealer may import a veterinary medicine if —
| (a) |
the authorization covers the veterinary product; and
|
| (b) |
the wholesale dealer has acquired the written consent of the holder of the market authorisation in writing before importation.
|
|
| (5) |
A veterinary surgeon may, with approval of the Council, import an orphan veterinary medicine.
|
|
PART VIII – VETERINARY MEDICINE INSPECTORS
| 45. |
Appointment of veterinary medicine inspectors
The Council shall appoint duly qualified persons on such terms and conditions of service as it may deem appropriate, to serve as veterinary medicine inspectors.
|
| 46. |
Identification of veterinary medicines inspector
| (1) |
The Council shall publish in the Kenya Gazette every inspector appointed under regulation 45 and issue each inspector with an official identity card which shall have a passport size photo of the inspector, duly stamped and signed by the Chief Executive Officer or his authorized agent.
|
| (2) |
Every inspector shall, in conducting inspections under these Regulations, identify himself using the identity card issued under paragraph (1).
|
| (3) |
A person who ceases to be an inspector shall surrender his identify card to the Chief Executive Officer or his authorized agent.
|
|
| 47. |
Powers of veterinary medicines inspectors
| (1) |
An inspector shall have the power, at all reasonable times, to —
| (a) |
enter upon the premises of any manufacturer, distributor or veterinary pharmacy and to inspect any books, papers, records or writings, veterinary medicines, whether patent or otherwise, or any article stored or offered for sale or used in the business; or
|
| (b) |
enter any premises in which he has reasonable cause to suspect that a breach of the law has been or is being committed, and to make such examination and inquiry and to do such other activities, including impounding and seizing suspect veterinary products, closing suspect premises and the taking of samples, as may be necessary for the purpose of ascertaining whether the provisions of these Regulations are being complied with.
|
|
| (2) |
| (a) |
for the purpose of inspecting a veterinary medicine use Form F set out in the Third Schedule; and
|
| (b) |
for any impounding of suspect veterinary medicines, use Form G set out in the Third Schedule.
|
|
| (3) |
An inspector shall observe confidentiality in the findings of his inspection.
|
| (4) |
An inspector shall be liable for any act of negligence he may commit in the performance of his duties.
|
|
PART IX – FINANCIAL PROVISIONS
| 48. |
Funds of the Directorate
| (1) |
The funds of the Directorate shall comprise of —
| (a) |
such monies as may be appropriated by Parliament for the purposes of the Directorate;
|
| (b) |
such monies as may accrue to or vest in the Directorate in the course of the exercise of its functions under these Regulations; and
|
| (c) |
monies from any other source provided for, donated or lent to the Directorate.
|
|
|
| 49. |
Financial year
The financial year of the Directorate shall be the period of twelve months from first July to thirtieth of June.
|
| 50. |
Annual estimates
| (1) |
Three months before the commencement of each financial year, the Council shall cause to be prepared estimates of revenue and expenditure of the Directorate for that year.
|
| (2) |
The annual estimates shall make provision for all the estimated expenditure of the Directorate for the financial year concerned, and in particular shall provide for—
| (a) |
the payment of salaries, allowances and other charges in respect of the staff of the Directorate;
|
| (b) |
the payment of pensions, gratuities and other charges in respect of retirement benefits which are payable out of the funds of the Directorate;
|
| (c) |
the proper maintenance of buildings and grounds of the Directorate;
|
| (d) |
the acquisition, maintenance, repair and replacement of the equipment and other movable property of the Directorate; and
|
| (e) |
the creation of such reserve funds to meet future or contingent liabilities in respect of retirement benefits, insurance or replacement of buildings or equipment or in respect of such other matter as the Directorate may find appropriate.
|
|
| (3) |
The annual estimates shall be approved by the Council before the commencement of the financial year to which they relate and shall be submitted to the Cabinet Secretary for approval.
|
| (4) |
The Directorate shall not increase any sum provided in the estimates without the consent of the Cabinet Secretary.
|
|
| 51. |
Investment of funds
The Council may invest any of the funds of the Directorate in a manner approved by the Treasury for the investment of trust funds.
|
| 52. |
Accounts and Audit
| (1) |
The Council shall cause to be kept all proper books and records of accounts of the income, expenditure, assets and liabilities of the Directorate.
|
| (2) |
The accounts of the Directorate shall be audited in accordance with the Public Finance Management Act, 2012 (No. 18 of 2012).
|
|
PART X – OFFENCES
| 53. |
Offences
A person commits an offence if that person —
| (a) |
imports, exports, manufactures, stores, distributes, sells or otherwise handles a veterinary medicine that has not been registered under these Regulations;
|
| (b) |
imports a veterinary medicine without a permit issued under these Regulations;
|
| (c) |
manufactures, stores, distributes or sells a veterinary medicine in premises which have not been registered under these Regulations;
|
| (d) |
presents for sale or distribution, expired, adulterated, Counterfeit or unlabeled veterinary medicines;
|
| (e) |
sells a veterinary medicine in any form other than the original sealed and labeled package, as registered with the Directorate;
|
| (f) |
sells or distributes a veterinary medicine in any other area other than a registered premises;
|
| (g) |
uses a veterinary medicine for purposes other than that for which it was registered, prescribed or dispensed;
|
| (h) |
supplies human medicinal product for administration to an animal in contravention of these Regulations;
|
| (i) |
is in possession of a Category I or Category II veterinary medicine other than in accordance with these Regulations;
|
| (j) |
advertises a veterinary medicine in contravention of these Regulations;
|
| (k) |
buys a veterinary medicine for resale in bulk while not being in possession of a wholesale dealer permit issued under these Regulations;
|
| (l) |
sells a veterinary medicine to the public while not being in possession of a retail dealer's permit issued in accordance with these Regulations;
|
| (m) |
undertakes research on imported and unregistered veterinary medicine without authorization from the Council;
|
| (n) |
obstructs or fails to assist the Council or veterinary medicine inspector in the performance of their lawful duties under these Regulations;
|
| (o) |
provides the Council or inspector with false or misleading information;
|
| (p) |
operates a veterinary pharmacy after it has been closed by an inspector; or
|
| (q) |
contravenes any provision of these Regulations.
|
|
| 54. |
Officers of corporations
If an institution commits an offence under these Regulations, any officer, director or agent of the corporation who directed, authorized, assented to, acquiesced in, or participated in the commission of the offence shall be a party to and shall be considered to have committed the offence and shall be liable on conviction to the punishment provided for the offence, whether or not the corporation has been prosecuted or convicted.
|
| 55. |
Passing confidential information without authorization
A member of the Council or staff of the Directorate, who —
| (a) |
being in possession of confidential information, however obtained without authorization of the Council —
| (ii) |
attempts, offers or threatens to divulge it, |
|
other than in accordance with these Regulations or other written law; or
| (b) |
willfully obtains or seeks to obtain confidential information to which he is not entitled, commits an offence.
|
|
| 56. |
Penalties
A person who contravenes these Regulations where no Penalties. penalty has been prescribed, shall on conviction be liable to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months, or to both.
|
PART XI – GENERAL PROVISIONS
| 57. |
Advertising veterinary standards for medicines
| (1) |
A person who intends to advertise a veterinary medicine, shall make an application to the Council for authority to advertise.
|
| (2) |
The Council may, on payment of the requisite fee and subject to such conditions as the Council or these Regulations may impose, grant the authorization applied for under paragraph (1).
|
| (3) |
An advertisement for a veterinary medicine shall not be misleading or contain any medicinal claim that is not in the summary of the product characteristics registered with the Council.
|
| (4) |
A veterinary medicine listed under Category I and II shall not be advertised unless —
| (a) |
in the case of a veterinary medicine listed under Category I, the advertisement is aimed at veterinary surgeons; and
|
| (b) |
in the case of a veterinary medicine listed under Category II, the advertisement is aimed at veterinary surgeons or suitably qualified persons as recognized by the Council.
|
|
| (5) |
A veterinary medicine listed in Category III and IV may be advertised to general members of the public.
|
|
| 58. |
Exemptions to Regulations
These Regulations do not apply to —
| (a) |
inactivated autogenous vaccine that is manufactured, on the instructions of a veterinary surgeon, from pathogens or antigens obtained from an animal and used for the treatment of that animal or —
| (i) |
other animals on the same site; |
| (ii) |
animals intended to be sent to those premises; or |
| (iii) |
animals on a site that receives animals from those premises; or |
|
| (b) |
animal foodstuff supplements with no therapeutic claim.
|
|
| 59. |
Discretion of Council
The Council shall have the discretion to —
| (a) |
approve the importation and use of any veterinary medicine for handling emergency situations;
|
| (b) |
cancel the registration of any veterinary medicine that is considered to be harmful to animal and human health and the environment; or
|
| (c) |
restrict the use of any specified veterinary medicine.
|
|
| 60. |
Copy of certificate or authorization
| (1) |
A person who has been issued with a certificate or authorization under these Regulations may, if the certificate or authorization is defaced, damaged or lost, on application to the Council and on payment of the prescribed fee, be issued with a copy of the certificate.
|
| (2) |
The copy of a certificate or authorization issued under paragraph (1) shall —
| (i) |
be issued only where the document is in force at the time the application is made; |
| (ii) |
be valid for the same period as the original document; and |
| (iii) |
bear the words "DUPLICATE COPY". |
|
|
| 61. |
Fees
Any fees payable under this Act is as set in the Fifth Schedule.
|
| 62. |
Supercession
These Regulations shall supersede any other Regulations on matters concerning veterinary medicines.
|
| 63. |
Transition and validity of licences and permits
Upon the commencement of these Regulations any certificate in force shall be deemed to have been issued under these Regulations and shall remain in force until its expiry.
|
FIRST SCHEDULE [r. 8(6).]
CONDUCT OF THE AFFAIRS OF THE COUNCIL
| 1. |
Meetings of the Council.
The Council shall meet at least four times in a year and not more than eight times a year, except in case of an emergency, for the transaction of its business, and such meetings shall be held at such places and times and on such days as the Council may determine.
|
| 2. |
Chairperson and vice-charperson
| (1) |
The Council shall at its first meeting elect its vice-chairperson from among the three members appointed under regulation 8(3)(f).
|
| (2) |
The Chairperson shall preside at all meetings of the Council at which he is present, and in case of his absence the vice-chairperson shall preside, but in the absence of both the chairperson and vice-chairperson, members present and constituting a quorum shall elect one among their number to be the chairperson for purposes of the meeting.
|
|
| 3. |
Quorum
The quorum of a meeting of the Council shall be five, at least three of whom shall be veterinary surgeons.
|
| 4. |
Decisions of Council
The decisions of the Council shall be by a simple majority of the votes of the members present, but in the case of an equality of votes, the Chairperson or person presiding shall have a casting vote.
|
| 5. |
Minutes
| (1) |
The Minutes of each meeting shall be kept in the minute book, after they have been confirmed by the Council and signed by the Chairperson at a subsequent meeting of the Council.
|
| (2) |
The deliberations and minutes of meetings of the Council shall be confidential.
|
|
| 6. |
Vacancy not to invalidate proceedings
No proceedings of the Council shall be invalid by reason only of a vacancy among the members thereof.
|
| 7. |
Council to determine own procedure
Subject to this Schedule, the Council may determine its own procedure.
|
| 8. |
Invitation of other persons
The Council may invite any person to participate in its deliberations but a person who has been invited shall have no right to vote.
|
SECOND SCHEDULE [rr. 32(1) & 33(1).]
CONDUCT OF THE AFFAIRS OF THE COUNCIL
PART I – CLASSES OF VETERINARY MEDICINES
| 1. |
List of veterinary pharmaceuticals.
| (iii) |
Analgesics and anti-inflammatories |
| (iv) |
Drugs acting on the nervous system |
| (vi) |
Other systemic therapeutic agents |
| (vii) |
Local therapeutic agents |
|
| 2. |
| (v) |
Probiotics and enzymes. |
|
| 4. |
| (ii) |
Veterinary medicine administration devices. |
| (iii) |
Any other material and equipment of veterinary relevance. |
|
| 5. |
List of alternative medicines.
| (iii) |
Performance enhancers. |
|
| 6. |
| (v) |
Any other pesticides of veterinary relevance |
| (vi) |
And other ecto-parasiticides |
| (vii) |
substances used for euthanasia |
|
PART II – CATEGORIES OF VETERINARY MEDICINES
(a) Prescription Only Medicine—Veterinary surgeon (abbreviated to POM-V);
Category I A POM-V Controlled Veterinary Medicine
| 11. |
Morphine and its derivatives.
|
| 13. |
Methadone (amidone); its salts.
|
| 16. |
Phenoperidine; its salts.
|
| 19. |
Any other veterinary medicine relevant in the category.
|
Category IB — Other POM - V
CATEGORY II —PRESCRIPTION ONLY MEDICINE (POM-VT)
Prescription Only Medicine—Veterinary surgeon
CATEGORY III — Authorized Veterinary Medicines —General Sales List (AVM-GSL)
CATEGORY IV- Alternative Veterinary Medicine — General Sales List (AItVM-GSL)
THIRD SCHEDULE [rr. 23(1) & 38(6).]
FORM A
APPLICATION FOR REGISTRATION OF A VETERINARY MEDICINE
(to be submitted as one original hard-copy and one electronic copy in MS-Word)
The Registrar
Veterinary Medicines Directorate
KABETE









APPLICATION FOR THE REGISTRATION OF VETERINARY PESTICIDES
[to be submitted as one original hard-copy and one electronic copy (in MS-Word)]
The Director
Veterinary Medicines Directorate
KABETE
Information for Applicants
| 1. |
The application form must be completed by a duly authorized person.
|
| 2. |
Every application must be accompanied by 3 copies of the draft label as per VMD requirements.
|
| 3. |
The applicant may be required to submit:-
| (a) |
a sample of the veterinary pesticide;
|
| (b) |
a sample of the technical grade of its active ingredient;
|
| (c) |
a sample of the laboratory standard of its active ingredient;
|
| (d) |
any other sample as may be required by the Council.
|
|
| 4. |
The application must be accompanied by a technical dossier as per VMD data Requirements.
|
| 5. |
An applicant who is not a resident in Kenya shall appoint as an agent a person who is permanently resident in Kenya and duly recognized by the Veterinary Medicines Directorate.











|
SUMMARY OF THE DATA SUBMITTED TO THE VMD FOR REGISTRATION OF A VETERINARY PESTICIDES
FOURTH SCHEDULE [r. 17.]
CODE OF CONDUCT FOR MEMBERS OF AND EMPLOYEES OF THE DIRECTORATE
| 1. |
Impartiality and independence of members
| (1) |
Every member and employee of the Directorate shall impartially and independently perform the functions of a member in good faith and without fear, favour or prejudice, and without influence from—
| (a) |
the National or County Government;
|
| (d) |
any candidate participating in an election; or
|
| (e) |
any other person or authority.
|
|
|
| 2. |
Independence from political or public office
| (1) |
A member or employee of the Directorate shall not, during tenure of office, be eligible for—
| (a) |
appointment or nomination to a political office; or
|
| (b) |
appointment to another public office.
|
|
| (2) |
A member of the Directorate may not—
| (a) |
by their membership, association, statement, conduct or in any other manner place in jeopardy the perceived independence of the member, or in any other manner harm the credibility, impartiality, independence or integrity of the Directorate;
|
| (b) |
make private use of or profit from any confidential information gained as a result of being a member of the Directorate; or
|
| (c) |
divulge any information to any third party, save in the of official duty.
|
|
|
| 3. |
Disclosure of conflicting interests
| (1) |
If a member or an employee is directly or indirectly interested in any contract, proposed contract or other matter before the Council and is present at any meeting of the Council at which the contract, proposed contract or other matter is the subject of consideration, the member or employee shall, at the meeting and as soon as practicable after the commencement thereof, disclose the fact and shall not take part in the consideration or discussion of, or vote on, any questions with respect to the contract or other matter or be counted in the quorum of the meeting during consideration of the matter.
|
| (2) |
A member or employee whose personal interest conflicts with their official duties shall—
| (a) |
declare the personal interests to their supervisor or other appropriate person or body in writing and comply with any directions given to avoid the conflict; and
|
| (b) |
refrain from participating in any deliberations with respect to the matter.
|
|
| (3) |
No member of staff of the Directorate shall transact business with the Directorate directly or indirectly.
|
|
| 4. |
Professionalism
A member or employee of the Directorate shall—
| (a) |
perform their duties in a manner that promotes and maintains public confidence in the Directorate;
|
| (b) |
treat the public and colleagues with courtesy and respect;
|
| (c) |
discharge all their duties in a professional, timely and efficient manner and in line with the rule of law; and
|
| (d) |
respect the rights and freedom of all persons that he may interact with.
|
|
| 5. |
Improper enrichment
A member or employee of the Directorate shall not—
| (a) |
use their office or organization to improperly enrich themselves or others;
|
| (b) |
accept or request gifts or personal favours from any person who may have a commercial interest with the Directorate or any other interest that may be affected by the normal business of the Service; or
|
| (c) |
use information that is acquired during the course of their duties or connected to their duties for their benefit or for the benefit of others
|
|
| 6. |
Integrity in private affairs
A member or employee shall conduct their private affairs in manner that maintains public confidence in the integrity of their office and the Directorate as a whole and shall—
| (b) |
not neglect their financial obligations;
|
| (c) |
submit an annual declaration of their income, assets and liabilities to the Commission responsible for such declarations from public officers;
|
| (d) |
not engage in political activity that may compromise or be seen to compromise the neutrality of their office, or the Directorate; and
|
| (e) |
not preside over or play a central role in the organization of a fundraising activity.
|
|
| 7. |
Sexual harassment
| (1) |
A member or employee shall not sexually harass a member of the public or colleague.
|
| (2) |
Sexual harassment includes —
| (a) |
making a request or exerting pressure for sexual activity or favours;
|
| (b) |
making intentional or careless physical contact that is sexual in nature; or
|
| (c) |
making gestures, jokes or comments, including innuendoes regarding another person's sexuality.
|
|
|
| 8. |
Nepotism
A member or employee shall not practice favouritism on the grounds of tribe, race, kin, culture, sex or acquaintance or otherwise in performance of their duties.
|
| 9. |
Application of the Public Officers Ethics Act
This Code is in addition to the provisions of the Public Officers Ethics Act and where there is a conflict between the Code and these Regulations, the provisions of the Act shall prevail.
|
| 10. |
Breach of code
Any breach of the Code by a member or officer of the Service shall be treated as gross misconduct.

|
THE VETERINARY SURGEONS AND VETERINARY PARA-PROFESSIONALS (VETERINARY MEDICINES DIRECTORATE) REGULATIONS
PART I – PRELIMINARY
| 1. |
Citation
These Regulations may be cited as the Veterinary Surgeons and Veterinary Para-Professionals (Veterinary Medicines Directorate) Regulations.
|
| 2. |
Interpretation
In these Regulations, unless the context otherwise requires—
"accredited laboratory" means a laboratory recognized as an accredited laboratory by the Directorate;
"advertisement" means any written or visual notice, circular, label, or wrapper, or other descriptive matter, verbal statement or reference appearing in any newspaper, television, film or mass media or brought to the attention of the public in any other form, which is intended to promote the sale of a veterinary medicine;
"alternative medicine" means the unrefined plant, animal and mineral substances commonly used in traditional animal treatments;
"Cabinet Secretary" means the Cabinet Secretary responsible for matters relating to veterinary service;
"Chief Executive Officer" means the Chief Executive Officer of the Directorate appointed under regulation 13;
"Committee" means the Veterinary Medicines Registration Committee established under regulation 27;
"controlled veterinary medicine" means a veterinary medicine specified in the Fourth Schedule as a Category I or Category II of veterinary medicine;
"Conventional medicines" means the regular and standardized veterinary medicines;
"Council" means the Council of the Directorate comprised under regulation 8(1);
"crude drug" means an unrefined medicine of biological origin;
"Directorate" means the Veterinary Medicines Directorate established under regulation 5 and as envisaged under section 39(2)(a) of the Act;
"dispense" means the sale or supply of a veterinary medicine by a veterinary surgeon or other person authorised in accordance with these Regulations;
"inspector" means a person appointed as an inspector under regulation 45;
"manufacture" means any stage in the manufacturing of a veterinary medicine until the finished product is ready for sale in its final form as specified in the marketing authorization, and includes repackaging, repacking or labeling of a veterinary medicine in an authorized facility but does not include the breaking open of the package of a veterinary medicine by retailers;
"market authorization" means registration of a veterinary medicine by the Council and the issuance of a registration certificate under regulation 23;
"orphan veterinary medicine" means a veterinary medicine that is not economical to trade in but is required for specific medical use;
"pharmaco-vigilance" means the routine inspections and surveys carried out in the veterinary medicines market to safeguard general animal and human health and trade;
"quality assurance standards" means the good manufacturing practice, good laboratory practice, good veterinary practice or any other standard developed by an international standardization body, the East African Community Standards Committee or the Kenya Bureau of Standards which the Cabinet Secretary, on the advice of the Council, shall recognize through the Gazette as a quality assurance standard for the purposes of these Regulations;
"register" means a register maintained by the Council containing the details of—
| (a) |
premises which have been issued with a permit in accordance with these Regulations; or
|
| (b) |
veterinary medicines registered in the country;
|
"retailer" means a veterinary pharmacy registered by the Council for the sale of veterinary medicines to the end users;
"specialized committees" means persons who are not Council members;
"veterinary medicines inspector" means an inspector appointed by the Council pursuant to regulation 45;
"veterinary pesticide" means a veterinary medicine used as a pest control product on animals or the animals' environment;
"veterinary pharmaceutical" means a chemical substance formulated or compounded as a single active ingredient or in any combination of the chemical substances, for veterinary curative use;
"veterinary pharmacy" means a business authorized by the Council to stock, dispense or distribute veterinary medicines for sale;
"veterinary pharmacy assistant" means a veterinary para-professional trading in veterinary medicines listed under Category III of the Fourth Schedule;
"veterinary pharmacy practice" means the business of veterinary pharmacy carried out by veterinary surgeons, veterinary para-professionals or any other person permitted by the Council to carry out veterinary pharmacy practice;
"veterinary product" means veterinary medicines and veterinary pesticides;
"wholesale" means a manufacturer or veterinary pharmacy approved by the Council to trade in bulk in the supply of veterinary medicines to wholesalers or retailers in the original outer pack-sizes.
|
| 3. |
Objectives and purpose of the Regulations
The object and purpose of these Regulations is to—
| (a) |
regulate the manufacture, importation, exportation, registration, distribution, prescription and dispensing of veterinary medicines and the practice of veterinary pharmacy in Kenya; and
|
| (b) |
advise the Kenya Veterinary Board in relation to all aspects listed under paragraph (a).
|
|
| 4. |
Application
Subject to regulation 58, these Regulations shall apply to all conventional and alternate veterinary medicine.
|
PART II – THE VETERINARY MEDICINES DIRECTORATE
| 5. |
Establishment of the Directorate
| (1) |
There is hereby established a Directorate to be known as the Veterinary Medicines Directorate whose management shall vest on a Council appointed under regulation 8.
|
| (2) |
The Directorate shall be a body corporate with perpetual succession and a common seal and shall in its corporate name be capable of—
| (b) |
taking, purchasing, or otherwise acquiring, holding, charging or disposing of movable and immovable property;
|
| (c) |
borrowing and lending money; and
|
| (d) |
doing all such other things or acts as may lawfully be done by a body corporate.
|
|
|
| 6. |
Functions of the Directorate
The functions of the Directorate are to—
| (a) |
formulate and enforce quality assurance standards in the in the manufacture, distribution and use of veterinary medicines to safeguard human and animal health and the environment;
|
| (b) |
in consultation with the Director of Veterinary Services, regulate the use of veterinary medicine for the treatment of animals under the Animal Diseases Act (Cap. 364);
|
| (c) |
consider applications for approval for market authorization of veterinary medicines;
|
| (d) |
set quality assurance standards for training in the management of veterinary medicines as directed by the Kenya Veterinary Board;
|
| (e) |
collaborate with the Kenya Veterinary Board in regulating training in the management of veterinary medicines;
|
| (f) |
inspect and approve premises in which the manufacture, sale or supply of veterinary medicine is conducted;
|
| (g) |
appoint and gazette veterinary medicine inspectors;
|
| (h) |
establish the standard operating procedures for veterinary medicine inspectors;
|
| (i) |
regulate veterinary pharmacy practices;
|
| (j) |
categorize veterinary medicines and the qualification of persons authorized to trade in each category and review the categories every five years;
|
| (k) |
regulate clinical and non-clinical trials of veterinary medicines by individuals and institutions to be involved in the trials;
|
| (l) |
regulate the manufacture, importation, exportation, handling, advertisement, labeling, sale and disposal of veterinary medicines;
|
| (m) |
register all veterinary medicines manufactured or imported for use in the country or exported from the country;
|
| (n) |
monitor the market for and take measures necessary for the elimination of trade in illegal and counterfeit veterinary medicines;
|
| (o) |
establish systems of pharmaco-vigilance and conduct pharmaco-vigilance of veterinary medicines through regular inspections and surveys;
|
| (p) |
enforce good manufacturing practice for veterinary medicines as approved by the Council;
|
| (q) |
develop, apply and from time to time review guidelines to be used in the inspection and ensuring compliance with good manufacturing practice;
|
| (r) |
ensure that the promotion and marketing of veterinary medicine is in accordance with the approved product information;
|
| (s) |
publish, on an annual basis, a notice in the Kenya Gazette inviting the public to note and inspect the register of veterinary medicines and the list of approved veterinary pharmacy practices within such period and at such place as may be specified in the notice;
|
| (t) |
consider, grant, issue or revoke authorizations and certificates in accordance with these Regulations;
|
| (u) |
collaborate with other regulatory agencies including the Public Health (Standards) Board in section 27 of the Food, Drugs and Chemical Substances Act (Cap. 254), the Pest Control Products Board established under section 5 of the Pest Control Products Act (Cap. 346) and the Central Board of Health established under section 3 of the Public Health Act (Cap. 242) to carry out its mandate; and
|
| (v) |
undertake any other thing necessary for the effective carrying out of its mandate under this or any other Act.
|
|
| 7. |
Powers of the Council
The Council shall have all the power necessary or expedient for the effective discharge of its functions under these Regulations and any other law, and in particular the power to—
| (a) |
control, supervise and manage the assets and liabilities of the Directorate;
|
| (b) |
determine the provision to be made for capital and recurrent expenditure and for the reserves of the Directorate;
|
| (c) |
seek and receive any grants or donations and make legitimate disbursements from such grants and donations for its purposes;
|
| (d) |
levy fees and charges for its services as provided in these Regulations;
|
| (e) |
enter into association with other bodies within or outside Kenya which the Council may consider desirable or appropriate;
|
| (f) |
invest funds of the Directorate not immediately required in securities in which trustees are empowered to invest under the Trustee Act (Cap. 167), and in other securities which may be approved for the purpose, by the Cabinet Secretary for the time being responsible for Finance;
|
| (g) |
establish and support investment and trust funds for the benefit of employees or ex-employees of the Directorate or dependants of such persons, to grant pension, benefits and allowances and to make such payments towards insurance as required under the relevant laws;
|
| (h) |
open and operate such accounts as are necessary for the funds of the Directorate, with a bank or financial institution licensed to conduct banking business under the Banking Act (Cap. 488);
|
| (i) |
recruit, support, discipline or dismiss the staff and inspectors of the Directorate;
|
| (j) |
in consultation with Salaries Remuneration Commission, determine the terms and conditions of employment of the staff and inspectors of the Directorate;
|
| (k) |
establish branch offices of the Directorate, to the extent that is practicable, to ensure accessibility of its services by all Kenyans;
|
| (l) |
superintend, regulate and assist branch offices, auxiliaries, committees and other forms of organizations established to advance the interest of the Directorate;
|
| (m) |
perform all things necessary or incidental to attain the objectives of the these Regulations or any other written law.
|
|
| 8. |
Composition and appointment of the Council
| (1) |
The Council shall be appointed by the Cabinet Secretary and shall consist of—
| (a) |
the Director of Veterinary Services who shall be the Chairperson;
|
| (b) |
the Registrar of the Pharmacy and Poisons Board;
|
| (c) |
the Chief Executive Officer of the Kenya Veterinary Board;
|
| (d) |
the Principal Secretary for the time being responsible for Finance;
|
| (e) |
the Principal Secretary for the time being responsible for animal health matters;
|
| (f) |
three veterinary surgeons nominated by the Kenya Veterinary Board;
|
| (g) |
one veterinary technologist from the veterinary pharmaceutical industry nominated by the Kenya Veterinary Board; and
|
| (h) |
the Chief Executive Officer of the Directorate who shall be the Secretary to the Council and shall be an ex officio member.
|
|
| (2) |
A person appointed under paragraph (1)(f) shall be nominated by the Kenya Veterinary Board from a list of five names drawn from the veterinary pharmaceutical industry including a trainer in veterinary pharmacology submitted by a registered professional association representing the interests of veterinary surgeons countrywide.
|
| (3) |
A person appointed under paragraph (1)(g) shall be nominated by the Kenya Veterinary Board from a list of three names from the veterinary pharmaceutical industry submitted by the registered association representing the interests of veterinary paraprofessionals.
|
| (4) |
The nominating bodies referred to under paragraphs (2) and (3) shall observe Constitutional principles relating to gender, youth, persons with disability and minorities in identifying the persons whose names shall be submitted for nomination by the Kenya Veterinary Board for appointment to the Council.
|
| (5) |
The Cabinet Secretary shall appoint the members of the Council by notice in the Kenya Gazette.
|
| (6) |
The Council shall conduct its affairs in the manner set out in the First Schedule.
|
| (7) |
The members of the Council shall hold office for a term of three years and shall be eligible for re-appointment for one further term.
|
| (8) |
The members appointed under paragraph (1)(b), (d) and (e) may, in writing, designate representatives to attend the meetings of the Council on their behalf.
|
| (9) |
The members of the Council in paragraphs (f), (g) and (h) may—
| (a) |
at any time resign from office by notice in writing to the Cabinet Secretary;
|
| (b) |
be removed from office by the Cabinet Secretary, on the advice of the Council if the member —
| (i) |
is declared bankrupt; |
| (ii) |
is absent from three consecutive meetings of the Council, without the permission of the Chairperson; |
| (iii) |
is convicted of a criminal offence and, sentenced to a term of imprisonment of more than six months; |
| (iv) |
is unable or unfit, due to physical or mental illness, to perform the functions of his office; or |
| (v) |
has failed to comply with the provisions of Chapter Six of the Constitution. |
|
|
| (10) |
The Cabinet Secretary shall, on the recommendation of the Council, appoint a relevant person to serve in the place of any member of the Council in the case of death, resignation, absence from Kenya for six consecutive months without authorization or more or removal from office under paragraph (9)(b) and the person appointed under this subregulation shall serve until the end of the term of the Council.
|
| (11) |
Where a vacancy arises under paragraph (10), the Cabinet Secretary shall undertake a fresh recruitment process to fill the vacancy.
|
|
| 9. |
Co-option of specialized persons
Pursuant to section 10 of the Act, the Council may for the effective discharge of its functions co-opt into the committees such persons with technical expertise or knowledge for the better carrying out of the functions of the Directorate.
|
| 10. |
Meetings of the Council
The conduct and regulation of the business and affairs of the Council shall be as provided in the First Schedule but subject thereto, the Council shall regulate its own procedure.
|
| 11. |
Conflict of interest
| (1) |
If any person is present at a meeting of the Council or any committee at which any matter is the subject of consideration and in which matter that person or that person's spouse or relative is directly or indirectly interested in a private capacity, that person shall as soon as is practicable after the commencement of the meeting declare such interest and shall not, unless the Council or committee otherwise directs, take part in any consideration or discussion of, or vote on any question connected to such matter.
|
| (2) |
The disclosure of interest shall be recorded in the minutes of the meeting at which it is made.
|
| (3) |
A member or employee of the Council shall not carry out an business or trade with the Council.
|
| (4) |
A member or staff of the Council who contravenes this Regulation commits an offence and is liable, on conviction, to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months or to both.
|
|
| 12. |
Common seal and logo
| (1) |
The Directorate shall have a common seal which shall be kept by the Chief Executive Officer.
|
| (2) |
The affixing of the seal shall be authenticated by signature of the chairperson and the Chief Executive Officer and in his absence, the signature of any other member authorized by resolution of the Council.
|
|
| 13. |
Chief Executive Officer
| (1) |
There shall be a Chief Executive Officer who shall be appointed by the Council through a competitive process and whose terms and conditions of service shall be determined by the Council in the instrument of appointment.
|
| (2) |
A person shall be qualified for appointment as a Chief Executive officer, if that person—
| (b) |
holds a degree in veterinary medicine from a university recognized by the Kenya Veterinary Board;
|
| (c) |
has at least ten years professional experience in veterinary medicine regulation, of which at least five years are at senior management level; and
|
| (d) |
satisfies the requirements of Chapter Six of the Constitution.
|
|
| (3) |
The Chief Executive Officer shall be an ex officio member of the Council but shall have no right to vote at any meeting of the Council.
|
| (4) |
The Chief Executive Officer shall hold office for a term of five years and shall be eligible for reappointment for one further term.
|
| (5) |
The Chief Executive Officers shall, in the performance of the functions and duties of office be responsible to the Council.
|
| (6) |
Without prejudice to the provisions of paragraph (5), the Chief Executive Officer shall—
| (a) |
| (i) |
carrying into effect the decisions of the Council; |
| (ii) |
day-to-day administration and management of the affairs of the Directorate; |
| (iii) |
supervision of the staff of the Directorate; |
|
| (b) |
be the Registrar of veterinary medicines; and
|
| (c) |
perform such other duties as may be assigned by the Council.
|
|
|
| 14. |
Removal of the Chief Executive Officer
| (1) |
The Chief Executive Officer may be removed from office by the Council in accordance with the terms and condition of service, for—
| (a) |
inability to perform functions of the office arising out of physical or mental infirmity;
|
| (b) |
gross misconduct or misbehavior;
|
| (c) |
incompetence or negligence of duty;
|
| (d) |
violation of the Constitution and any other written law; or
|
| (e) |
any other grounds specified in the terms and conditions of service.
|
|
| (2) |
Where the question of the removal of the Chief Executive Officer under paragraph (1) arises, the Council shall —
| (a) |
inform the Chief Executive Officer in writing of the reasons for the intended removal; and
|
| (b) |
provide the Chief Executive Officer with the opportunity to be heard in accordance with the principles of fair administrative action safeguarded under Article 47 of the Constitution.
|
|
|
| 15. |
Veterinary medicines inspectors
| (1) |
The Council shall employ such veterinary medicines inspectors as it deems necessary.
|
| (2) |
A person shall be qualified for appointment as a veterinary medicines inspector if that person—
| (a) |
is a veterinary surgeon or technologist; or
|
| (b) |
holds at least a diploma in animal health or other qualification recognized by the Council; and
|
| (c) |
has at least five years post qualification experience in a relevant field.
|
|
|
| 16. |
Staff
The Council may employ such staff as it deems appropriate for the effective performance of its functions.
|
| 17. |
Code of conduct
The members and the employees of the Council shall subscribe to the leadership and integrity code set out in the Fourth Schedule.
|
| 18. |
Protection from personal liability
Nothing done by a member of the Council or by any person working under the instructions of the Council shall, if done in good faith for the purpose of executing the powers, functions or duties of the Council under these Regulations render such member or officer personally liable for any action, claim or demand.
|
PART III – MANUFACTURING, IMPORTATION AND REGISTRATION OF VETERINARY MEDICINES
| 19. |
Veterinary medicines to be registered
| (1) |
A person shall not import, manufacture sell, transport or distribute any veterinary medicine in Kenya unless that veterinary medicine has been registered in accordance with the provisions of these Regulations.
|
| (2) |
Every person transporting a veterinary medicine in transit shall declare the veterinary medicine at the port of entry and exit.
|
|
| 20. |
Manufacturing permit
A person who intends to manufacture veterinary medicine shall make an application to the Council for a manufacturing permit.
|
| 21. |
Manufacturing of veterinary medicines
| (1) |
The Council shall, on receipt of an application made under regulation 20, before issuing a certificate of registration, cause the premises in which the manufacturing of the veterinary medicine is proposed to be conducted be inspected in order to ensure compliance to good manufacturing practice.
|
| (2) |
The inspector instructed under paragraph (1) shall submit the results of the inspection to the Council and the applicant in writing.
|
| (3) |
A manufacturer shall ensure that every stage in the manufacture of a veterinary medicine is carried out based on good manufacturing practices.
|
| (4) |
A manufacturer shall not manufacture a veterinary medicine unless the manufacturing plant has been inspected and licensed in accordance with this Regulation.
|
| (5) |
A person shall not use any premises, for the purposes of manufacturing. formulating, packaging, selling or storing veterinary medicine unless that person is in possession of a licence issued under these Regulations in respect of that premises.
|
| (6) |
Where an inspector has approved the compliance by the manufacturer, the Council shall on payment of the prescribed fee, issue a licence as set out in Form 1.3 set out in the a Third Schedule.
|
| (7) |
A person in executive authority of manufacturing company which manufactures veterinary medicines without a licence commits an offence and shall be liable, on conviction, to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months or to both.
|
|
| 22. |
Powers of inspectors with regard manufacturing plants
| (1) |
An inspector appointed under these Regulations shall have power with regard to manufacturing plants to —
| (a) |
enter the premises and inspect the plant and the process of manufacture intended to be employed in the manufacturing of the veterinary medicine and shall make a report to the Council; or
|
| (b) |
order the immediate closure of a manufacturing plant where its continued operation appears to pose a serious threat to life and safety.
|
|
| (2) |
An inspector who has ordered the closure of a plant under paragraph (1)(b) shall within twenty-four hours of the closure notify the Council of the closure and shall provide reasons for the closure.
|
| (3) |
On receipt of the notification under paragraph (2), the Council shall direct the owner of the plant to take such measures as the Council may direct to ensure conformity with these Regulations.
|
| (4) |
A manufacturer who fails, within fourteen days of receipt of the directive made by the Council under paragraph (3), to take the measures indicated in the directive shall have its licence revoked.
|
| (5) |
The Council shall publish in at least one newspaper of wide national circulation the name of a manufacturing plant closed and whose licence has been revoked under this Regulation and shall place a notice of the closure at the entrance of such plant.
|
| (6) |
The protection of right to property under Article 40 of the Constitution shall be limited for purposes of maintenance of veterinary medicine standards for the protection and safety of the public.
|
|
| 23. |
Application for registration of a veterinary medicine
| (1) |
An application for registration of—
| (a) |
a veterinary medicine shall be in accordance with Form A set out in the Third Schedule; and
|
| (b) |
a veterinary medicine used as pesticides shall be Form B set out in the Third Schedule.
|
|
| (2) |
An applicant shall, in addition to the information required to be paragraph (1), furnish such further information and material as may be required by the Council for the proper evaluation of the veterinary medicine in respect of which the application is made.
|
| (3) |
The Council shall, on approval of an application made under this Regulation and on payment of the prescribed fees, register the veterinary medicine or veterinary pesticide and issue a certificate in Form D1 or D2, respectively set out in the Third Schedule.
|
| (4) |
A certificate of registration issued under these Regulations shall, unless suspended or revoked, be in force for a period of five years.
|
| (5) |
The Council shall not approve an application made under paragraph (1) unless satisfied that the applicant has attained the prescribed standards and satisfied all requirements for registration.
|
|
| 24. |
Renewal of registration of veterinary medicine
| (1) |
A person may make an application to the Council, for renewal of registration of a veterinary medicine in Form E set out in the Third Schedule.
|
| (2) |
The Council shall, before renewing registration under this Regulation conduct a pharmaco-vigilance study on the manufacturer.
|
| (3) |
An applicant shall on receipt of approval for renewal of registration from the Council, make payment of the fees as prescribed in the Fifth Schedule.
|
|
| 25. |
Clinical and non-clinical trials and toxicity testing
| (1) |
The Council may require that a clinical and non-clinical trial and toxicity testing be done before the registration of a veterinary medicine.
|
| (2) |
Despite paragraph (1) a veterinary pesticide shall not be registered unless an efficacy and an acute toxicological study is carried out by an accredited laboratory.
|
| (3) |
A person who or institution which intends to apply for accreditation under this Regulation shall, before making such application, obtain approval from the Council.
|
| (4) |
A person who or institution which desires to conduct clinical and non-clinical trials and toxicity testing shall apply to the Council for accreditation.
|
| (5) |
The cost of any clinical trial or toxicity testing shall be borne by the applicant.
|
|
| 26. |
Conditions for registration of veterinary medicines and pesticides
| (1) |
A veterinary medicine and pesticide shall not be registered unless a laboratory analysis has been carried out by an accredited laboratory.
|
| (2) |
A person who intends to register a veterinary medicine shall submit to the Council a certificate of analysis from an accredited laboratory together with the application for registration of the veterinary medicine
|
|
| 27. |
Veterinary Medicines Registration Committee
| (1) |
There is hereby established a standing committee of the Council to be known as the Veterinary Medicines Registration Committee.
|
| (2) |
The Committee shall be responsible for matters relating to registration of veterinary medicines under these Regulations and in particular shall be responsible for—
| (a) |
evaluating applications for veterinary medicines registration and shall make recommendations to the Council; and
|
| (b) |
issuing provisional approval pending the issuance of the veterinary medicines registration certificate by the Council.
|
|
| (3) |
A provisional certificate issued under paragraph (2)(b) shall be valid for a period of not more than three months.
|
| (4) |
The Council shall consider an application made under this Regulation, and if satisfied of the safety, efficacy and quality of the veterinary medicine or veterinary pesticide, shall register the veterinary medicine in accordance with Form C1 and C2 set out in the Third Schedule.
|
| (5) |
Upon registration of a veterinary medicine under paragraph (4), the applicant shall be issued with a certificate of registration in Form D1 or D2 set out in the Third Schedule.
|
| (6) |
The Council may, while considering a veterinary medicine for registration under paragraph (4), approve the details as supplied by the applicant or approve it with such modifications as it may consider appropriate in respect of the following particulars—
| (a) |
the name under which the veterinary medicine may be sold;
|
| (c) |
the statement of the representations to be made for the promotion of the veterinary medicine in respect of the—
| (i) |
claim to be made for the veterinary medicine; |
| (ii) |
route of administration; |
| (iv) |
contra-indications, side effects and precautions, if any; |
| (vi) |
withdrawal period for food producing animals; |
| (vii) |
proposed category; and |
|
|
|
| 28. |
Rejection of application for registration of a veterinary medicine
The Council shall, if it is not satisfied as to the safety, efficacy or quality of the veterinary medicine, reject the application for the registration of the veterinary medicine and inform the applicant, in writing, the reasons for rejection.
|
| 29. |
Registration of veterinary medicine researched outside Kenya
| (1) |
The Council may, before registering a new veterinary medicine for which the research work has been conducted in another country and its efficacy, safety, and quality established in that country, require an investigation on the pharmaceutical, pharmacological and other aspects of the veterinary medicine to be conducted and clinical trials to be carried out as necessary to establish its safety, efficacy, quality and where applicable the biological availability to be established under local conditions.
|
| (2) |
Despite paragraph (1), the Council may register a new veterinary medicine and require the investigations and clinical trials specified therein to be conducted after its registration.
|
|
| 30. |
Suspension and revocation of certificate of registration
| (1) |
The Council may suspend or revoke a certificate of registration issued under these Regulations for such period as the Council may determine.
|
| (2) |
The Council shall not revoke or cancel the certificate of registration unless—
| (a) |
where matters stated in the application on which the certificate of registration was granted were false or incomplete in any material particular;
|
| (b) |
where provision of the certificate of registration has to a material extent been contravened by the holder of the certificate;
|
| (c) |
where the premises on which, or on part of which, a veterinary medicine is manufactured, assembled or stored by or on behalf of the holder of the certificate of registration are unsuitable for the manufacturing, assembling or storage of veterinary medicines; or
|
| (d) |
where new information has been discovered by the Council which renders the veterinary medicines unsafe or dangerous.
|
|
| (3) |
Where a certificate has been revoked under paragraph (2)(d), the Council shall on delivery of the information to the manufacturer —
| (a) |
inspect the plant to ensure all the medicine or the relevant batch has been destroyed;
|
| (b) |
disseminate the information to the public through a newspaper advertisement in at least two daily newspapers of nationwide circulation; and
|
| (c) |
recall any medicine which has been distributed.
|
|
|
| 31. |
Period for renewal of registration and retention of veterinary medicine
| (1) |
The Council may grant the renewal of registration as provided under regulation 24 for a period not exceeding five years.
|
| (2) |
On the expiry of five years a veterinary medicine may be retained in the register upon an application made by the certificate holder which shall—
| (a) |
include a declaration that the requirements met by the veterinary medicine during registration has not changed; and
|
| (b) |
include payment of the requisite fees.
|
|
|
PART IV – CLASSIFICATION AND CATEGORIZATION OF VETERINARY MEDICINES
| 32. |
Classification of Veterinary Medicines
| (1) |
The Cabinet Secretary shall classify veterinary medicines in the classes set out in Part I of the Second Schedule.
|
| (2) |
The Cabinet Secretary may, on the advice of the Council, vary the classification of the various veterinary medicines depending on the therapeutic use and current information for the medicine.
|
|
| 33. |
Categorization of Veterinary Medicines
| (1) |
The Cabinet Secretary shall categorize veterinary medicines as set out in Part II of the Second Schedule.
|
| (2) |
The Cabinet Secretary shall be guided by the Council on the categorization of veterinary medicines and shall place the veterinary medicines in the following categories—
| (a) |
Category I relating to prescription only medicine—Veterinary Surgeon which shall comprise two sub-categories as follows—
| (i) |
Category IA relating to Controlled Veterinary Medicines includes products which contain narcotic or psychotropic substances or other substances very dangerous at small quantities; |
| (ii) |
Category IB relating to other prescription only medicine, comprising products intended for administration following a diagnosis or clinical assessment by a veterinary surgeon or dispensed by a veterinary surgeon or a person with equivalent qualification; |
|
| (b) |
Category II relating to prescription only medicine veterinary surgeon, pharmacist, and veterinary technologist who has served for at least five years in a veterinary pharmacy which contains—
| (i) |
veterinary medicines for use in food-producing animals; |
| (ii) |
veterinary medicines in respect of which special precautions shall be taken in order to avoid any unnecessary risk to the target species, the person administering the products to the animal and the environment; Provided that the requirement in this paragraph shall not apply if all the following criteria are met— |
|
| (c) |
Category III relating to authorised veterinary medicine general sales list which may be traded by veterinary surgeons and all categories of veterinary paraprofessionals and includes—
| (i) |
pest control veterinary medicines provided that the medicine are sold in the original package without repackaging and with an intact label; |
| (ii) |
endoparasiticide veterinary medicines considered safe for use in food animals; |
| (iii) |
endoparasiticide veterinary medicines for use in non-food animals; or |
| (iv) |
any other product the Directorate may prescribe. |
|
| (d) |
Category IV which includes alternative veterinary medicine general sales list.
|
|
|
| 34. |
Use of veterinary pesticides
| (1) |
A veterinary pesticide shall be—
| (a) |
sold only in the original sealed and labeled package as registered by the Council;
|
| (b) |
stored in accordance with the instructions on the label on the original container; and
|
| (c) |
used according to the instructions on the label.
|
|
| (2) |
The empty container of a veterinary pesticide shall be disposed in accordance with the instructions given on the label.
|
| (3) |
A person who removes or defaces a label on a veterinary pesticide or package commits an offence.
|
| (4) |
A person who sells, dispenses or otherwise gives out a veterinary pesticide in contravention of this Regulation commits an offence and shall be liable, on conviction, to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months or to both.
|
|
PART V – VETERINARY PHARMACY
| 35. |
Standards of Veterinary Pharmacy
| (1) |
A veterinary pharmacy shall satisfy the following minimum standards—
| (a) |
be located away from known fire hazards;
|
| (b) |
be a separate entity from other veterinary operations such as veterinary clinic, ambulatory and laboratory services;
|
| (c) |
be separated from non-complementary businesses;
|
| (d) |
have restricted access by personnel to Category I and II veterinary medicine; and
|
| (e) |
be vermin-proofed, including protection against insects and rodents.
|
|
| (2) |
A veterinary pharmacy shall—
| (a) |
demonstrate adequate security for the safety of veterinary medicine;
|
| (b) |
demonstrate appropriate storage conditions that shall maintain the temperature, lighting and humidity requirements as prescribed in the manufacturers' specifications of veterinary medicines;
|
| (c) |
have the floor and the wall of the building constructed from materials that are easy to clean, impervious and resistant to corrosion by chemicals;
|
| (d) |
have lockable safety cabinets to protect the controlled veterinary medicines;
|
| (e) |
provide the personnel with protective clothing for use only within the premises;
|
| (f) |
be of appropriate size and have sufficient space for carrying out of all necessary operations provided for the orderly receipt, warehousing and dispatch of the various classes of veterinary medicines and, in particular, a quarantine area for isolation when necessary, including isolation of faulty packs and recalled or expired veterinary products;
|
| (g) |
provide a disposal system, acceptable to the Council, for safe disposal of expired veterinary medicines;
|
| (h) |
be operated by competent staff as provided for in the permit;
|
| (i) |
display warning notices indicating hazardous areas;
|
| (j) |
label the various sections in the business;
|
| (k) |
store veterinary medicines only in the designated areas, which shall be labeled;
|
| (l) |
have emergency lighting, fire-fighting equipment and fully equipped first aid kit;
|
| (m) |
display emergency protocols within the veterinary pharmacy advising personnel on procedures to follow in case of emergencies;
|
| (n) |
display standard operating procedures;
|
| (o) |
retain records of the movement of all veterinary medicines for at least five years; and
|
| (p) |
provide separate sanitary facilities for each gender.
|
|
|
| 36. |
Qualifications to operate a veterinary pharmacy
A person who is a veterinary surgeon, or holds an equivalent qualification in matters of pertaining to veterinary medicines as determined by the Council, may apply to the Council for a permit to undertake a veterinary pharmacy.
|
| 37. |
Council to approve veterinary pharmacy businesses
| (1) |
The Council shall have power to approve any of the following types of veterinary pharmacy businesses—
| (a) |
manufacturing of veterinary medicines;
|
| (b) |
wholesaling of veterinary medicines; and
|
| (c) |
retailing of veterinary medicines.
|
|
| (2) |
Upon approval of the veterinary business specified under paragraph (1), the Council shall register the veterinary pharmacy premises in Form J set out in the Third Schedule.
|
|
| 38. |
Practising permit
| (1) |
A person shall not practice the business of veterinary pharmacy unless he holds a valid practicing permit issued by the Council.
|
| (2) |
A person who intends to practice the business of veterinary pharmacy shall make an application in Forms H1, H2 and H2, respectively of the Third Schedule.
|
| (3) |
The Council shall issue a practicing permit if the premises in which the applicant intends to operate has been approved by the Council in accordance with these Regulations.
|
| (4) |
An application for a practicing permit shall be in—
| (a) |
Form 1.1 for veterinary pharmacy practitioner;
|
| (b) |
Form 1.2 for a wholesaler dealer permit; or
|
| (c) |
Form 1.3 for manufacturers' permit.
|
|
| (5) |
An applicant under paragraph (1) shall, on approval of the application, by the Council, pay the fee prescribed in the Fifth Schedule.
|
| (6) |
The Council, upon confirming that the applicant has satisfied the conditions of a permit, and upon payment of the fee under paragraph (4) issue an applicant with a practicing permit as set out in Form A set out in the Third Schedule for veterinary pharmacy practices and Form B for wholesale pharmacy practice.
|
| (7) |
The practice permit shall be valid for a period of one year commencing on the 1st of January and ending on the 31st of December of each year.
|
| (8) |
The Council shall by the 31st March of every year publish, in the Kenya Gazette or in a newspaper of wide national circulation, the registered veterinary pharmacy businesses.
|
| (9) |
A person who conducts the business of veterinary pharmacy in premises not registered by the Council under these Regulations commits an offence, and shall be liable, on conviction, to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months or to both.
|
|
| 39. |
Practice permit to be displayed
| (1) |
A person who carries on the business of a veterinary pharmacy shall display his practice permit on a conspicuous place within the premises in which the business is being carried on.
|
| (2) |
A person who contravenes the provisions of this Regulation commits an offence and shall be liable, on conviction, to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months or to both.
|
|
| 40. |
Wholesale permit
A person who intends to trade in veterinary medicines, in bulk, shall make an application to the Council for a wholesale permit and support the application with the following—
| (a) |
a certified copy of the certificate of registration or incorporation of the business name or body corporate and the memorandum and articles of association;
|
| (b) |
provide evidence that the business is under the management of a supervisor who—
| (i) |
is a registered veterinary surgeon permitted to carry out the business of veterinary pharmacy; and |
| (ii) |
is a member of the Board of directors of the body corporate, and who is not acting in a similar capacity for any other body corporate. |
|
|
| 41. |
Retail permit
| (1) |
A person who intends to trade in veterinary medicines as a retailer shall make an application to the Council for a retail permit in Form H set out in the Third Schedule.
|
| (2) |
This regulation shall be applicable to a manufacturer and a wholesaler of veterinary medicines who desires to carry out retail trade.
|
|
PART VI – VETERINARY PHARMACY AND COMPLEMENTARY BUSINESSES
| 42. |
Complementary businesses
| (1) |
A veterinary pharmacy may be conducted alongside complementary businesses including the sale of human medicines, horticultural chemicals, agro-forestry chemicals and fertilizer, animal foodstuffs, seeds and agricultural equipment.
|
| (2) |
The building or premises of a veterinary pharmacy stocking or trading in complementary businesses referred to in paragraph (1), shall be built or adapted in such manner as to provide a separate and distinct partition for the veterinary products from any other products.
|
| (3) |
In addition to the provisions of paragraph (2), the premises shall be built or adapted in such manner as to provide separate and distinct partitions for veterinary medicines and veterinary pesticides such that no mixing of the two classes of products is permissible.
|
|
| 43. |
Stock, supply and distribution of veterinary medicine
| (1) |
A veterinary medicine shall be supplied or distributed only through a wholesale or retail business registered as a veterinary pharmacy business.
|
| (2) |
A holder of wholesale or veterinary pharmacy permit issued by the Council may sell veterinary medicines in bulk to registered retail dealers.
|
| (3) |
A holder of a retail dealer permit, issued by the Council, may sell veterinary medicines to the public.
|
| (4) |
A person shall not supply authorised human medicinal product for administration to an animal other than a product supplied by a veterinary surgeon or in accordance with a written prescription from a veterinary surgeon.
|
| (5) |
A veterinary pharmacy practitioner who dispenses a veterinary medicine belonging to Category I or II to any member of the public without a prescription, commits an offence and shall be liable, on conviction, to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months or to both.
|
|
PART VII – MARKET AUTHORIZATION
| 44. |
Authorization to import or export veterinary medicine
| (1) |
A person who intends to import or export a veterinary medicine shall apply to the Council for a permit in Form K set out in the Third Schedule.
|
| (2) |
The Council shall in determining the application under paragraph (1) consider whether the applicant has met the conditions for importation and exportation in these Regulations, and if so, shall on payment of the prescribed fee, issue the applicant with an import permit in Form L set out in the Third Schedule.
|
| (3) |
A holder of a market authorization may apply to the Council for a wholesale permit to authorize him to import the veterinary medicine specified in the authorization.
|
| (4) |
An authorised wholesale dealer may import a veterinary medicine if—
| (a) |
the authorization covers the veterinary product; and
|
| (b) |
the wholesale dealer has acquired the written consent of the holder of the market authorisation in writing before importation.
|
|
| (5) |
A veterinary surgeon may, with approval of the Council, import an orphan veterinary medicine.
|
|
PART VIII – VETERINARY MEDICINE INSPECTORS
| 45. |
Appointment of veterinary medicine inspectors
The Council shall appoint duly qualified persons on such terms and conditions of service as it may deem appropriate, to serve as veterinary medicine inspectors.
|
| 46. |
Identification of veterinary medicines inspector
| (1) |
The Council shall publish in the Kenya Gazette every inspector appointed under regulation 45 and issue each inspector with an official identity card which shall have a passport size photo of the inspector, duly stamped and signed by the Chief Executive Officer or his authorized agent.
|
| (2) |
Every inspector shall, in conducting inspections under these Regulations, identify himself using the identity card issued under paragraph (1).
|
| (3) |
A person who ceases to be an inspector shall surrender his identify card to the Chief Executive Officer or his authorized agent.
|
|
| 47. |
Powers of veterinary medicines inspectors
| (1) |
An inspector shall have the power, at all reasonable times, to—
| (a) |
enter upon the premises of any manufacturer, distributor or veterinary pharmacy and to inspect any books, papers, records or writings, veterinary medicines, whether patent or otherwise, or any article stored or offered for sale or used in the business; or
|
| (b) |
enter any premises in which he has reasonable cause to suspect that a breach of the law has been or is being committed, and to make such examination and inquiry and to do such other activities, including impounding and seizing suspect veterinary products, closing suspect premises and the taking of samples, as may be necessary for the purpose of ascertaining whether the provisions of these Regulations are being complied with.
|
|
| (2) |
| (a) |
for the purpose of inspecting a veterinary medicine use Form F set out in the Third Schedule; and
|
| (b) |
for any impounding of suspect veterinary medicines, use Form G set out in the Third Schedule.
|
|
| (3) |
An inspector shall observe confidentiality in the findings of his inspection.
|
| (4) |
An inspector shall be liable for any act of negligence he may commit in the performance of his duties.
|
|
PART IX – FINANCIAL PROVISIONS
| 48. |
Funds of the Directorate
| (1) |
The funds of the Directorate shall comprise of—
| (a) |
such monies as may be appropriated by Parliament for the purposes of the Directorate;
|
| (b) |
such monies as may accrue to or vest in the Directorate in the course of the exercise of its functions under these Regulations; and
|
| (c) |
monies from any other source provided for, donated or lent to the Directorate.
|
|
|
| 49. |
Financial year
The financial year of the Directorate shall be the period of twelve months from first July to thirtieth of June.
|
| 50. |
Annual estimates
| (1) |
Three months before the commencement of each financial year, the Council shall cause to be prepared estimates of revenue and expenditure of the Directorate for that year.
|
| (2) |
The annual estimates shall make provision for all the estimated expenditure of the Directorate for the financial year concerned, and in particular shall provide for—
| (a) |
the payment of salaries, allowances and other charges in respect of the staff of the Directorate;
|
| (b) |
the payment of pensions, gratuities and other charges in respect of retirement benefits which are payable out of the funds of the Directorate;
|
| (c) |
the proper maintenance of buildings and grounds of the Directorate;
|
| (d) |
the acquisition, maintenance, repair and replacement of the equipment and other movable property of the Directorate; and
|
| (e) |
the creation of such reserve funds to meet future or contingent liabilities in respect of retirement benefits, insurance or replacement of buildings or equipment or in respect of such other matter as the Directorate may find appropriate.
|
|
| (3) |
The annual estimates shall be approved by the Council before the commencement of the financial year to which they relate and shall be submitted to the Cabinet Secretary for approval.
|
| (4) |
The Directorate shall not increase any sum provided in the estimates without the consent of the Cabinet Secretary.
|
|
| 51. |
Investment of funds
The Council may invest any of the funds of the Directorate in a manner approved by the Treasury for the investment of trust funds.
|
| 52. |
Accounts and Audit
| (1) |
The Council shall cause to be kept all proper books and records of accounts of the income, expenditure, assets and liabilities of the Directorate.
|
| (2) |
The accounts of the Directorate shall be audited in accordance with the Public Finance Management Act (Cap. 412A).
|
|
PART X – OFFENCES
| 53. |
Offences
A person commits an offence if that person—
| (a) |
imports, exports, manufactures, stores, distributes, sells or otherwise handles a veterinary medicine that has not been registered under these Regulations;
|
| (b) |
imports a veterinary medicine without a permit issued under these Regulations;
|
| (c) |
manufactures, stores, distributes or sells a veterinary medicine in premises which have not been registered under these Regulations;
|
| (d) |
presents for sale or distribution, expired, adulterated, Counterfeit or unlabeled veterinary medicines;
|
| (e) |
sells a veterinary medicine in any form other than the original sealed and labeled package, as registered with the Directorate;
|
| (f) |
sells or distributes a veterinary medicine in any other area other than a registered premises;
|
| (g) |
uses a veterinary medicine for purposes other than that for which it was registered, prescribed or dispensed;
|
| (h) |
supplies human medicinal product for administration to an animal in contravention of these Regulations;
|
| (i) |
is in possession of a Category I or Category II veterinary medicine other than in accordance with these Regulations;
|
| (j) |
advertises a veterinary medicine in contravention of these Regulations;
|
| (k) |
buys a veterinary medicine for resale in bulk while not being in possession of a wholesale dealer permit issued under these Regulations;
|
| (l) |
sells a veterinary medicine to the public while not being in possession of a retail dealer's permit issued in accordance with these Regulations;
|
| (m) |
undertakes research on imported and unregistered veterinary medicine without authorization from the Council;
|
| (n) |
obstructs or fails to assist the Council or veterinary medicine inspector in the performance of their lawful duties under these Regulations;
|
| (o) |
provides the Council or inspector with false or misleading information;
|
| (p) |
operates a veterinary pharmacy after it has been closed by an inspector; or
|
| (q) |
contravenes any provision of these Regulations.
|
|
| 54. |
Officers of corporations
If an institution commits an offence under these Regulations, any officer, director or agent of the corporation who directed, authorized, assented to, acquiesced in, or participated in the commission of the offence shall be a party to and shall be considered to have committed the offence and shall be liable on conviction to the punishment provided for the offence, whether or not the corporation has been prosecuted or convicted.
|
| 55. |
Passing confidential information without authorization
A member of the Council or staff of the Directorate, who—
| (a) |
being in possession of confidential information, however obtained without authorization of the Council—
| (ii) |
attempts, offers or threatens to divulge it, |
|
other than in accordance with these Regulations or other written law; or
| (b) |
willfully obtains or seeks to obtain confidential information to which he is not entitled, commits an offence.
|
|
| 56. |
Penalties
A person who contravenes these Regulations where no Penalties penalty has been prescribed, shall on conviction be liable to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months, or to both.
|
PART XI – GENERAL PROVISIONS
| 57. |
Advertising veterinary standards for medicines
| (1) |
A person who intends to advertise a veterinary medicine, shall make an application to the Council for authority to advertise.
|
| (2) |
The Council may, on payment of the requisite fee and subject to such conditions as the Council or these Regulations may impose, grant the authorization applied for under paragraph (1).
|
| (3) |
An advertisement for a veterinary medicine shall not be misleading or contain any medicinal claim that is not in the summary of the product characteristics registered with the Council.
|
| (4) |
A veterinary medicine listed under Category I and II shall not be advertised unless—
| (a) |
in the case of a veterinary medicine listed under Category I, the advertisement is aimed at veterinary surgeons; and
|
| (b) |
in the case of a veterinary medicine listed under Category II, the advertisement is aimed at veterinary surgeons or suitably qualified persons as recognized by the Council.
|
|
| (5) |
A veterinary medicine listed in Category III and IV may be advertised to general members of the public.
|
|
| 58. |
Exemptions to Regulations
These Regulations do not apply to—
| (a) |
inactivated autogenous vaccine that is manufactured, on the instructions of a veterinary surgeon, from pathogens or antigens obtained from an animal and used for the treatment of that animal or—
| (i) |
other animals on the same site; |
| (ii) |
animals intended to be sent to those premises; or |
| (iii) |
animals on a site that receives animals from those premises; or |
|
| (b) |
animal foodstuff supplements with no therapeutic claim.
|
|
| 59. |
Discretion of Council
The Council shall have the discretion to—
| (a) |
approve the importation and use of any veterinary medicine for handling emergency situations;
|
| (b) |
cancel the registration of any veterinary medicine that is considered to be harmful to animal and human health and the environment; or
|
| (c) |
restrict the use of any specified veterinary medicine.
|
|
| 60. |
Copy of certificate or authorization
| (1) |
A person who has been issued with a certificate or authorization under these Regulations may, if the certificate or authorization is defaced, damaged or lost, on application to the Council and on payment of the prescribed fee, be issued with a copy of the certificate.
|
| (2) |
The copy of a certificate or authorization issued under paragraph (1) shall—
| (i) |
be issued only where the document is in force at the time the application is made; |
| (ii) |
be valid for the same period as the original document; and |
| (iii) |
bear the words "DUPLICATE COPY". |
|
|
| 61. |
Fees
Any fees payable under this Act is as set in the Fifth Schedule.
|
| 62. |
Supercession
These Regulations shall supersede any other Regulations on matters concerning veterinary medicines.
|
| 63. |
Transition and validity of licences and permits
Upon the commencement of these Regulations any certificate in force shall be deemed to have been issued under these Regulations and shall remain in force until its expiry.
|
FIRST SCHEDULE [r. 8(6)]
CONDUCT OF THE AFFAIRS OF THE COUNCIL
1. Meetings of the Council.
The Council shall meet at least four times in a year and not more than eight times a year, except in case of an emergency, for the transaction of its business, and such meetings shall be held at such places and times and on such days as the Council may determine.
2. Chairperson and vice-Chairperson
| (1) |
The Council shall at its first meeting elect its vice-Chairperson from among the three members appointed under regulation 8(3)(f).
|
| (2) |
The Chairperson shall preside at all meetings of the Council at which he is present, and in case of his absence the vice-Chairperson shall preside, but in the absence of both the Chairperson and vice-Chairperson, members present and constituting a quorum shall elect one among their number to be the Chairperson for purposes of the meeting.
3. Quorum
The quorum of a meeting of the Council shall be five, at least three of whom shall be veterinary surgeons.
4. Decisions of Council
The decisions of the Council shall be by a simple majority of the votes of the members present, but in the case of an equality of votes, the Chairperson or person presiding shall have a casting vote.
5. Minutes
|
| (1) |
The Minutes of each meeting shall be kept in the minute book, after they have been confirmed by the Council and signed by the Chairperson at a subsequent meeting of the Council.
|
| (2) |
The deliberations and minutes of meetings of the Council shall be confidential.
6. Vacancy not to invalidate proceedings
No proceedings of the Council shall be invalid by reason only of a vacancy among the members thereof.
7. Council to determine own procedure
Subject to this Schedule, the Council may determine its own procedure.
8. Invitation of other persons
The Council may invite any person to participate in its deliberations but a person who has been invited shall have no right to vote.
|
SECOND SCHEDULE [rr. 32(1), 33(1)]
CATEGORIES AND CLASSES OF VETERINARY MEDICINES
Part I - CLASSES OF VETERINARY MEDICINES
1. List of veterinary pharmaceuticals.
| (iii) |
Analgesics and anti-inflammatories |
| (iv) |
Drugs acting on the nervous system |
| (vi) |
Other systemic therapeutic agents |
| (vii) |
Local therapeutic agents |
| (v) |
Probiotics and enzymes. |
4. Equipment and materials.
| (ii) |
Veterinary medicine administration devices. |
| (iii) |
Any other material and equipment of veterinary relevance. |
5. List of alternative medicines.
| (iii) |
Performance enhancers. |
| (v) |
Any other pesticides of veterinary relevance |
| (vi) |
And other ecto-parasiticides |
| (vii) |
substances used for euthanasia |
Part II - CATEGORIES OF VETERINARY MEDICINES
| (a) |
Prescription Only Medicine—Veterinary surgeon (abbreviated to POM-V);
|
Category IA POM-V Controlled Veterinary Medicine
| 11. |
Morphine and its derivatives.
|
| 13. |
Methadone (amidone); its salts.
|
| 16. |
Phenoperidine; its salts.
|
| 19. |
Any other veterinary medicine relevant in the category.
|
Category IB — Other POM - V
| (1) |
Alkali fluorides, other than those specified in Part II of this List.
|
| (2) |
Chlorpropamide; its salts.
|
| (7) |
Alkaloids, the following; their salts, simple or complex.
|
| (8) |
Amidopyrine; its salts; amidopyrine sulphonates; their salts.
|
| (9) |
Amino-alcohols esterified with benzoic acid, phenylacetic acid, phenylpropionic acid, cinnamic acid or the deriv.
|
| (10) |
Amitriptyline; its salts.
|
| (14) |
B-Aminopropylbenzene and B-aminoisopropylbenzene and any compound structurally derived from either of the
|
| (15) |
Barbituric acid; its salts; derivatives of barbituric acid; their salts; compounds of barbituric acid, their salts, their
|
| (16) |
Belladonna, alkaloids of.
|
| (22) |
Chlordiazepoxide; its salts.
|
| (23) |
Chlormethiazole; its salts.
|
| (28) |
Curare, alkaloids of; curare bases.
|
| (31) |
Dehydroemetine; its salts.
|
| (32) |
Derivatives of stilbene, dibenzyl or naphthalene with oestrogenic activity; their esters.
|
| (34) |
Dextromethorphan; its salts.
|
| (37) |
Digitalis, glycosides of; other active principles of digitalis.
|
| (44) |
Ergot, alkaloids of, homologues and hydrogenated.
|
| (54) |
Hydroxypethidine; its salts.
|
| (65) |
Mercaptopurine; its salts; derivatives of mercaptopurine; their salts.
|
| (69) |
Monofluroacetic acid; its salts.
|
| (76) |
p-Aminobenzenesulphonamide; its salts, derivatives of p-amino-benzenesulphonamide having any of the hydrogen
|
| (77) |
p-Aminobenzoic acid, esters of; their salts.
|
| (78) |
p-Amino-salicylic acid; its salts; any preparation of p-Amino salicylic acid; its salts.
|
| (80) |
Parenteral Anti-histamine substances, the following; their salts; their molecular compounds—
|
| (81) |
Parenteral Arsenical substances
|
| (85) |
Phencyclidine; its salts.
|
| (89) |
Phenothiazine, derivatives of
|
| (97) |
Quinapyramine and analogous substances; their salts.
|
| (99) |
Rauwolfia, alkaloids of; their derivatives.
|
| (100) |
Steroid compounds with androgenic or oestrogenic or progestational activity; their esters.
|
| (112) |
Any other veterinary medicine relevant in the category
|
CATEGORY II — PRESCRIPTION ONLY MEDICINE (POM-VT)
Prescription Only Medicine—Veterinary surgeon
| (2) |
Androgenic, oestrogenic and progestational substances,
|
| (4) |
Anti-histamine substances, the following; their salts; their molecular compounds
|
| (22) |
Dextromethorphan; its salts.
|
| (24) |
Digitalis, glycosides of; other active principles of digitalis.
|
| (36) |
Halogeneted Salicylanides and Nitrophenols
|
| (39) |
Hydroxypethidine; its salts.
|
| (49) |
Mercaptopurine; its salts; derivatives of mercaptopurine; their salts.
|
| (50) |
Mercury and its compound.
|
| (52) |
Metallic oxalates, other than potassium quadroxalate, if in the form of photographic solutions
|
| (62) |
Phenylene diamines; toluene diamines; other alkylated-benzenediamines; their salts.
|
| (75) |
Sulphonamides, parenteral.
|
| (78) |
Vaccines, administered by a veterinary surgeon or veterinary para-professional
|
| (81) |
Any other veterinary medicine relevant in the category
|
CATEGORY III — Authorized Veterinary Medicines-General Sales List (AVM-GSL)
| (2) |
Benzimidazoles, oral preparation of
|
| (4) |
Creosot obtained from wood.
|
| (6) |
Dyflos, except the preparations in Part 2
|
| (12) |
Hydroxy-N-N-dimethyltryptamines, esters or ethers of these; salts of any of the foregoing.
|
| (13) |
Imidazothiozoles, oral preparation of
|
| (15) |
Monofluroacetic except the preparation in Part 2
|
| (18) |
p-Amino-salicylic acid; its salts; any preparation of p-Amino salicylic acid; its salts.
|
| (20) |
polymethylene diguanidines; di-p-anisyl-p-phenetylguanidine.
|
| (21) |
Praziquantel oral preparation
|
| (25) |
Sulphonamides, oral and topical preparations.
|
| (29) |
Any other veterinary medicine relevant in the category
|
CATEGORY IV- Alternative Veterinary Medicine — General Sales List (Alt VM-GSL)
| (1) |
Anthelmintic preparations
|
| (2) |
Anti-inflammatory preparations
|
| (3) |
Antibacterial preparations
|
| (5) |
Antispasmodic preparations
|
| (11) |
Laxatives, purgatives and cathartics.
|
| (12) |
Biopesticide preparations
|
| (13) |
IGalactagogue preparations
|
| (14) |
Antiprotozoa preparations
|
| (15) |
Disinfectants and antiseptics
|
| (16) |
Any other veterinary medicine relevant in the category
|
THIRD SCHEDULE [rr. 23(1), 38(6)]
FORMS
|
FORM A
|
|
|
|
APPLICATION FOR REGISTRATION OF A VETERINARY MEDICINE
|
|
(to be submitted as one original hard-copy and one electronic copy (in MS-Word)
|
|
The RegistrarVeterinary Medicines DirectorateKABETE
|
|
Application Number
|
|
|
|
Date of submission of the dossier
|
|
|
|
Name of the 1st Evaluator
|
|
|
Signature
|
|
|
Name of the 2nd Evaluator
|
|
|
Signature
|
|
|
Date of 1st evaluation
|
|
|
|
Date of 2nd Evaluation
|
|
|
|
Number of files received
|
|
|
|
CONCLUSION OF THE ASSESSMENTRECOMMENDED (no outstanding issues)QUERY RAISED (Indicate the sections where query is raised)REJECTED (indicate the module(s) that led to the rejection)
(Please delete which does not apply)
|
|
|
|
TYPE OF APPLICATION — VETERINARY PHARMACEUTICALS, BIOLOGICALS, NUTRIENTS, EQUIPMENT AND MATERIALS, ALTERNATIVE MEDICINE, POISONS (tick the applicable class)
|
|
|
PART 1: ADMINISTRATIVE INFORMATION
|
|
|
SECTION 1: PARTICULARS OF THE VETERINARY MEDICINE
|
|
|
1.1 Name and address of Applicant
|
|
|
(Company) Name:Address:Country:Country Code:Office telephone number:Mobile telephone number:E-Mail:
|
|
|
For VMD use only
|
|
|
1.2
|
Trade Name of the veterinary medicine (Proprietary Veterinary Medicine Name)
|
|
|
For VMD use only
|
|
|
1.3
|
International Non-proprietary Name (INN) of the Active Ingredient (AI)
|
|
|
For VMD use only
|
|
|
1.4
|
Strength of Active Ingredient (AI) per unit dosage of the veterinary medicine:
|
|
|
For VMD use only
|
|
|
1.5
|
Pharmaceutical Dosage form and route of administration of the veterinary medicine
|
|
|
1.5.1
|
Pharmaceutical Dosage form of the veterinary medicine:
|
|
|
1.5.2
|
Route(s) of administration (use current list of standard terms — British Pharmacopoeia)
|
|
|
For VMD use only
|
|
|
1.6
|
Packing/pack size of the veterinary medicine:
|
|
|
For VMD use only
|
|
|
1.7
|
Visual description of the veterinary medicine(Add as many rows as necessary)
|
|
|
|
|
|
For VMD use only
|
|
|
1.8
|
Proposed shelf life (in months):
|
|
|
1.8.1
|
Proposed shelf life (after reconstitution or dilution):
|
|
|
1.8.2
|
Proposed shelf life (after first opening container):
|
|
|
1.8.3
|
Proposed storage conditions:
|
|
|
1.8.4
|
Proposed storage conditions after first opening:
|
|
|
For VMD use only
|
|
|
1.9
|
Pharmacotherapeutic group and Anatomical Therapeutic Chemical (ATC) Code
|
|
|
1.9.1
|
Pharmacotherapeutic group:
|
|
|
1.9.2
|
ATC Code: (Please use current ATC code)
|
|
|
1.9.3
|
If no ATC code has been assigned, please indicate if an application for ATC code has been made: ☐
|
|
|
For VMD use only
|
|
|
1.10
|
Legal category
|
|
|
1.10.1
|
Proposed dispensing category:
|
|
|
1.10.2
|
For veterinary medicine subject to veterinary prescription: Controlled Veterinary Medicine (POM-V Category IA) or Other Prescription Only Medicine, (Category IB-POM-V)(Please delete which does not apply)
|
|
|
1.10.3
|
For veterinary medicine not subject to veterinary prescription:
|
|
|
For VMD use only
|
|
|
1.11
|
Country of origin or country of release:
|
|
|
For VMD use only
|
|
|
1.12
|
Marketing Authorisation in the country of origin and other countries. (Attach certificate of veterinary medicine from competent regulatory authority)If not registered, state reasons
|
|
☐ Authorised Country:Date of authorisation (dd-mm-yyyy):Proprietary name:Authorisation Certificate number:☐ Refused Country:Date of refusal (dd-mm-yyyy):Reason for Refusal:
|
☐ Withdrawn (by applicant after authorisation)Country:Date of withdrawal (dd-mm-yyyy):Proprietary name:Reason for withdrawal:☐ Suspended/revoked (by competent authority) date of suspension/revocation (dd-mm-yyyy):Reason for suspension/revocation:Proprietary name:
|
|
|
For VMD use only
|
|
|
1.13
|
Pre-registration analysis of the Veterinary Medicine (Attach certificate of analysis from a laboratory recognized by the Directorate)
|
|
|
For VMD use only
|
|
|
1.14
|
Name(s) and complete address(es) of the manufacturer(s)
|
|
|
1.14.1
|
Name(s) and complete address(es) of the manufacturer(s) of the finished veterinary medicine, including the final company releasing the veterinary medicine if different from the manufacturer. (Add as many rows as necessary)
|
|
|
Name:Company name:Address: Country:Country code:Office telephone number:Mobile telephone number:E-Mail: If the manufacturer is different to 1.1 above, explain the relationship:
|
|
|
1.14.2
|
Name(s) and complete address(es) of the manufacturer(s) of the active ingredient(s) (AI)(Add as many rows as necessary)
|
|
|
|
|
|
Name:Company name:Address:Country:Country code Office Telephone number:Mobile number:E-Mail:
|
|
|
For VMD use only
|
|
|
1.15
|
Good Manufacturing Practice (GMP) status of the manufacturer(s) of the veterinary medicine
|
|
|
For VMD use only
|
|
|
1.16
|
Name and complete address of the Market Authorization holder of Manufacturer
|
|
|
Name:Company name:Address: Country:Country Code Office telephone number:Mobile number:E-Mail: If the Market Authorization holder is different to 1.1 above, explain and provide evidence for the relationship:
|
|
|
For VMD use only
|
|
|
1.17
|
Summary Veterinary Medicine Characteristics
|
|
|
For VMD use only
|
|
|
1.18 Batch number(s) of the veterinary medicine used in(Add as many rows as necessary)
|
|
|
Clinical/bioequivalence studies
|
|
|
|
Stability studies
|
|
|
|
|
|
Validation/production scale batches
|
|
|
|
|
|
Comments [e.g., batch size, explanation of NA (not applicable) answers]
|
|
|
Composition of clinical, primary stability and validation/production finished veterinary medicine batches (kg)
|
|
|
Ingredients
|
Administration Unit
|
Bioequivalence<batch number>
|
Primary stability<batch number>
|
Production<batch number>
|
|
|
Mg
|
%*
|
Kg
|
%*
|
Kg
|
%*
|
Kg
|
%*
|
|
|
Core tablet/bolus/capsule contents/injections/suspensions, etc. (Please delete/change which does not apply)
|
|
|
AI 1
|
|
|
|
|
|
|
|
|
|
|
AI 2
|
|
|
|
|
|
|
|
|
|
|
AI 3
|
|
|
|
|
|
|
|
|
|
|
Please add/delete as many rows as necessary
|
|
|
|
|
|
|
|
|
|
|
Excipient 1
|
|
|
|
|
|
|
|
|
|
|
Excipient 2
|
|
|
|
|
|
|
|
|
|
|
Excipient 3
|
|
|
|
|
|
|
|
|
|
|
Please add/delete as many rows as necessary
|
|
|
|
|
|
|
|
|
|
|
Subtotal 1
|
|
|
|
|
|
|
|
|
|
|
Purified water/other solvent(s)
|
|
|
|
|
|
|
|
|
|
|
Capsule shell/printing ink(Please delete/change which does not apply)
|
|
|
Please add/delete as many rows as necessary
|
|
|
|
|
|
|
|
|
|
|
Subtotal 2
|
|
|
|
|
|
|
|
|
|
|
Grand total
|
|
|
|
|
|
|
|
|
|
|
Equivalence of the composition or justified differences
|
The compositions of the bioequivalence, stability and validation batches are the same and differences are justified. (Please delete/change which does not apply)
|
|
|
*Each ingredient is expressed as a percentage of the grand total.**All components (..........) of the proprietary mixture are described in the compendiaFor VMD use onlyOVERALL QUERIES AND RECOMMENDATIONS FOR THIS PART
|
|
|
PART 2: CHEMICAL, PHARMACEUTICAL, NON-CLINICAL AND CLINICAL OVERVIEWS AND SUMMARIES
|
|
|
2.1
|
OVERALL TABLE OF CONTENTS OF PARTS 2, 3, 4 AND 5
|
|
|
2.2
|
INTRODUCTION
|
|
|
2.3
|
OVERALL QUALITY SUMMARY
|
|
|
For VMD use only
|
|
|
2.3.1
|
OVERVIEW OF ACTIVE INGREDIENT(S) [AI(S)]
|
|
|
2.3.1.1
|
General Information of the AI(S)
|
|
|
2.2.1.1.1
|
Nomenclature
|
|
|
|
|
|
For VMD use only
|
|
|
2.2.1.1.2
|
Structure
|
|
|
|
|
|
For VMD use only
|
|
|
2.2.1.1.3
|
General Properties of the AI(s)
|
|
|
For VMD use only
|
|
|
2.3.1.2
|
Manufacture of the AI(S)
|
|
|
2.3.1.2.1
|
Name and address of AI(s) Manufacturer
|
|
|
For VMD use only
|
|
|
2.3.1.2.2
|
Description of Manufacturing Process and Process Controls
|
|
|
2.3.1.2.3
|
Control of Materials used in Manufacture of AI
|
|
|
2.3.1.2.4
|
Controls of Critical Steps and Intermediates
|
|
|
2.2.1.2.5
|
Process Validation and/or Evaluation
|
|
|
For VMD use only
|
|
|
2.3.1.3
|
Characterization of the AI(S)
|
|
|
2.3.1.4
|
Control of the AI(S))
|
|
|
2.3.1.5
|
Reference Standards or Materials of the AI(S)
|
|
|
2.3.1.6
|
Container Closure System of the AI(S)
|
|
|
2.3.1.7
|
Stability of the AI(S)
|
|
|
For VMD use only
|
|
|
2.3.2
|
APPENDICES
|
|
|
2.3.2.1
|
Facilities and Equipment
|
|
|
2.3.2.2
|
Adventitious Agents Safety Evaluation
|
|
|
2.3.2.3
|
Novel Excipients
|
|
|
|
|
|
For VMD use only
|
|
|
2.4
|
SUMMARY OF NON-CLINICAL DOCUMENTATION AND CLINICAL DOCUMENTATION
|
|
|
2.4.1
|
FOR NEW CHEMICAL ENTITIES
|
|
|
2.4.1.1
|
Non-clinical overview
|
|
|
2.4.1.2
|
Non-clinical written and tabulated summaries
|
|
|
2.4.1.3
|
Clinical overview
|
|
|
2.4.1.3
|
Clinical summary
|
|
|
For VMD use only
|
|
|
2.4.2
|
GENERIC VETERINARY MEDICINE APPLICATIONS
|
|
|
2.4.2.1
|
Clinical Overview and Summary
|
|
|
2.4.2.1.1
|
Veterinary Medicine Development Rationale
|
|
|
2.4.2.1.2
|
Overview of Biopharmaceutics Studies
|
|
|
2.4.2.1.3
|
Summary of Biopharmaceutics Studies and Associated Analytical Methods
|
|
|
2.4.2.1.4
|
Overview and Summary of In Vitro Dissolution Tests complementary to Bioequivalence Studies
|
|
|
2.4.2.1.5
|
Overview and Summary of In Vitro Dissolution Tests in support of a Biowaiver
|
|
|
For VMD use only
|
|
|
For VMD use onlyOVERALL QUERIES AND RECOMMENDATIONS FOR THIS PART
|
|
|
PART 3: CHEMICAL-PHARMACEUTICAL DOCUMENTATION
|
|
|
3.1
|
TABLE OF CONTENTS OF PART 3
|
|
|
3.2
|
BODY OF DATA
|
|
|
3.2.1
|
PARTICULARS OF ACTIVE INGREDIENT(s) [AI(s)]
|
|
|
3.2.1.1
|
General Information of the AI(S)
|
|
|
3.2.1.2
|
Manufacture of the AI(S)
|
|
|
3.2.1.3
|
Characterization of the AI(S)
|
|
|
3.2.1.4
|
Control of the AI(S))
|
|
|
3.2.1.5
|
Reference Standards or Materials of the AI(S)
|
|
|
3.2.1.6
|
Container Closure System of the AI(S)
|
|
|
3.2.1.7
|
Stability of the AI(S)
|
|
|
3.2.2
|
PARTICULARS OF FINISHED VETERINARY MEDICINE
|
|
|
3.2.2.1
|
Description and Composition of the Veterinary Medicine(s)
|
|
|
3.2.2.2
|
Pharmaceutical Development of the Veterinary Medicine(s)
|
|
|
3.2.2.3
|
Manufacture of the Veterinary Medicine(s)
|
|
|
3.2.2.4
|
Control of Excipients for the Veterinary Medicine(s)
|
|
|
3.2.2.5
|
Control of the Veterinary Medicine(s)
|
|
|
3.2.2.6
|
Reference Standards or Materials of the Veterinary Medicine(s)
|
|
|
3.2.2.7
|
Container Closure System of the Veterinary Medicine(s)
|
|
|
3.2.2.8
|
Stability of the Veterinary Medicine(s)
|
|
|
3.2.3
|
APPENDICES
|
|
|
3.2.3.1
|
Facilities and Equipment
|
|
|
3.2.3.2
|
Adventitious Agents Safety Evaluation
|
|
|
3.2.3.3
|
Novel Excipients
|
|
|
PART4: NON-CLINICAL STUDY REPORTS FOR NEW CHEMICAL ENTITIES ONLY
|
|
|
4.1
|
TABLE OF CONTENTS OF PART 4
|
|
|
4.2
|
STUDY REPORTS
|
|
|
4.3
|
LITERATURE REFERENCES
|
|
|
PARTS: CLINICAL STUDY REPORTS
|
|
|
5.1
|
NEW CHEMICAL ENTITIES ONLY
|
|
|
5.1.1
|
Table of Contents of Part 5
|
|
|
5.1.2
|
Tabular Listing of All Clinical Studies
|
|
|
5.1.3
|
Clinical Study Reports
|
|
|
5.1.4
|
Literature References
|
|
|
5.2
|
INTERCHANGEABILITY OF GENERIC VETERINARY MEDICINE — (GENERIC VETERINARY MEDICINE APPLICATIONS ONLY)
|
|
|
5.2.1
|
REPORTS OF BIOPHARMACEUTIC STUDY(IES)
|
|
|
5.2.1.1
|
Bioavailability (BA) study report
|
|
|
5.2.1.2
|
In Vitro Dissolution Tests
|
|
|
5.2.2.1.1
|
In vitro dissolution tests complementary to bioequivalence studies
|
|
|
5.2.2.1.2
|
In vitro dissolution tests in support of biowaiver
|
|
|
5.2.3
|
Other Clinical study data done to support efficacy and safety of the product
|
|
|
5.3
|
SAFETY AND RESIDUES DOCUMENTATION (FOR VETERINARY MEDICINES USED IN FOOD ANIMALS)
|
|
|
5.3.1
|
Requirements for Animal Safety
|
|
|
5.3.1.1
|
Laboratory Animal Studies
|
|
|
5.3.1.2
|
Target Animal Safety Studies
|
|
|
5.3.2
|
Requirements for Human Safety
|
|
|
5.3.2.1
|
Laboratory Animal Toxicity Studies
|
|
|
5.3.2.2
|
Microbiological Safety Studies (for antimicrobial products)
|
|
|
DECLARATION BY AN APPLICANT
|
|
|
|
1. I, the undersigned certify that all the information in this application form and accompanying documentation is correct, complete and true to the best of my knowledge.2. I further confirm that the information referred to in my application dossier is available for verification during current GMP inspection.3. I agree that the undersigned has not marketed or advertised this product in Kenya and will follow the Veterinary Medicines Directorate requirements for advertisements of veterinary medicines4. I also agree that I am obliged to follow the requirements of the Veterinary Medicines Directorate Regulations.Name: .............................Position in the company: ..................Signature: ........................Date: ..............................Official stamp: ..........................
|
|
|
FORM B
|
APPLICATION FOR THE REGISTRATION OF VETERINARY PESTICIDES
|
(r. 23(b))
|
|
|
(to be submitted as one original hard-copy and one electronic copy (in MS-Word)
|
|
[to be submitted as one original hard-copy and one electronic copy (in MS-Word)]
The Director
Veterinary Medicines Directorate
KABETE
Information for Applicants
| 1. |
The application form must be completed by a duly authorized person.
|
| 2. |
Every application must be accompanied by 3 copies of the draft label as per VMD requirements.
|
| 3. |
The applicant may be required to submit:—
| (a) |
a sample of the veterinary pesticide;
|
| (b) |
a sample of the technical grade of its active ingredient;
|
| (c) |
a sample of the laboratory standard of its active ingredient;
|
| (d) |
any other sample as may be required by the Council.
|
|
| 4. |
The application must be accompanied by a technical dossier as per VMD data Requirements.
|
| 5. |
An applicant who is not a resident in Kenya shall appoint as an agent a person who is permanently resident in Kenya and duly recognized by the Veterinary Medicines Directorate.
PURPOSE OF APPLICATION (tick as appropriate)
|
a. Veterinary pesticide containing a new active ingredient ☐
|
|
b. Veterinary pesticide where source of active and/or formulation is not identical to that of a registered product ☐
|
|
|
|
c. Registration transfer ☐
|
|
d. Amendments to existing registration ☐
|
|
e. Other (Explain) ...........................................................
|
|
Will the product be marketed under own label? If no specify Yes ☐ No ☐
|
|
Proposed date of marketing .............................................
|
|
1. APPLICANT
|
|
1.1 Identification
|
|
|
Name of applicant / Corporate name of company
|
|
|
Business Reg No.:
|
|
|
Name of registration holder
|
|
|
Name of local agent in country: (if different from registration holder)
|
|
|
1.2 Status: (Importer/formulator/distributor)
|
|
|
Country of origin and country code
|
|
|
1.3 Physical Address
|
|
|
1.4 Postal Address:
|
|
|
1.5 Office Telephone (and code):
|
|
|
1.6 Mobile:
|
|
|
1.7 e-Mail:
|
|
|
1.8 Status:
|
|
|
1.9 (Importer/formulator/distributor)
|
|
|
Business Registration No. of Importer/formulator/distributor (if different from applicant):
|
|
|
1.2 Identification
|
|
|
Name of applicant / Corporate name of company
|
|
|
Business Reg No.:
|
|
|
Name of registration holder
|
|
|
Name of local agent in country: (if different from registration holder)
|
|
|
1,2.1 Status: (Importer/formulator/distributor)
|
|
|
1.2.2 Name:
|
|
|
1.2.2 Country of origin and area code
|
|
|
1.2.3 Physical Address
|
|
|
1.2.4 Postal Address:
|
|
|
1.2.5 Office Telephone (and code):
|
|
|
1.2.6 Mobile (and code):
|
|
|
1.3 e-Mail:
|
|
|
Veterinary Pesticide details
|
|
2.1 Designation (Description of product)
|
Trade name:
|
|
Trade mark:
|
|
Trade mark holder:
|
|
2.2. Function of veterinary pesticide: (eg. Insecticide, acaricides etc.)
|
|
|
2.3 Intended use:.
|
|
|
2.4 Target pest(s), vector(s) and host(s)
|
|
|
2.5 Method, dosage rates and frequency of application:
|
|
|
2.6 Type of formulation: (eg. Emulsifiable Concentrate. Wetable powder, etc.)
|
|
|
2.7(a) Is the veterinary pesticide registered in country of manufacture?b) Is the veterinary pesticide registered in the country of formulation?
|
Yes ☐ No ☐If no, give reasons Yes☐ NoIf no, give No reasons
|
|
2.8 Registration in SEARCH* country/ies: (names)
|
|
|
2.9 Existing registration No(s) and country(s).
|
|
|
2.10 Customs Tariff Code:
|
|
|
3. COMPOSITION OF ACTIVE INGREDIENT(S) (Technical grade) (Information on active ingredient(s) (A.I) may be attached in sealed envelope)
|
|
Active ingredient(s): (Common name/s)
|
Manufacturer: (Name and address)
|
Minimum A.I.% purity
|
MaximumA.I.% purity
|
|
|
|
|
|
|
4. FORMULATION
|
|
|
4.1 Formulator: (Name)Country and code:Postal Address: Physical address:Office TelephoneMobile:Email address:
|
|
4.2 Internal code:
|
|
|
4.3 Composition (Information on composition may be attached in sealed envelope)
|
|
|
Ingredients and Function:
|
g1
|
g/kg
|
Range
|
|
|
|
* Formerly GCPF
* SEARCH - Southern and Eastern African Regulation Committee on Harmonisation of Pesticide Registration
|
5. TOXICOLOGY (formulated veterinary pesticide)
|
|
5.1 Rat:
|
Acute Oral (LDso. mg/kg)
|
Acute Dermal (LDso Ingficg)
|
Inhalation **LCso (mg/I/hour)
|
|
|
Experimental
|
Experimental
|
Experimental
|
|
|
Calculated
|
Calculated
|
Calculated
|
|
5.2 Rabbit:
|
Skin irritation
|
Eye irritation
|
|
None
|
|
|
|
|
Mild
|
|
|
|
|
Moderate
|
|
|
|
|
Severe
|
|
|
|
|
5.3 Skin Sensitization in guinea pig: (tick)
|
None ☐ Mild ☐ Moderate ☐ Severe ☐
|
|
5.4 WHO classification:
|
Ia
|
Ib
|
II
|
III
|
|
a Summary of other mammalian toxicological studies with references: eg. livestock, wildlife, poultry, pets
|
|
5.6 Summary of environmental effects
|
|
5.6.1 Toxicity to bees:
|
|
|
5.6.2 Toxicity to fish and other aquatic organisms:
|
|
|
5.6.3 Toxicity to birds:
|
|
|
5.6.4 Toxicity to earthworms and soil micro-organisms:
|
|
|
5.6.5 Toxicity to other non-target organisms:
|
|
|
5.6.6 Persistence in environment:
|
|
|
5.6.7 Other effects: Specify
|
|
|
6. PACKAGING
|
|
|
6.1 Packaging material or container:
|
|
|
6.2 Pack size(s):
|
|
|
6.3 Method of disposal of empty container(s):
|
|
|
7. OTHER SPECIFIC REQUIREMENTS
|
|
7.1 Operator exposure
|
|
a). Dermal absorption.
|
|
b). Likely operator exposure under field conditions
|
|
c). Available toxicological data relating to other ingredients in formulation (non-active additives in formulation).
|
|
8. DECLARATION
|
|
For and on behalf of ........................ I ................................. hereby certify that the above mentioned information and data provided in support of this application is to the best of my knowledge true, correct and complete.
|
|
.........................................Name in full (printed)
|
.....................Signature
|
|
....................................Official Title
|
.................................Date
|
|
Official Stamp of Applicant / Company
|
|
Remarks..........................................................................Signed: ............. Date: ...................
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
NOTE: The format of this application is recognized by all SEARCH countries.
*LD50=medium lethal dose
**LC50—medium Lethal Concentrate
GUIDELINE: ACTIVE INGREDIENT DOSSIER (OFFICIAL INFORMATION ONLY)
The dossier accompanying this form should provide details of the information requested on the methods used (physical and chemical), details of the methods used in and results of toxicological and ecotoxicological studies, methods of analysis etc. Numbering used in the dossier must correspond with that used in the application form.
ACTIVE INGREDIENT
|
REQUIREMENTS:
|
REMARKS
|
|
1. DESIGNATION/IDENTITY OF A.I.
|
Specify accordingly.
|
|
1.1 Common name
|
|
|
1.2 Manufacturer or Development code
|
|
|
1.3 Source, Name and Address of manufacturer and address and location of manufacturing plants.
|
|
|
1.4 Methods of manufacture (synthesis pathways)
|
|
|
1.5 Chemical name (IUPAC)
|
|
|
1.6 Chemical group
|
|
|
1.7 Structural formula
|
|
|
1.8 Empirical formula
|
|
|
1.9 Patent status
|
|
|
Is the A.I. under patent?
|
|
|
Who is patent holder
|
|
|
Expiry date
|
|
|
1.10 Molecular mass
|
|
|
1.11 CAS Number
|
|
|
| 2. |
PHYSICAL AND CHEMICAL PROPERTIES
(active ingredient)
|
REQUIREMENTS:
|
REMARKS
|
|
2.1. Physical state
|
Where relevant indicate method/test used.
|
|
2.2 Colour
|
|
|
2.3 Odour
|
|
|
2.4 Density at 20°C
|
|
|
2.5 Vapour pressure at 20/25°C
|
|
|
2.6 Volatility
|
|
|
2.7 Hydrolysis DT50 ..... Days °C pH
|
Give the DT50 of the active ingredient, with mention of temperature and pH parameters employed during the determination.
|
|
2.8 Photolysis
|
Give the DT50 of the active ingredient (in days).
|
|
2.9 Solubility in water °C ..... pH
|
Where relevant indicate method/test used.
|
|
2.10 Solubility in organic solvents
|
|
|
2.11 n-octanol/water partition coefficient
|
|
|
2.12 Boiling point °C
|
|
|
2.13 Melting point °C
|
|
|
2.14 Decomposition temperature °C
|
|
|
2.15 Method of Analysis and Impurities
|
|
|
2.16 Stability in water, hydrolysis rate, effect of light, identity of breakdown products
|
|
|
2.17 Stability in organic solvents used in Formulation
|
|
|
2.18 Stability in air; effect of light, identity of breakdown products
|
|
|
REQUIREMENTS:
|
REMARKS:
|
|
2.19 Thermal stabilty, identity of breakdown product.
|
|
|
2.20 Flammability
|
|
|
2.21 Flash point
|
|
|
2.22 Explosive properties
|
|
|
2.23 Oxidizing properties
|
|
|
2.24 Absorption spectra — UV/Visible, infra-red, NMR, MS
|
|
|
2.25 Reactivity towards container material
|
|
|
| 3. |
TOXICOLOGY
(Active Ingredient)
Include a copy of an executive summary discussing ALL ISSUES named under Part 3 or provide copies of the individual summaries from each study relating to issues mentioned under Part 3. Information on the methods of testing must be provided.
|
REQUIREMENTS:
|
REMARKS
|
|
ADI
|
Acceptable Daily Intake in mg product/kg
|
|
|
body weight.
|
|
NOAEL
|
Non observable adverse effect level (NOAEL) (expressed in mg product / kg weight on animal)
|
|
Short term toxicity
|
|
Oral cumulative toxicity (28 days study)
|
Not mandatory, but can be useful
|
|
Subchronic toxicity test of 90-day duration.
|
Oral route on two species — one rodent(rat) and one non-rodent.
|
|
Dermal route — 28-days dermal, 90-days dermal.
|
Specify accordingly.
|
|
Inhalation route 28-days inhalation, 90-days inhalation
|
Specify accordingly.
|
|
3.1 Eye irritation (rabbit)
|
|
|
3.2 Skin sensitization (guinea pig)
|
|
|
3.3 Reproduction (specify species)
|
|
|
3.4 Subchronic toxicity 90 day NOAEL mg/kg/day
|
|
|
3.5 Chronic toxicity NOAEL mg/kg/day
|
|
|
3.6 Carcinogenicity (life time) NOAEL mg/kg/day
|
|
|
3.7 Neurotoxicity NOAEL mg/kg/day
|
|
|
3.8 Teratogenicity NOAEL mg/kg/day
|
|
|
3.9 Mutagenicity /Genotoxicity
|
|
|
3.10 Metabolism (rat)
|
|
|
3.11 Other studies
|
Provide further information relevant to the toxicity profile of the product e.g. Toxicity of major metabolites, reaction or breakdown products of the pest control products formed in/or on treated plant/crop etc, which are likely to be consumed — in cases where different from those identified in animal studies. Toxic effects on livestock, poultry, pests etc. should be given.
|
|
| 4. |
ECO-TOXICOLOGY
Provide either an executive summary or individual summaries of studies on the behaviour of the veterinary pesticide in the environment. Provide information requested for in the application form.
|
REQUIREMENTS:
|
REMARKS:
|
|
4.1 Birds (2 species)
|
LD50 mg/kg
|
Provide details of at least one land and one water bird. LD50 in mg product/kg bird weight and the NOAEL. Furthermoreprovide information on the effect on reproduction
|
|
NOAEL
|
|
LDsomg/kg
|
|
NOAEL
|
|
Reproduction
|
|
4.2 Fish (2 species)
|
LD50 mg/kg
|
Provide details on at least two species studied. LC50 (in mg of product / litre of water) and the NOAEL. Furthermore provide information on the effect on reproduction. Indicate the bio-concentration factor (BCF) on the active ingredient in tissues.
|
|
NOAEL
|
|
LD5omg/kg
|
|
Reproduction
|
|
BCF
|
|
4.3 Daphnia
|
LC50 mg/1
|
Specify and provide details on other organisms according to the information requested on the form.
|
|
NOAEL
|
|
4.4 Algae
|
LC50mg/1
|
|
NOAEL
|
|
4.5 Bees
|
LD50.1g/bee
|
|
NOAEL
|
|
4.6 Earthworms
|
LC50 mg/kg
|
|
4.7 Soil microorganisms
|
|
|
| 5. |
BEHAVIOUR IN ENVIRONMENT
(active ingredient)
Provide an executive summary or copies of summaries from each study relating to the issues highlighted in the application form.
|
REQUIREMENTS:
|
REMARKS:
|
|
5.1 Behaviour, ways of degradation. degradation products in soil:
|
Indicate the degradation path of the active ingredient in the soil and the degradation products formed.
|
|
5.11 Major metabolites
|
Specify the major metabolites in the soil and their behaviour.
|
|
5.12 DT 50 (days)
|
Specify the half-life of the active ingredient in various types of soils.
|
|
5.13 Mobility of the A.I.
|
Specify the degree of mobility of the active ingredient in the soil hence leaching potential and possibility for ground water contamination. If high, provide details on further studies i.e. lysimeter study.
|
|
5.14 Adsorption
|
Indicate the degree of adsorption of the active ingredient in the soil.
|
|
5.15 Mobility of metabolites
|
Indicate the degree of mobility of the metabolites in the soil.
|
|
5.2 Behaviour, ways of degradation, degradation products in water:
|
Describe ways and speed of degradation of the active ingredient in water.
|
|
5.21 Major Metabolites
|
Specify the major break down products formed and their adsorption/desorption on sediments.
|
|
5.22 DT50 (days)
|
Specify the half-life of the active ingredient in water
|
|
5.23. Surface
|
Describe ways and speed of degradation in surface and ground water.Provide an executive summary or copies of summaries from each study relating to the issues highlighted in the form
|
|
5.24 Ground
|
|
5.3 Behaviour, ways of degradation, degradation products in air:
|
Describe ways and speed of degradation in air and break down products formed. (for fumigants and volatile products).
|
|
| 7. |
RESIDUES
Provide either an executive summary or individual summaries of studies conducted concerning the issues listed in the application form.
|
REQUIREMENTS:
|
REMARKS:
|
|
7.1 Major metabolites
|
Provide either an executive summary or individual summaries of studies conducted concerning the metabolites in plants.- Specify the metabolites- State their toxicological effects
|
|
7.2 Metabolism
|
Describe the principle of metabolization of the active ingredient in the plant and the degradation products formed.
|
|
7.3 Behaviour of residues
|
Indicate the action and the persistence of the metabolites in the animals.
|
|
|
|
|
7.5 MRL codex
|
MRL's (if available)
|
|
7.5.1 MRL of country of origin
|
|
7.5.2 Proposed MRL
|
|
|
|
7.9 Method of residue analysis
|
Provide a copy in the dossier for countries requiring it.
|
|
| 8. |
OTHER SPECIFIC REQUIREMENTS
|
REQUIREMENTS:
|
REMARKS:
|
|
8.1 Residue data from a GLP certified lab or as directed by the Directorate.
|
Provide an executive summary or copies of summaries from each study relating to residues.
|
|
8.2 Proposed withholding periods after use.
|
|
8.3 Effects on taint, odour, taste or other quality aspects due to residues in or on fresh or processed products.
|
|
8.4 Effects of industrial processing and/or household preparation on the nature and magnitude of residues.
|
|
SUMMARY OF THE DATA SUBMITTED TO THE VMD FOR REGISTRATION OF A VETERINARY PESTICIDES
Part I —
Trade Name ................................................
The Name and Address of Formulator ..................................
Common Name of the active ingredient(s) ..................................
Concentration of active ingredient(s) ..................................
Source of active ingredient(s) ..................................
Chemical Name ..........................................
Formulation type ..........................
Proposed Uses ..........................
Packaging/Containers (Material size) ..........................
Registrant (Name, Address, Status) ..........................
Agents/Distributors in Kenya ..........................
Premises (Reg. No. Date of issue) ..........................
Part II - CHEMISTRY DATA
a) Physical /Chemical Properties of the A.I .........................
................................................................
b) Physical/Chemical properties of the active ingredient (AI) ..........................
....................................................
c) Composition of the veterinary pesticide (purity%, nature and content of impurities, isomers, by-products — other details should be provided in the dossier)
....................................................
d) Physical/Chemical Properties of the Formulated veterinary pesticide
................................................
e) Composition of the Formulated veterinary pesticide (Concentration of A.I. in the formulation, other details should be provided in the dossier)
..........................................................
f) Method of analysis for determination of the A.I. in technical and formulated veterinary pesticide
..............................................................
Part III - Biological (efficacy) Data
a) Target Pest(s), Vectors, Diseases(s), Host(s) ..............................
b) Method, Rate, Frequency of application ..............................
c) Recommendations for use in Kenya ..............................
d) Recommendations for use by authorized bodies outside Kenya ..............................
Part IV - Toxicological data
a) Acute Toxicological Data of the active ingredient(s)
......................................................
b) Acute toxicity data of the formulated veterinary pesticide: ...........................
c) Short term toxicity studies ...........................
d) Other toxicological studies: ...........................
1) Reproduction studies ...................................
2) Teratological studies ...................................
3) Neurotoxicity studies ...................................
4) Mutagenecity studies ...................................
5) Long term toxicity/carcinogenicity studies ...........................
6) Accumulation of compound in tissues .............................
7) Metabolic studies ..........................................................
8) Effects on animals ..........................................................
9) Toxicity Data on impurities .............................
10) Toxicity Data on metabolites .............................
11) Human toxicology and medical aspects:
1) Hazards to humans ........................................
2) Symptoms of poisoning ........................................
3) Antidote ........................................
4) First Aid Measures ........................................
5) Treatment ...................................................
6) Safety Precautions/Restrictions ........................................
Part V - RESIDUE DATA
a) Principal Residues ..............................................................................
b) Disappearance and fate of residues .......................................
c) Method(s) of analysis (soil, water, feedstuffs etc.)
..............................................................................
Part VI
Environment and wildlife hazards
a) Degradation and mobility studies (soil, water, air) ..................................
b)Toxicity to birds ..................................
c)Toxicity to fish ..................................
d)Toxicity to honeybees/beneficial insects ..................................
e)Toxicity to earthworms, other soil invertebrates ..................................
f) Changes in soil ecology ..................................
Part VII
Information on Approvals/Registrations in other countries .....................
..............................................................................
..............................................................................
Part VIII
Draft of local label (paste
......................................................................
......................................................................
Part IX
Brief prepared by ..........................................................
Signature ..........................................................
Official stamp ..........................................................
Date ..........................................................
Part X
Decision of the VMD technical committee
Recommended/Not Recommended for registration
Reasons:
..........................................................................
Date: ..........................................................................
|
FORM C. 1
|
(r. 27 (4))
|
|
REGISTER FOR VETERINARY MEDICINES
|
|
S/No
|
Trade Name
|
Active Ingredient(s)
|
Reg. No
|
Class/ Category
|
Intended Use
|
Dosage Form
|
Manufacturer & Contact
|
Date Reg./Date Retained
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
FORM C. 2
|
(r. 27 (4))
|
|
REGISTER FOR VETERINARY PESTICIDES
|
|
S/No.
|
Trade Name
|
Active Ingredient(s)
|
Reg. No
|
Class
|
Category
|
Function
|
Intended Use
|
Dosage Form
|
Manufacturer & Contact
|
Date Reg.
|
Date Retained
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
FORM D. 1
|
(r. 23 (3))
|
|
REPUBLIC OF KENYA
|
|
THE VETERINARY MEDICINE DIRECTORATE
|
|
CERTIFICATE OF REGISTRATION OF VETERINARY MEDICINE (Valid for a maximum of 3 months)
|
|
Registration Number ..................................................
|
It is hereby certified that the veterinary medicine as described hereunder has been registered subject to the conditions indicated below:
1. Trade name under which marketed ...............................
2. Approved name (in pharmacopoeia) ..............................................................
3. Form of preparation ........................................................
4. Active ingredient(s) and strength(s) ...............................
5. Condition(s) under which veterinary medicine is registered
..............................................................
Name and business address of manufacturer ...............................
Registered in the name of ..........................................
Business address ...................................................
6. Date of registration ......................................
7. Expiry date of registration ...............................
|
Date ...................
|
|
|
Registrar of Veterinary MedicinesThe Veterinary Medicines Directorate
|
|
FORM D. 2
|
(r. 23(3))
|
|
REPUBLIC OF KENYA
|
|
THE VETERINARY MEDICINE DIRECTORATE
|
|
CERTIFICATE OF REGISTRATION OF VETERINARY MEDICINE
|
|
Registration Number ...........................................
|
It is hereby certified that the veterinary medicine as described hereunder has been registered subject to the conditions indicated below:
1. Trade name under which marketed ...............................
2. Approved name (in pharmacopoeia) ..............................................................
3. Form of preparation ........................................................
4. Active ingredient(s) and strength(s) ...............................
5. Condition(s) under which veterinary medicine is registered
..............................................................
6. Name and business address of manufacturer ...............................
7. Registered in the name of ..........................................
8. Business address ...................................................
9. Date of registration ......................................
10. Expiry date of registration ...............................
|
Date ...................
|
|
|
Registrar of Veterinary MedicinesThe Veterinary Medicines Directorate
|
|
FORM E
|
(r. 24)
|
|
APPLICATION FOR RENEWAL FOR RETENTION OF A VETERINARY MEDICINE/ PESTICIDE IN THE REGISTER
|
|
(to be submitted as one original hard-copy and one electronic copy in MS-Word)
|
|
The DirectorVeterinary Medicines DirectorateKABETE
|
|
Application number
|
|
|
Name of applicant
|
|
|
Address
|
|
|
Registration number of veterinary medicine/veterinary pesticide for retention
|
|
|
Declaration on GMP compliance
|
|
|
Declaration on change on physical address if applicable
|
|
|
Declaration of applicant that the registered veterinary medicine/ pesticide has not changed since registration/ previous retention
|
|
|
FOR OFFICIAL USE
|
|
|
Findings of pharmaco-vigilance
|
|
|
Recommendations of the committee
|
|
|
Approved/rejected (if rejected give reasons)
|
|
|
Name ......................................................................................
|
|
Signature ...................................................
|
Date ........................................................
|
|
CEO, VMD
|
|
|
FORM F
|
(r. 47(2)(a))
|
|
REPUBLIC OF KENYATHE VETERINARY MEDICINE DIRECTORATE VETERINARY MEDICINES PREMISES INSPECTION FORM
|
(to be filled in triplicate, one copy to be retained at the inspected premises)
I. the undersigned of (postal address) ............................... have today carried out an inspection of ......................... as required by Regulation 18 of the Veterinary Surgeons and Veterinary Para-Professionals (Veterinary Medicines Directorate) Regulations.
Identification of premises;
| (1) |
Name of owner/proprietor....................................
|
| (2) |
Physical location(specify) .................................
|
| (3) |
Address..............................................
|
| (4) |
Premise Permit No. .......................................
|
| (5) |
Authorized classes and categories of medicines dispensed.............................
The following findings are reported—
Location with respect to fire hazards.........................................
Separation from other veterinary operations..................................
Separation from non-complementary businesses.................................
Restriction of access to Category I and II veterinary medicine by personnel............................
Vermin and insect proofing...................................................
Security and safety measures for veterinary medicine.........................................
Storage conditions..........................................................
Descriptions of floors and the walls of the building...............................
Description of safety cabinets for medicines.................................
Personnel protection equipment used in premises...............................
Description of size and space for operations.........................................
Description of disposal system for expired veterinary medicines...................................
Competency of staff.........................................................
Identification of hazard areas.............................................
Labeling of sections.......................................................
Labeling of veterinary medicines designated areas............................
Emergency lighting, firefighting equipment and first aid kit(s)...........................
Emergency protocols displayed..........................................
Standard operating procedures displayed.......................................
Records of movement of all veterinary medicines..............................
Sanitary facilities ........................................
7. Other comments
Summary of significant observations ..............................................
Inspection carried out in presence of:
Name.......................... ID. No .................... Position in the business..................... Signature...................... Date ................................
I have the following recommendations to make—
.............................................
The previous inspection was carried out on ................................................
Signature................................................................
Designation..................................................................
Date ...........................................................
For official use
Premise approved/rejected
Action taken............................................
Signature ................................ Date.........................
The Chief Executive Officer, Veterinary
Medicines Directorate
|
FORM G
|
(r. 47(2)(b))
|
|
REPUBLIC OF KENYATHE VETERINARY MEDICINE DIRECTORATEIMPOUNDING OF SUSPECT VETERINARY MEDICINE FORM
|
(to be filled in triplicate, and one copy be retained at the inspected premises)
I ................... being a veterinary medicines inspector Registration number have .........................today (date) ..................... impounded the following veterinary medicines from located at ...................in .......................... county and registered under ..................................
|
No.
|
Veterinary medicine/pesticides
|
Category
|
Dosage form
|
Quantity
|
Reasons
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Impounding carried out in presence of:-
Name: ....................ID................................. Position in the business.........................Date: .........................................
Signature........................................
Date ............................................
|
FORM H.1
|
(r.38(2)
|
|
APPLICATION FOR A PREMISES PERMIT FOR A VETERINARY PHARMACY
|
The Chief Executive Officer,
Veterinary Medicines Directorate,
KABETE.
1. Applicant Details
Applicant's Name: ............................... Professional Reg. No...........................
Email address: ............................. Cell Phone No. ............................
ID/Passport/Alien ID No: ..................... Nationality..........................
Premise Name & Address.................................................
.........................................................
Qualification ...................................................
Period of experience working in a veterinary pharmacy ................years.
1. Premise Location:
County: ................................... Town: ...........................
Road: ................................... Building: ..............................
2. Proposed category of vet medicine that will be traded in:
3. Other professionals working in this premise
|
No.
|
Names
|
Position in the Business
|
Registration/enrollment no. new column qualification and experience
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Date .................... Signature of the Applicant.........................
Note: all fields are MANDATORY. Attach a copy of previous premise licence, if any, a copy of current registration with the professional regulator and the business registration details where applicable. Non-Kenyans to attach current work permit. Incomplete forms will not be processed.
|
FORM H.2
|
(r. 38(2))
|
|
APPLICATION FOR A VETERINARY WHOLESALE DEALERS PREMISES LICENCE
|
The Chief Executive Officer,
Veterinary Medicines Directorate
KABETE.
1. Applicant's details:
Applicants name............................ Professional Reg. No.................
Business Name:........................ Business Reg. No...................
Email address: ........................Cell Phone No............................
ID/Passport/Alien ID No:....................... Nationality....................
Premise Name & Address-....................................................
Premise Location:....................................................
County:....................................................................
Town: ...............................................................
Road: ...........................Building: .................................
2. Supervising Veterinary Surgeon
Name.......................... Professional Reg. No.......................
Email address:......................... Cell Phone No............................
ID/Passport/Alien ID No: .........................Nationality................
3. Other professionals working in this premise.
|
No.
|
Names
|
Position in the business
|
Qualification
|
Registration / enrollment no.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Signature of the Applicant ............................. Date ..........................
Note: all fields are MANDATORY. Attach a certified copy of certificate of registration/incorporation of the business and memorandum and articles of incorporation, previous premise licence, a copy of current registration with the professional regulator and the business registration details where applicable. Non-Kenyans to attach current work permit. Incomplete forms will not be processed.
|
FORM H.3
|
(r.38(2))
|
|
APPLICATION FORM FOR GOOD MANUFACTURING PRACTICE INSPECTIONFOR VETERINARY PHARMACEUTICAL MANUFACTURING FACILITIES
|
The Chief Executive Officer,
Veterinary Medicines Directorate
KABETE.
1. PARTICULARS OF APPLICANT/LICENCE HOLDER
Name...........................................
Physical Address
Country.............................. Telephone......................
Mobile......................... E-mail.....................
2. PARTICULARS OF SITE TO BE INSPECTED
Name of contact person ..............................
Physical Address (if different from 1. above).................................
Country ........................ Tel .................................
Mobile ......... .....E-mail:...............................
Note: Separate application to be filled in for each individual site
3. CONTACT PERSON ON SITE
Name of contact person.................................................
Tel: .................... Fax:...........................
E-mail:.............................................
4. AUTHORISED REPRESENTATIVE/AGENT IN KENYA
Name of Local Technical Representative
Tel: ______________________________
Mobile:___________________________
Email:___________________________________
5. TYPE OF VETERINARY MEDICINES
Class of veterinary medicines manufactured (Tick where applicable)
• Veterinary pharmaceuticals
• Biologicals
• Nutrients
• Equipment and materials
• Alternative medicines
• Poisons
6. (Please tick where applicable)
☐ First Inspection
☐ Re-inspection after failure
☐ Routine Re- inspection
☐ Previous inspection date
☐ Other (please specify) ................................................
7. LINES TO BE INSPECTED
|
DOSAGE FORM
|
Tick where applicable
|
*ACTIVITIES
|
|
Tablets
|
|
|
|
Capsules
|
|
|
|
Injections
|
|
|
|
Oral liquids
|
|
|
|
Creams/Ointments/lotions
|
|
|
|
Others (specify)
|
|
|
|
|
|
|
*Activity means any of the following:
• Formulation (dispensing, mixing, blending)
• Processing(compression, emulsification etc)
• Packing
• Quality Control
• Warehousing (raw material, finished products)
8. REGISTRATION OF PRODUCTS
Have you registered any products in Kenya; or
Have you submitted dossier for registration? YES ☐ NO ☐
If YES, list the veterinary medicines applicable. (Attach a separate sheet if needed) ............................................
I hereby certify that the above information is correct and apply for Good Manufacturing Practice inspection of the above-named site(s).
Signature of applicant...................... Date:............................
Print Name ..........................................
_________________________________________
|
FORM I.1
|
(r.38(4)(a))
|
|
REPUBLIC OF KENYATHE VETERINARY MEDICINE DIRECTORATEPREMISES PERMIT FOR A VETERINARY PHARMACY
|
SERIAL NO............................
Messrs...............................................
of..................................................
Plot No. ......................... is permitted to carry on business of veterinary pharmacy as provided in Regulation 22(4) of Veterinary Medicines Directorate Regulations.
This licence allows trade in veterinary medicines in category ..........................
Date ............................
Chief Executive Officer, Veterinary Medicines Directorate.
Note:
| (a) |
This registration expires on 31st December, 20..............
|
| (b) |
No change of premises is permitted without the authority of the Directorate.
|
| (c) |
This registration shall become void immediately upon any change of ownership of the
|
| (d) |
Directorate shall be notified immediately the licensee changes
|
________________________________________________
|
FORM I. 2
|
(r38 (4)(b))
|
|
REPUBLIC OF KENYATHE VETERINARY MEDICINE DIRECTORATEVETERINARY WHOLESALEDEALERS PREMISES PERMIT
|
SERIAL NO. ...........................
Messrs........................................
of ..........................................
Plot No. ................... is permitted to carry on business of veterinary medicines wholesaling as provided in Regulation 22(4) of Veterinary Medicines Directorate Regulations.
The business shall be under the supervision of ............................. KVB Reg. No. ....................(where applicable)
Date ...............................
Chief Executive Officer,
Veterinary Medicines Directorate.
NB:
| (a) |
This registration expires on 31st December, 20.................
|
| (b) |
No change of premises is permitted without the authority of the Directorate.
|
| (c) |
This registration shall become void immediately upon any change of the supervisor of the business and may resume practice when a new supervisor has been approved by the Council and brought on board.
|
| (d) |
Directorate must be notified immediately the licensee changes.
|
________________________________________-
|
FORM I 3
|
(r.38(4)(c))
|
|
REPUBLIC OF KENYATHE VETERINARY MEDICINE DIRECTORATEVETERINARY MEDICINES GOOD MANUFACTURING PRACTICE LICENCE
|
SERIAL NO. ................................
Messrs. .................................. of ................................Land Registration No. ......................... has complied with good manufacturing practices and is hereby licenced to carry out business of veterinary medicine manufacturing as provided in Regulation 8(2) of Veterinary Medicines Directorate Regulations.
Date ..........................
Chief Executive Officer,
Veterinary Medicines Directorate.
Note:
| (a) |
This licence expires on 31st December, 20.....................
|
| (b) |
No change of premises is permitted without the authority of the Directorate
|
_____________________________________________
|
FORM J
|
(r. 37(2))
|
|
REGISTER OF APPROVED VETERINARY PHARMACY PREMISES
|
|
S/No of Permit.
|
Type of Veterinary Pharmacy
|
Physical Location
|
Name of Business
|
Reg. No. of Business
|
Name & Contact of Proprietor
|
Name, Contact & Qualification of Professional In Charge
|
Date of Permit
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
FORM K
|
(r. 44(1))
|
|
APPLICATION FOR IMPORT/ EXPORT PERMIT FOR A VETERINARY MEDICINE
|
Application No. ...................... Date......................
I. Name of Importer/Exporter .....................................
Address .................Tel. No .................. Business Location ...........................
Land Reg.NO. ............. Street/Road .............. Town ............
PIN NO. ................ VAT NO. .................................
Commodity................... Value (FOB) Kshs.....................
Quantity............... Date of Manufacture.............. Expiry Date....................
Purpose of Importation/Exportation........................................
Country of Origin ......................Destination............................
Last Imports/Exports Quantity.................. Value Kshs...................
Date ........................................
N.B. Part Ito be completed by the applicant. Misleading information in Part I may lead to invalidation of the application and or prosecution.
II. EVALUATION BY VETERINARY MEDICINES DIRECTORATE (VMD)
| (a) |
The Directorate has evaluated the product ................... and confirms that it is registered/not registered for .........................under Reg. No. .............................
|
OR for Experimental/ Raw materials(delete as appropriate)
| (b) |
recommended/ not recommended for importation/exportation.
|
Reasons:
1.
2.
3.
Evaluating Officer
Name: .............................................................
Signature: ........................................................
CHIEF EXECUTIVE OFFICER: Approved/Not Approved
Name............................................................
Signed ...................... Date.........................
Valid for Three Months, from the date of approval, for one consignment
__________________________________
|
FORM L
|
(r.44(2))
|
|
REPUBLIC OF KENYA
|
|
|
|
THE VETERINARY MEDICINE DIRECTORATE
|
|
PERMIT FOR IMPORTATION/ EXPORTATION OF A VETERINARY MEDICINE
|
CHIEF EXECUTIVE OFFICER
VETERINARY MEDICINES DIRECTORATE
KABETE
Permit No. ....................................................
Application No. ....................................................
This permit is granted to ....................................................
To import/export a veterinary medicine(s) ....................................................
Trade name ....................................................
Registration number ....................................................
Date of expiry of registration ....................................................
Registered uses ....................................................
Country of origin (if being imported) ....................................................
Country of destination (if being exported) ....................................................
Harmonisation Code (HS) Code ....................................................
Approved common name ....................................................
Chemical name ....................................................
Formulation type ....................................................
Concentration (A.I. %) .................................................
State of veterinary medicine (Tick appropriately) A.I. ........................or Formulated.......................
Category of veterinary medicine ....................................................
Purpose of import/export ....................................................
Registered use ..................................................................
Quantity authorized for importation or exportation ......................................
Date .....................................
..........................................
Chief Executive Officer
Name ......................................................................
Signature ................................................................
Stamp and Seal ...............................................................
Fee paid................ Date ....................Receipt No.....................
This permit is not transferable to any other person without the approval of the Directorate.
NB This permit is valid for one consignment only, for three (3) months from the date of issue.
_______________________________________________
|
FOURTH SCHEDULE [r. 17]
CODE OF CONDUCT FOR MEMBERS OF AND EMPLOYEES OF THE DIRECTORATE
1. Impartiality and independence of members
Every member and employee of the Directorate shall impartially and independently perform the functions of a member in good faith and without fear, favour or prejudice, and without influence from—
| (a) |
the National or County Government;
|
| (d) |
any candidate participating in an election; or
|
| (e) |
any other person or authority.
|
2. Independence from political or public office
| (1) |
A member or employee of the Directorate shall not, during tenure of office, be eligible for—
| (a) |
appointment or nomination to a political office; or
|
| (b) |
appointment to another public office.
|
|
| (2) |
A member of the Directorate may not—
| (a) |
by their membership, association, statement, conduct or in any other manner place in jeopardy the perceived independence of the member, or in any other manner harm the credibility, impartiality, independence or integrity of the Directorate;
|
| (b) |
make private use of or profit from any confidential information gained as a result of being a member of the Directorate; or
|
| (c) |
divulge any information to any third party, save in the of official duty.
|
3. Disclosure of conflicting interests
|
| (1) |
If a member or an employee is directly or indirectly interested in any contract, proposed contract or other matter before the Council and is present at any meeting of the Council at which the contract, proposed contract or other matter is the subject of consideration, the member or employee shall, at the meeting and as soon as practicable after the commencement thereof, disclose the fact and shall not take part in the consideration or discussion of, or vote on, any questions with respect to the contract or other matter or be counted in the quorum of the meeting during consideration of the matter.
|
| (2) |
A member or employee whose personal interest conflicts with their official duties shall—
| (a) |
declare the personal interests to their supervisor or other appropriate person or body in writing and comply with any directions given to avoid the conflict; and
|
| (b) |
refrain from participating in any deliberations with respect to the matter.
|
|
| (3) |
No member of staff of the Directorate shall transact business with the Directorate directly or indirectly.
4. Professionalism
A member or employee of the Directorate shall—
| (a) |
perform their duties in a manner that promotes and maintains public confidence in the Directorate;
|
| (b) |
treat the public and colleagues with courtesy and respect;
|
| (c) |
discharge all their duties in a professional, timely and efficient manner and in line with the rule of law; and
|
| (d) |
respect the rights and freedom of all persons that he may interact with.
|
5. Improper enrichment
A member or employee of the Directorate shall not—
| (a) |
use their office or organization to improperly enrich themselves or others;
|
| (b) |
accept or request gifts or personal favours from any person who may have a commercial interest with the Directorate or any other interest that may be affected by the normal business of the Service; or
|
| (c) |
use information that is acquired during the course of their duties or connected to their duties for their benefit or for the benefit of others
|
6. Integrity in private affairs
A member or employee shall conduct their private affairs in manner that maintains public confidence in the integrity of their office and the Directorate as a whole and shall—
| (b) |
not neglect their financial obligations;
|
| (c) |
submit an annual declaration of their income, assets and liabilities to the Commission responsible for such declarations from public officers;
|
| (d) |
not engage in political activity that may compromise or be seen to compromise the neutrality of their office, or the Directorate; and
|
| (e) |
not preside over or play a central role in the organization of a fundraising activity.
|
|
| (1) |
A member or employee shall not sexually harass a member of the public or colleague.
|
| (2) |
Sexual harassment includes—
| (a) |
making a request or exerting pressure for sexual activity or favours;
|
| (b) |
making intentional or careless physical contact that is sexual in nature; or
|
| (c) |
making gestures, jokes or comments, including innuendoes regarding another person's sexuality.
|
8. Nepotism
A member or employee shall not practice favouritism on the grounds of tribe, race, kin, culture, sex or acquaintance or otherwise in performance of their duties.
9. Application of the Public Officers Ethics Act
This Code is in addition to the provisions of the Public Officers Ethics Act and where there is a conflict between the Code and these Regulations, the provisions of the Act shall prevail.
10. Breach of code
Any breach of the Code by a member or officer of the Service shall be treated as gross misconduct.
|
FIFTH SCHEDULE [r. 24(3), 38(5) & 61]
FEES
|
Purpose
|
Fees
|
Frequency
|
|
Application forms under these regulations shall be issued free of charge
|
|
Inspection fee:(i) Retail Veterinary Pharmacy
|
Kshs 15,000
|
Once
|
|
Inspection fee:(ii) Wholesale Veterinary Pharmacy
|
Kshs 30,000
|
Once
|
|
Good Manufacturing Practice inspection per site
|
|
|
|
1) Local manufacturing site.
|
Kshs 100,000
|
Every three (3) years
|
|
2) Foreign manufacturing site
|
USD 4,000
|
Every three (3) years
|
|
Product registration fees per imported veterinary medicine
|
USD 1,000
|
Once
|
|
Product registration fees for locally manufactured veterinary medicine
|
USD 500
|
Once
|
|
Appeals for rejected application for registration of veterinary medicine
|
USD 300
|
|
|
Retention of veterinary medicine in the register
|
USD 300
|
Annually
|
|
Veterinary pharmacy practice fee:
|
|
|
|
1) Wholesaler
|
Kshs 30,000
|
Annual
|
|
2) Retailer
|
Kshs 10,000
|
Annual
|
|
Manufacturer fee
|
Kshs 30,000
|
|
|
Import permit
|
Kshs 1,000
|
Per consignment
|
|
Inspection/verification fee
|
0.75% of the consignment value
|
Per FOB consignment
|
|
Advertisement per veterinary medicine
|
Kshs50,000
|
Annual
|
|
Fees for duplicate permit or licence
|
Kshs 1,000
|
Per copy
|
THE VETERINARY SURGEONS AND VETERINARY PARA-PROFESSIONALS (VETERINARY MEDICINES DIRECTORATE) REGULATIONS
PART I – PRELIMINARY
| 1. |
Citation
These Regulations may be cited as the Veterinary Surgeons and Veterinary Paraprofessionals (Veterinary Medicines Directorate) Regulations, 2015.
|
| 2. |
Interpretation
In these Regulations, unless the context otherwise requires—
"accredited laboratory" means a laboratory recognized as an accredited laboratory by the Directorate;
"advertisement" means any written or visual notice, circular, label, or wrapper, or other descriptive matter, verbal statement or reference appearing in any newspaper, television, film or mass media or brought to the attention of the public in any other form, which is intended to promote the sale of a veterinary medicine;
"alternative medicine" means the unrefined plant, animal and mineral substances commonly used in traditional animal treatments;
"Cabinet Secretary" means the Cabinet Secretary responsible for matters relating to veterinary service;
"Chief Executive Officer" means the Chief Executive Officer of the Directorate appointed under regulation 13;
"Committee" means the Veterinary Medicines Registration Committee established under regulation 27;
"controlled veterinary medicine" means a veterinary medicine specified in the Fourth Schedule as a Category I or Category II of veterinary medicine;
"Conventional medicines" means the regular and standardized veterinary medicines;
"Council" means the Council of the Directorate comprised under regulation 8 (1);
"crude drug" means an unrefined medicine of biological origin;
"Directorate" means the Veterinary Medicines Directorate established under regulation 5 and as envisaged under section 39(2)(a) of the Act;
"dispense" means the sale or supply of a veterinary medicine by a veterinary surgeon or other person authorised in accordance with these Regulations;
"inspector" means a person appointed as an inspector under regulation 45;
"manufacture" means any stage in the manufacturing of a veterinary medicine until the finished product is ready for sale in its final form as specified in the marketing authorization, and includes repackaging, repacking or labeling of a veterinary medicine in an authorized facility but does not include the breaking open of the package of a veterinary medicine by retailers;
"market authorization" means registration of a veterinary medicine by the Council and the issuance of a registration certificate under regulation 23;
"orphan veterinary medicine" means a veterinary medicine that is not economical to trade in but is required for specific medical use;
"pharmaco-vigilance" means the routine inspections and surveys carried out in the veterinary medicines market to safeguard general animal and human health and trade;
"quality assurance standards" means the good manufacturing practice, good laboratory practice, good veterinary practice or any other standard developed by an international standardization body, the East African Community Standards Committee or the Kenya Bureau of Standards which the Cabinet Secretary, on the advice of the Council, shall recognize through the Gazette as a quality assurance standard for the purposes of these Regulations;
"register" means a register maintained by the Council containing the details of—
| (a) |
premises which have been issued with a permit in accordance with these Regulations; or
|
| (b) |
veterinary medicines registered in the country;
|
"retailer" means a veterinary pharmacy registered by the Council for the sale of veterinary medicines to the end users;
"specialized committees" means persons who are not Council members;
"veterinary medicines inspector" means an inspector appointed by the Council pursuant to regulation 45;
"veterinary pesticide" means a veterinary medicine used as a pest control product on animals or the animals' environment;
"veterinary pharmaceutical" means a chemical substance formulated or compounded as a single active ingredient or in any combination of the chemical substances, for veterinary curative use;
"veterinary pharmacy" means a business authorized by the Council to stock, dispense or distribute veterinary medicines for sale;
"veterinary pharmacy assistant" means a veterinary para-professional trading in veterinary medicines listed under Category III of the Fourth Schedule;
"veterinary pharmacy practice" means the business of veterinary pharmacy carried out by veterinary surgeons, veterinary para-professionals or any other person permitted by the Council to carry out veterinary pharmacy practice;
"veterinary product" means veterinary medicines and veterinary pesticides;
"wholesale" means a manufacturer or veterinary pharmacy approved by the Council to trade in bulk in the supply of veterinary medicines to wholesalers or retailers in the original outer pack-sizes.
|
| 3. |
Objectives and purpose of the Regulations
The object and purpose of these Regulations is to—
| (a) |
regulate the manufacture, importation, exportation, registration, distribution, prescription and dispensing of veterinary medicines and the practice of veterinary pharmacy in Kenya; and
|
| (b) |
advise the Kenya Veterinary Board in relation to all aspects listed under paragraph (a).
|
|
| 4. |
Application
Subject to regulation 58, these Regulations shall apply to all conventional and alternate veterinary medicine.
|
PART II – THE VETERINARY MEDICINES DIRECTORATE
| 5. |
Establishment of the Directorate
| (1) |
There is hereby established a Directorate to be known as the Veterinary Medicines Directorate whose management shall vest on a Council appointed under regulation 8.
|
| (2) |
The Directorate shall be a body corporate with perpetual succession and a common seal and shall in its corporate name be capable of—
| (b) |
taking, purchasing, or otherwise acquiring, holding, charging or disposing of movable and immovable property;
|
| (c) |
borrowing and lending money; and
|
| (d) |
doing all such other things or acts as may lawfully be done by a body corporate.
|
|
|
| 6. |
Functions of the Directorate
The functions of the Directorate are to —
| (a) |
formulate and enforce quality assurance standards in the in the manufacture, distribution and use of veterinary medicines to safeguard human and animal health and the environment;
|
| (b) |
in consultation with the Director of Veterinary Services, regulate the use of veterinary medicine for the treatment of animals under the Animal Diseases Act (Cap. 364);
|
| (c) |
consider applications for approval for market authorization of veterinary medicines;
|
| (d) |
set quality assurance standards for training in the management of veterinary medicines as directed by the Kenya Veterinary Board;
|
| (e) |
collaborate with the Kenya Veterinary Board in regulating training in the management of veterinary medicines;
|
| (f) |
inspect and approve premises in which the manufacture, sale or supply of veterinary medicine is conducted;
|
| (g) |
appoint and gazette veterinary medicine inspectors;
|
| (h) |
establish the standard operating procedures for veterinary medicine inspectors;
|
| (i) |
regulate veterinary pharmacy practices;
|
| (j) |
categorize veterinary medicines and the qualification of persons authorized to trade in each category and review the categories every five years;
|
| (k) |
regulate clinical and non-clinical trials of veterinary medicines by individuals and institutions to be involved in the trials;
|
| (l) |
regulate the manufacture, importation, exportation, handling, advertisement, labeling, sale and disposal of veterinary medicines;
|
| (m) |
register all veterinary medicines manufactured or imported for use in the country or exported from the country;
|
| (n) |
monitor the market for and take measures necessary for the elimination of trade in illegal and counterfeit veterinary medicines;
|
| (o) |
establish systems of pharmaco-vigilance and conduct pharmaco-vigilance of veterinary medicines through regular inspections and surveys;
|
| (p) |
enforce good manufacturing practice for veterinary medicines as approved by the Council;
|
| (q) |
develop, apply and from time to time review guidelines to be used in the inspection and ensuring compliance with good manufacturing practice;
|
| (r) |
ensure that the promotion and marketing of veterinary medicine is in accordance with the approved product information;
|
| (s) |
publish, on an annual basis, a notice in the Kenya Gazette inviting the public to note and inspect the register of veterinary medicines and the list of approved veterinary pharmacy practices within such period and at such place as may be specified in the notice;
|
| (t) |
consider, grant, issue or revoke authorizations and certificates in accordance with these Regulations;
|
| (u) |
collaborate with other regulatory agencies including the Public Health (Standards) Board in section 27 of the Food, Drugs and Chemical Substances Act (Cap. 254), the Pest Control Products Board established under section 5 of the Pest Control Products Act (Cap. 346) and the Central Board of Health established under section 3 of the Public Health Act (Cap. 242) to carry out its mandate; and
|
| (v) |
undertake any other thing necessary for the effective carrying out of its mandate under this or any other Act.
|
|
| 7. |
Powers of the Council
The Council shall have all the power necessary or expedient for the effective discharge of its functions under these Regulations and any other law, and in particular the power to—
| (a) |
control, supervise and manage the assets and liabilities of the Directorate;
|
| (b) |
determine the provision to be made for capital and recurrent expenditure and for the reserves of the Directorate;
|
| (c) |
seek and receive any grants or donations and make legitimate disbursements from such grants and donations for its purposes;
|
| (d) |
levy fees and charges for its services as provided in these Regulations;
|
| (e) |
enter into association with other bodies within or outside Kenya which the Council may consider desirable or appropriate;
|
| (f) |
invest funds of the Directorate not immediately required in securities in which trustees are empowered to invest under the Trustee Act (Cap. 167), and in other securities which may be approved for the purpose, by the Cabinet Secretary for the time being responsible for Finance;
|
| (g) |
establish and support investment and trust funds for the benefit of employees or ex-employees of the Directorate or dependants of such persons, to grant pension, benefits and allowances and to make such payments towards insurance as required under the relevant laws;
|
| (h) |
open and operate such accounts as are necessary for the funds of the Directorate, with a bank or financial institution licensed to conduct banking business under the Banking Act (Cap. 488);
|
| (i) |
recruit, support, discipline or dismiss the staff and inspectors of the Directorate;
|
| (j) |
in consultation with Salaries Remuneration Commission, determine the terms and conditions of employment of the staff and inspectors of the Directorate;
|
| (k) |
establish branch offices of the Directorate, to the extent that is practicable, to ensure accessibility of its services by all Kenyans;
|
| (l) |
superintend, regulate and assist branch offices, auxiliaries, committees and other forms of organizations established to advance the interest of the Directorate;
|
| (m) |
perform all things necessary or incidental to attain the objectives of the these Regulations or any other written law.
|
|
| 8. |
Composition and appointment of the Council
| (1) |
The Council shall be appointed by the Cabinet Secretary and shall consist of—
| (a) |
a chairperson, who shall be a registered veterinary surgeon with at least ten years’ post-qualification experience, appointed by the President;
|
| (b) |
the Director of Veterinary Services;
|
| (c) |
the Chief Executive Officer of the Kenya Veterinary Board;
|
| (d) |
the Principal Secretary for the time being responsible for Finance;
|
| (e) |
the Principal Secretary for the time being responsible for animal health matters;
|
| (f) |
two veterinary surgeons nominated by the Kenya Veterinary Board;
|
| (g) |
one veterinary technologist from the veterinary pharmaceutical industry nominated by the Kenya Veterinary Board;
|
| (ga) |
one livestock sector stakeholder who is not a veterinary surgeon or a veterinary para-professional, appointed by the Cabinet Secretary; and
|
| (h) |
the Chief Executive Officer of the Directorate who shall be the Secretary to the Council and shall be an ex-officio member.
|
|
| (2) |
A person appointed under paragraph (1)(f) shall be nominated by the Kenya Veterinary Board from a list of five names drawn from the veterinary pharmaceutical industry including a trainer in veterinary pharmacology submitted by a registered professional association representing the interests of veterinary surgeons countrywide.
|
| (3) |
A person appointed under paragraph (1)(g) shall be nominated by the Kenya Veterinary Board from a list of three names from the veterinary pharmaceutical industry submitted by the registered association representing the interests of veterinary paraprofessionals.
|
| (4) |
The nominating bodies referred to under paragraphs (2) and (3) shall observe Constitutional principles relating to gender, youth, persons with disability and minorities in identifying the persons whose names shall be submitted for nomination by the Kenya Veterinary Board for appointment to the Council.
|
| (5) |
The Cabinet Secretary shall appoint the members of the Council by notice in the Kenya Gazette.
|
| (6) |
The Council shall conduct its affairs in the manner set out in the First Schedule.
|
| (7) |
The members of the Council shall hold office for a term of three years and shall be eligible for re-appointment for one further term.
|
| (8) |
The members appointed under paragraph (1)(b), (d) and (e) may, in writing, designate representatives to attend the meetings of the Council on their behalf.
|
| (9) |
The members of the Council in paragraphs (f), (g) and (h) may—
| (a) |
at any time resign from office by notice in writing to the Cabinet Secretary;
|
| (b) |
be removed from office by the Cabinet Secretary, on the advice of the Council if the member —
| (i) |
is declared bankrupt; |
| (ii) |
is absent from three consecutive meetings of the Council, without the permission of the Chairperson; |
| (iii) |
is convicted of a criminal offence and, sentenced to a term of imprisonment of more than six months; |
| (iv) |
is unable or unfit, due to physical or mental illness, to perform the functions of his office; or |
| (v) |
has failed to comply with the provisions of Chapter Six of the Constitution. |
|
|
| (10) |
The Cabinet Secretary shall, on the recommendation of the Council, appoint a relevant person to serve in the place of any member of the Council in the case of death, resignation, absence from Kenya for six consecutive months without authorization or more or removal from office under paragraph (9)(b) and the person appointed under this sub-Regulation shall serve until the end of the term of the Council.
|
| (11) |
Where a vacancy arises under paragraph (10), the Cabinet Secretary shall undertake a fresh recruitment process to fill the vacancy.
[L.N. 113/2023, r. 2.]
|
|
| 9. |
Co-option of specialized persons
Pursuant to section 10 of the Act, the Council may for the effective discharge of its functions co-opt into the committees such persons with technical expertise or knowledge for the better carrying out of the functions of the Directorate.
|
| 10. |
Meetings of the Council
The conduct and regulation of the business and affairs of the Council shall be as provided in the First Schedule but subject thereto, the Council shall regulate its own procedure.
|
| 11. |
Conflict of interest
| (1) |
If any person is present at a meeting of the Council or any committee at which any matter is the subject of consideration and in which matter that person or that person's spouse or relative is directly or indirectly interested in a private capacity, that person shall as soon as is practicable after the commencement of the meeting declare such interest and shall not, unless the Council or committee otherwise directs, take part in any consideration or discussion of, or vote on any question connected to such matter.
|
| (2) |
The disclosure of interest shall be recorded in the minutes of the meeting at which it is made.
|
| (3) |
A member or employee of the Council shall not carry out an business or trade with the Council.
|
| (4) |
A member or staff of the Council who contravenes this Regulation commits an offence and is liable, on conviction, to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months or to both.
|
|
| 12. |
Common seal and logo
| (1) |
The Directorate shall have a common seal which shall be kept by the Chief Executive Officer.
|
| (2) |
The affixing of the seal shall be authenticated by signature of the chairperson and the Chief Executive Officer and in his absence, the signature of any other member authorized by resolution of the Council.
|
|
| 13. |
Chief Executive Officer
| (1) |
There shall be a Chief Executive Officer who shall be appointed by the Council through a competitive process and whose terms and conditions of service shall be determined by the Council in the instrument of appointment.
|
| (2) |
A person shall be qualified for appointment as a Chief Executive officer, if that person—
| (b) |
holds a degree in veterinary medicine from a university recognized by the Kenya Veterinary Board;
|
| (c) |
has at least ten years professional experience in veterinary medicine regulation, of which at least five years are at senior management level; and
|
| (d) |
satisfies the requirements of Chapter Six of the Constitution.
|
|
| (3) |
The Chief Executive Officer shall be an ex-officio member of the Council but shall have no right to vote at any meeting of the Council.
|
| (4) |
The Chief Executive Officer shall hold office for a term of three years renewable once subject to satisfactory performance.
|
| (4A) |
Despite subregulation (4), a person serving as a Chief Executive Officer immediately before the commencement of this Regulation shall remain in office for the unexpired period of that person’s tenure.
|
| (5) |
The Chief Executive Officers shall, in the performance of the functions and duties of office be responsible to the Council.
|
| (6) |
Without prejudice to the provisions of paragraph (5), the Chief Executive Officer shall—
| (a) |
| (i) |
carrying into effect the decisions of the Council; |
| (ii) |
day-to-day administration and management of the affairs of the Directorate; |
| (iii) |
supervision of the staff of the Directorate; |
|
| (b) |
be the Registrar of veterinary medicines; and
|
| (c) |
perform such other duties as may be assigned by the Council.
|
[L.N. 113 of 2023, r. 3.]
|
|
| 14. |
Removal of the Chief Executive Officer
| (1) |
The Chief Executive Officer may be removed from office by the Council in accordance with the terms and condition of service, for—
| (a) |
inability to perform functions of the office arising out of physical or mental infirmity;
|
| (b) |
gross misconduct or misbehavior;
|
| (c) |
incompetence or negligence of duty;
|
| (d) |
violation of the Constitution and any other written law; or
|
| (e) |
any other grounds specified in the terms and conditions of service.
|
|
| (2) |
Where the question of the removal of the Chief Executive Officer under paragraph (1) arises, the Council shall —
| (a) |
inform the Chief Executive Officer in writing of the reasons for the intended removal; and
|
| (b) |
provide the Chief Executive Officer with the opportunity to be heard in accordance with the principles of fair administrative action safeguarded under Article 47 of the Constitution.
|
|
|
| 15. |
Veterinary medicines inspectors
| (1) |
The Council shall employ such veterinary medicines inspectors as it deems necessary.
|
| (2) |
A person shall be qualified for appointment as a veterinary medicines inspector if that person —
| (a) |
is a veterinary surgeon or technologist; or
|
| (b) |
holds at least a diploma in animal health or other qualification recognized by the Council; and
|
| (c) |
has at least five years post qualification experience in a relevant field.
|
|
|
| 16. |
Staff
The Council may employ such staff as it deems appropriate for the effective performance of its functions.
|
| 17. |
Code of conduct
The members and the employees of the Council shall subscribe to the leadership and integrity code set out in the Fourth Schedule.
|
| 18. |
Protection from personal liability
Nothing done by a member of the Council or by any person working under the instructions of the Council shall, if done in good faith for the purpose of executing the powers, functions or duties of the Council under these Regulations render such member or officer personally liable for any action, claim or demand.
|
PART III – MANUFACTURING, IMPORTATION AND REGISTRATION OF VETERINARY MEDICINES
| 19. |
Veterinary medicines to be registered
| (1) |
A person shall not import, manufacture sell, transport or distribute any veterinary medicine in Kenya unless that veterinary medicine has been registered in accordance with the provisions of these Regulations.
|
| (2) |
Every person transporting a veterinary medicine in transit shall declare the veterinary medicine at the port of entry and exit.
|
|
| 20. |
Manufacturing permit
A person who intends to manufacture veterinary medicine shall make an application to the Council for a manufacturing permit.
|
| 21. |
Manufacturing of veterinary medicines
| (1) |
The Council shall, on receipt of an application made under regulation 20, before issuing a certificate of registration, cause the premises in which the manufacturing of the veterinary medicine is proposed to be conducted be inspected in order to ensure compliance to good manufacturing practice.
|
| (2) |
The inspector instructed under paragraph (1) shall submit the results of the inspection to the Council and the applicant in writing.
|
| (3) |
A manufacturer shall ensure that every stage in the manufacture of a veterinary medicine is carried out based on good manufacturing practices.
|
| (4) |
A manufacturer shall not manufacture a veterinary medicine unless the manufacturing plant has been inspected and licensed in accordance with this Regulation.
|
| (5) |
A person shall not use any premises, for the purposes of manufacturing. formulating, packaging, selling or storing veterinary medicine unless that person is in possession of a licence issued under these Regulations in respect of that premises.
|
| (6) |
Where an inspector has approved the compliance by the manufacturer, the Council shall on payment of the prescribed fee, issue a licence as set out in Form 1.3 set out in the a Third Schedule.
|
| (7) |
A person in executive authority of manufacturing company which manufactures veterinary medicines without a license commits an offence and shall be liable, on conviction, to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months or to both.
|
|
| 22. |
Powers of inspectors with regard manufacturing plants
| (1) |
An inspector appointed under these Regulations shall have power with regard to manufacturing plants to —
| (a) |
enter the premises and inspect the plant and the process of manufacture intended to be employed in the manufacturing of the veterinary medicine and shall make a report to the Council; or
|
| (b) |
order the immediate closure of a manufacturing plant where its continued operation appears to pose a serious threat to life and safety.
|
|
| (2) |
An inspector who has ordered the closure of a plant under paragraph (1)(b) shall within twenty-four hours of the closure notify the Council of the closure and shall provide reasons for the closure.
|
| (3) |
On receipt of the notification under paragraph (2), the Council shall direct the owner of the plant to take such measures as the Council may direct to ensure conformity with these Regulations.
|
| (4) |
A manufacturer who fails, within fourteen days of receipt of the directive made by the Council under paragraph (3), to take the measures indicated in the directive shall have its licence revoked.
|
| (5) |
The Council shall publish in at least one newspaper of wide national circulation the name of a manufacturing plant closed and whose licence has been revoked under this Regulation and shall place a notice of the closure at the entrance of such plant.
|
| (6) |
The protection of right to property under Article 40 of the Constitution shall be limited for purposes of maintenance of veterinary medicine standards for the protection and safety of the public.
|
|
| 23. |
Application for registration of a veterinary medicine
| (1) |
An application for registration of —
| (a) |
a veterinary medicine shall be in accordance with Form A set out in the Third Schedule; and
|
| (b) |
a veterinary medicine used as pesticides shall be Form B set out in the Third Schedule.
|
|
| (2) |
An applicant shall, in addition to the information required to be paragraph (1), furnish such further information and material as may be required by the Council for the proper evaluation of the veterinary medicine in respect of which the application is made.
|
| (3) |
The Council shall, on approval of an application made under this Regulation and on payment of the prescribed fees, register the veterinary medicine or veterinary pesticide and issue a certificate in Form D1 or D2, respectively set out in the Third Schedule.
|
| (4) |
A certificate of registration issued under these Regulations shall, unless suspended or revoked, be in force for a period of five years.
|
| (5) |
The Council shall not approve an application made under paragraph (1) unless satisfied that the applicant has attained the prescribed standards and satisfied all requirements for registration.
|
|
| 24. |
Renewal of registration of veterinary medicine
| (1) |
A person may make an application to the Council, for renewal of registration of a veterinary medicine in Form E set out in the Third Schedule.
|
| (2) |
The Council shall, before renewing registration under this Regulation conduct a pharmaco-vigilance study on the manufacturer.
|
| (3) |
An applicant shall on receipt of approval for renewal of registration from the Council, make payment of the fees as prescribed in the Fifth Schedule.
|
|
| 25. |
Clinical and non-clinical trials and toxicity testing
| (1) |
The Council may require that a clinical and non-clinical trial and toxicity testing be done before the registration of a veterinary medicine.
|
| (2) |
Despite paragraph (1) a veterinary pesticide shall not be registered unless an efficacy and an acute toxicological study is carried out by an accredited laboratory.
|
| (3) |
A person who or institution which intends to apply for accreditation under this Regulation shall, before making such application, obtain approval from the Council.
|
| (4) |
A person who or institution which desires to conduct clinical and non-clinical trials and toxicity testing shall apply to the Council for accreditation.
|
| (5) |
The cost of any clinical trial or toxicity testing shall be borne by the applicant.
|
|
| 26. |
Conditions for registration of veterinary medicines and pesticides
| (1) |
A veterinary medicine and pesticide shall not be registered unless a laboratory analysis has been carried out by an accredited laboratory.
|
| (2) |
A person who intends to register a veterinary medicine shall submit to the Council a certificate of analysis from an accredited laboratory together with the application for registration of the veterinary medicine
|
|
| 27. |
Veterinary Medicines Registration Committee
| (1) |
There is hereby established a standing committee of the Council to be known as the Veterinary Medicines Registration Committee.
|
| (2) |
The Committee shall be responsible for matters relating to registration of veterinary medicines under these Regulations and in particular shall be responsible for —
| (a) |
evaluating applications for veterinary medicines registration and shall make recommendations to the Council; and
|
| (b) |
issuing provisional approval pending the issuance of the veterinary medicines registration certificate by the Council.
|
|
| (3) |
A provisional certificate issued under paragraph (2)(b) shall be valid for a period of not more than three months.
|
| (4) |
The Council shall consider an application made under this Regulation, and if satisfied of the safety, efficacy and quality of the veterinary medicine or veterinary pesticide, shall register the veterinary medicine in accordance with Form C1 and C2 set out in the Third Schedule.
|
| (5) |
Upon registration of a veterinary medicine under paragraph (4), the applicant shall be issued with a certificate of registration in Form D1 or D2 set out in the Third Schedule.
|
| (6) |
The Council may, while considering a veterinary medicine for registration under paragraph (4), approve the details as supplied by the applicant or approve it with such modifications as it may consider appropriate in respect of the following particulars—
| (a) |
the name under which the veterinary medicine may be sold;
|
| (c) |
the statement of the representations to be made for the promotion of the veterinary medicine in respect of the —
| (i) |
claim to be made for the veterinary medicine; |
| (ii) |
route of administration; |
| (iv) |
contra-indications, side effects and precautions, if any; |
| (vi) |
withdrawal period for food producing animals; |
| (vii) |
proposed category; and |
|
|
|
| 28. |
Rejection of application for registration of a veterinary medicine
The Council shall, if it is not satisfied as to the safety, efficacy or quality of the veterinary medicine, reject the application for the registration of the veterinary medicine and inform the applicant, in writing, the reasons for rejection.
|
| 29. |
Registration of veterinary medicine researched outside Kenya
| (1) |
The Council may, before registering a new veterinary medicine for which the research work has been conducted in another country and its efficacy, safety, and quality established in that country, require an investigation on the pharmaceutical, pharmacological and other aspects of the veterinary medicine to be conducted and clinical trials to be carried out as necessary to establish its safety, efficacy, quality and where applicable the biological availability to be established under local conditions.
|
| (2) |
Despite paragraph (1), the Council may register a new veterinary medicine and require the investigations and clinical trials specified therein to be conducted after its registration.
|
|
| 30. |
Suspension and revocation of certificate of registration
| (1) |
The Council may suspend or revoke a certificate of registration issued under these Regulations for such period as the Council may determine.
|
| (2) |
The Council shall not revoke or cancel the certificate of registration unless —
| (a) |
where matters stated in the application on which the certificate of registration was granted were false or incomplete in any material particular;
|
| (b) |
where provision of the certificate of registration has to a material extent been contravened by the holder of the certificate;
|
| (c) |
where the premises on which, or on part of which, a veterinary medicine is manufactured, assembled or stored by or on behalf of the holder of the certificate of registration are unsuitable for the manufacturing, assembling or storage of veterinary medicines; or
|
| (d) |
where new information has been discovered by the Council which renders the veterinary medicines unsafe or dangerous.
|
|
| (3) |
Where a certificate has been revoked under paragraph (2)(d), the Council shall on delivery of the information to the manufacturer —
| (a) |
inspect the plant to ensure all the medicine or the relevant batch has been destroyed;
|
| (b) |
disseminate the information to the public through a newspaper advertisement in at least two daily newspapers of nationwide circulation; and
|
| (c) |
recall any medicine which has been distributed.
|
|
|
| 31. |
Period for renewal of registration and retention of veterinary medicine
| (1) |
The Council may grant the renewal of registration as provided under regulation 24 for a period not exceeding five years.
|
| (2) |
On the expiry of five years a veterinary medicine may be retained in the register upon an application made by the certificate holder which shall —
| (a) |
include a declaration that the requirements met by the veterinary medicine during registration has not changed; and
|
| (b) |
include payment of the requisite fees.
|
|
|
PART IV – CLASSIFICATION AND CATEGORIZATION OF VETERINARY MEDICINES
| 32. |
Classification of Veterinary Medicines
| (1) |
The Cabinet Secretary shall classify veterinary medicines in the classes set out in Part I of the Second Schedule.
|
| (2) |
The Cabinet Secretary may, on the advice of the Council, vary the classification of the various veterinary medicines depending on the therapeutic use and current information for the medicine.
|
|
| 33. |
Categorization of Veterinary Medicines
| (1) |
The Cabinet Secretary shall categorize veterinary medicines as set out in Part II of the Second Schedule.
|
| (2) |
The Cabinet Secretary shall be guided by the Council on the categorization of veterinary medicines and shall place the veterinary medicines in the following categories —
| (a) |
Category I relating to prescription only medicine—Veterinary Surgeon which shall comprise two sub-categories as follows —
| (i) |
Category IA relating to Controlled Veterinary Medicines includes products which contain narcotic or psychotropic substances or other substances very dangerous at small quantities; |
| (ii) |
Category IB relating to other prescription only medicine, comprising products intended for administration following a diagnosis or clinical assessment by a veterinary surgeon or dispensed by a veterinary surgeon or a person with equivalent qualification; |
|
| (b) |
Category II relating to prescription only medicine veterinary surgeon, pharmacist, and veterinary technologist who has served for at least five years in a veterinary pharmacy which contains —
| (i) |
veterinary medicines for use in food-producing animals; |
| (ii) |
veterinary medicines in respect of which special precautions shall be taken in order to avoid any unnecessary risk to the target species, the person administering the products to the animal and the environment; Provided that the requirement in this paragraph shall not apply if all the following criteria are met— |
|
| (c) |
Category III relating to authorised veterinary medicine general sales list which may be traded by veterinary surgeons and all categories of veterinary paraprofessionals and includes—
| (i) |
pest control veterinary medicines provided that the medicine are sold in the original package without repackaging and with an intact label; |
| (ii) |
endoparasiticide veterinary medicines considered safe for use in food animals; |
| (iii) |
endoparasiticide veterinary medicines for use in non-food animals; or |
| (iv) |
any other product the Directorate may prescribe. |
|
| (d) |
Category IV which includes alternative veterinary medicine general sales list.
|
|
|
| 34. |
Use of veterinary pesticides
| (1) |
A veterinary pesticide shall be—
| (a) |
sold only in the original sealed and labeled package as registered by the Council;
|
| (b) |
stored in accordance with the instructions on the label on the original container; and
|
| (c) |
used according to the instructions on the label.
|
|
| (2) |
The empty container of a veterinary pesticide shall be disposed in accordance with the instructions given on the label.
|
| (3) |
A person who removes or defaces a label on a veterinary pesticide or package commits an offence.
|
| (4) |
A person who sells, dispenses or otherwise gives out a veterinary pesticide in contravention of this Regulation commits an offence and shall be liable, on conviction, to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months or to both.
|
|
PART V – VETERINARY PHARMACY
| 35. |
Standards of Veterinary Pharmacy
| (1) |
A veterinary pharmacy shall satisfy the following minimum standards—
| (a) |
be located away from known fire hazards;
|
| (b) |
be a separate entity from other veterinary operations such as veterinary clinic, ambulatory and laboratory services;
|
| (c) |
be separated from non-complementary businesses;
|
| (d) |
have restricted access by personnel to Category I and II veterinary medicine; and
|
| (e) |
be vermin-proofed, including protection against insects and rodents.
|
|
| (2) |
A veterinary pharmacy shall—
| (a) |
demonstrate adequate security for the safety of veterinary medicine;
|
| (b) |
demonstrate appropriate storage conditions that shall maintain the temperature, lighting and humidity requirements as prescribed in the manufacturers' specifications of veterinary medicines;
|
| (c) |
have the floor and the wall of the building constructed from materials that are easy to clean, impervious and resistant to corrosion by chemicals;
|
| (d) |
have lockable safety cabinets to protect the controlled veterinary medicines;
|
| (e) |
provide the personnel with protective clothing for use only within the premises;
|
| (f) |
be of appropriate size and have sufficient space for carrying out of all necessary operations provided for the orderly receipt, warehousing and dispatch of the various classes of veterinary medicines and, in particular, a quarantine area for isolation when necessary, including isolation of faulty packs and recalled or expired veterinary products;
|
| (g) |
provide a disposal system, acceptable to the Council, for safe disposal of expired veterinary medicines;
|
| (h) |
be operated by competent staff as provided for in the permit;
|
| (i) |
display warning notices indicating hazardous areas;
|
| (j) |
label the various sections in the business;
|
| (k) |
store veterinary medicines only in the designated areas, which shall be labeled;
|
| (l) |
have emergency lighting, fire-fighting equipment and fully equipped first aid kit;
|
| (m) |
display emergency protocols within the veterinary pharmacy advising personnel on procedures to follow in case of emergencies;
|
| (n) |
display standard operating procedures;
|
| (o) |
retain records of the movement of all veterinary medicines for at least five years; and
|
| (p) |
provide separate sanitary facilities for each gender.
|
|
|
| 36. |
Qualifications to operate a veterinary pharmacy
A person who is a veterinary surgeon, or holds an equivalent qualification in matters of pertaining to veterinary medicines as determined by the Council, may apply to the Council for a permit to undertake a veterinary pharmacy.
|
| 37. |
Council to approve veterinary pharmacy businesses
| (1) |
The Council shall have power to approve any of the following types of veterinary pharmacy businesses—
| (a) |
manufacturing of veterinary medicines;
|
| (b) |
wholesaling of veterinary medicines; and
|
| (c) |
retailing of veterinary medicines.
|
|
| (2) |
Upon approval of the veterinary business specified under paragraph (1), the Council shall register the veterinary pharmacy premises in Form J set out in the Third Schedule.
|
|
| 38. |
Practicing permit
| (1) |
A person shall not practice the business of veterinary pharmacy unless he holds a valid practicing permit issued by the Council.
|
| (2) |
A person who intends to practice the business of veterinary pharmacy shall make an application in Forms H1, H2 and H2, respectively of the Third Schedule.
|
| (3) |
The Council shall issue a practicing permit if the premises in which the applicant intends to operate has been approved by the Council in accordance with these Regulations.
|
| (4) |
An application for a practicing permit shall be in —
| (a) |
Form I.1 for veterinary pharmacy practitioner;
|
| (b) |
Form I.2 for a wholesaler dealer permit; or
|
| (c) |
Form I.3 for manufacturers' permit.
|
|
| (5) |
An applicant under paragraph (1) shall, on approval of the application, by the Council, pay the fee prescribed in the Fifth Schedule.
|
| (6) |
The Council, upon confirming that the applicant has satisfied the conditions of a permit, and upon payment of the fee under paragraph (4) issue an applicant with a practicing permit as set out in Form A set out in the Third Schedule for veterinary pharmacy practices and Form B for wholesale pharmacy practice.
|
| (7) |
The practice permit shall be valid for a period of one year commencing on the 1st of January and ending on the 31st of December of each year.
|
| (8) |
The Council shall by the 31st March of every year publish, in the Kenya Gazette or in a newspaper of wide national circulation, the registered veterinary pharmacy businesses.
|
| (9) |
A person who conducts the business of veterinary pharmacy in premises not registered by the Council under these Regulations commits an offence, and shall be liable, on conviction, to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months or to both.
|
|
| 39. |
Practice permit to be displayed
| (1) |
A person who carries on the business of a veterinary pharmacy shall display his practice permit on a conspicuous place within the premises in which the business is being carried on.
|
| (2) |
A person who contravenes the provisions of this Regulation commits an offence and shall be liable, on conviction, to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months or to both.
|
|
| 40. |
Wholesale permit
A person who intends to trade in veterinary medicines, in bulk , shall make an application to the Council for a wholesale permit and support the application with the following—
| (a) |
a certified copy of the certificate of registration or incorporation of the business name or body corporate and the memorandum and articles of association;
|
| (b) |
provide evidence that the business is under the management of a supervisor who—
| (i) |
is a registered veterinary surgeon permitted to carry out the business of veterinary pharmacy; and |
| (ii) |
is a member of the Board of directors of the body corporate, and who is not acting in a similar capacity for any other body corporate. |
|
|
| 41. |
Retail permit
| (1) |
A person who intends to trade in veterinary medicines as a retailer shall make an application to the Council for a retail permit in Form H set out in the Third Schedule.
|
| (2) |
This regulation shall be applicable to a manufacturer and a wholesaler of veterinary medicines who desires to carry out retail trade.
|
|
PART VI – VETERINARY PHARMACY AND COMPLEMENTARY BUSINESSES
| 42. |
Complementary businesses
| (1) |
A veterinary pharmacy may be conducted alongside complementary businesses including the sale of human medicines, horticultural chemicals, agro-forestry chemicals and fertilizer, animal foodstuffs, seeds and agricultural equipment.
|
| (2) |
The building or premises of a veterinary pharmacy stocking or trading in complementary businesses referred to in paragraph (1), shall be built or adapted in such manner as to provide a separate and distinct partition for the veterinary products from any other products.
|
| (3) |
In addition to the provisions of paragraph (2), the premises shall be built or adapted in such manner as to provide separate and distinct partitions for veterinary medicines and veterinary pesticides such that no mixing of the two classes of products is permissible.
|
|
| 43. |
Stock, supply and distribution of veterinary medicine
| (1) |
A veterinary medicine shall be supplied or distributed only through a wholesale or retail business registered as a veterinary pharmacy business.
|
| (2) |
A holder of wholesale or veterinary pharmacy permit issued by the Council may sell veterinary medicines in bulk to registered retail dealers.
|
| (3) |
A holder of a retail dealer permit, issued by the Council, may sell veterinary medicines to the public.
|
| (4) |
A person shall not supply authorised human medicinal product for administration to an animal other than a product supplied by a veterinary surgeon or in accordance with a written prescription from a veterinary surgeon.
|
| (5) |
A veterinary pharmacy practitioner who dispenses a veterinary medicine belonging to Category I or II to any member of the public without a prescription, commits an offence and shall be liable, on conviction, to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months or to both.
|
|
PART VII – MARKET AUTHORIZATION
| 44. |
Authorization to import or export veterinary medicine
| (1) |
A person who intends to import or export a veterinary medicine shall apply to the Council for a permit in Form K set out in the Third Schedule.
|
| (2) |
The Council shall in determining the application under paragraph (1) consider whether the applicant has met the conditions for importation and exportation in these Regulations, and if so, shall on payment of the prescribed fee, issue the applicant with an import permit in Form L set out in the Third Schedule.
|
| (3) |
A holder of a market authorization may apply to the Council for a wholesale permit to authorize him to import the veterinary medicine specified in the authorization.
|
| (4) |
An authorised wholesale dealer may import a veterinary medicine if —
| (a) |
the authorization covers the veterinary product; and
|
| (b) |
the wholesale dealer has acquired the written consent of the holder of the market authorisation in writing before importation.
|
|
| (5) |
A veterinary surgeon may, with approval of the Council, import an orphan veterinary medicine.
|
|
PART VIII – VETERINARY MEDICINE INSPECTORS
| 45. |
Appointment of veterinary medicine inspectors
The Council shall appoint duly qualified persons on such terms and conditions of service as it may deem appropriate, to serve as veterinary medicine inspectors.
|
| 46. |
Identification of veterinary medicines inspector
| (1) |
The Council shall publish in the Kenya Gazette every inspector appointed under regulation 45 and issue each inspector with an official identity card which shall have a passport size photo of the inspector, duly stamped and signed by the Chief Executive Officer or his authorized agent.
|
| (2) |
Every inspector shall, in conducting inspections under these Regulations, identify himself using the identity card issued under paragraph (1).
|
| (3) |
A person who ceases to be an inspector shall surrender his identify card to the Chief Executive Officer or his authorized agent.
|
|
| 47. |
Powers of veterinary medicines inspectors
| (1) |
An inspector shall have the power, at all reasonable times, to —
| (a) |
enter upon the premises of any manufacturer, distributor or veterinary pharmacy and to inspect any books, papers, records or writings, veterinary medicines, whether patent or otherwise, or any article stored or offered for sale or used in the business; or
|
| (b) |
enter any premises in which he has reasonable cause to suspect that a breach of the law has been or is being committed, and to make such examination and inquiry and to do such other activities, including impounding and seizing suspect veterinary products, closing suspect premises and the taking of samples, as may be necessary for the purpose of ascertaining whether the provisions of these Regulations are being complied with.
|
|
| (2) |
| (a) |
for the purpose of inspecting a veterinary medicine use Form F set out in the Third Schedule; and
|
| (b) |
for any impounding of suspect veterinary medicines, use Form G set out in the Third Schedule.
|
|
| (3) |
An inspector shall observe confidentiality in the findings of his inspection.
|
| (4) |
An inspector shall be liable for any act of negligence he may commit in the performance of his duties.
|
|
PART IX – FINANCIAL PROVISIONS
| 48. |
Funds of the Directorate
| (1) |
The funds of the Directorate shall comprise of —
| (a) |
such monies as may be appropriated by Parliament for the purposes of the Directorate;
|
| (b) |
such monies as may accrue to or vest in the Directorate in the course of the exercise of its functions under these Regulations; and
|
| (c) |
monies from any other source provided for, donated or lent to the Directorate.
|
|
|
| 49. |
Financial year
The financial year of the Directorate shall be the period of twelve months from first July to thirtieth of June.
|
| 50. |
Annual estimates
| (1) |
Three months before the commencement of each financial year, the Council shall cause to be prepared estimates of revenue and expenditure of the Directorate for that year.
|
| (2) |
The annual estimates shall make provision for all the estimated expenditure of the Directorate for the financial year concerned, and in particular shall provide for—
| (a) |
the payment of salaries, allowances and other charges in respect of the staff of the Directorate;
|
| (b) |
the payment of pensions, gratuities and other charges in respect of retirement benefits which are payable out of the funds of the Directorate;
|
| (c) |
the proper maintenance of buildings and grounds of the Directorate;
|
| (d) |
the acquisition, maintenance, repair and replacement of the equipment and other movable property of the Directorate; and
|
| (e) |
the creation of such reserve funds to meet future or contingent liabilities in respect of retirement benefits, insurance or replacement of buildings or equipment or in respect of such other matter as the Directorate may find appropriate.
|
|
| (3) |
The annual estimates shall be approved by the Council before the commencement of the financial year to which they relate and shall be submitted to the Cabinet Secretary for approval.
|
| (4) |
The Directorate shall not increase any sum provided in the estimates without the consent of the Cabinet Secretary.
|
|
| 51. |
Investment of funds
The Council may invest any of the funds of the Directorate in a manner approved by the Treasury for the investment of trust funds.
|
| 52. |
Accounts and Audit
| (1) |
The Council shall cause to be kept all proper books and records of accounts of the income, expenditure, assets and liabilities of the Directorate.
|
| (2) |
The accounts of the Directorate shall be audited in accordance with the Public Finance Management Act, 2012 (No. 18 of 2012).
|
|
PART X – OFFENCES
| 53. |
Offences
A person commits an offence if that person —
| (a) |
imports, exports, manufactures, stores, distributes, sells or otherwise handles a veterinary medicine that has not been registered under these Regulations;
|
| (b) |
imports a veterinary medicine without a permit issued under these Regulations;
|
| (c) |
manufactures, stores, distributes or sells a veterinary medicine in premises which have not been registered under these Regulations;
|
| (d) |
presents for sale or distribution, expired, adulterated, Counterfeit or unlabeled veterinary medicines;
|
| (e) |
sells a veterinary medicine in any form other than the original sealed and labeled package, as registered with the Directorate;
|
| (f) |
sells or distributes a veterinary medicine in any other area other than a registered premises;
|
| (g) |
uses a veterinary medicine for purposes other than that for which it was registered, prescribed or dispensed;
|
| (h) |
supplies human medicinal product for administration to an animal in contravention of these Regulations;
|
| (i) |
is in possession of a Category I or Category II veterinary medicine other than in accordance with these Regulations;
|
| (j) |
advertises a veterinary medicine in contravention of these Regulations;
|
| (k) |
buys a veterinary medicine for resale in bulk while not being in possession of a wholesale dealer permit issued under these Regulations;
|
| (l) |
sells a veterinary medicine to the public while not being in possession of a retail dealer's permit issued in accordance with these Regulations;
|
| (m) |
undertakes research on imported and unregistered veterinary medicine without authorization from the Council;
|
| (n) |
obstructs or fails to assist the Council or veterinary medicine inspector in the performance of their lawful duties under these Regulations;
|
| (o) |
provides the Council or inspector with false or misleading information;
|
| (p) |
operates a veterinary pharmacy after it has been closed by an inspector; or
|
| (q) |
contravenes any provision of these Regulations.
|
|
| 54. |
Officers of corporations
If an institution commits an offence under these Regulations, any officer, director or agent of the corporation who directed, authorized, assented to, acquiesced in, or participated in the commission of the offence shall be a party to and shall be considered to have committed the offence and shall be liable on conviction to the punishment provided for the offence, whether or not the corporation has been prosecuted or convicted.
|
| 55. |
Passing confidential information without authorization
A member of the Council or staff of the Directorate, who —
| (a) |
being in possession of confidential information, however obtained without authorization of the Council —
| (ii) |
attempts, offers or threatens to divulge it, |
|
other than in accordance with these Regulations or other written law; or
| (b) |
willfully obtains or seeks to obtain confidential information to which he is not entitled, commits an offence.
|
|
| 56. |
Penalties
A person who contravenes these Regulations where no Penalties. penalty has been prescribed, shall on conviction be liable to a fine not exceeding one hundred thousand shillings or to imprisonment for a term not exceeding twelve months, or to both.
|
PART XI – GENERAL PROVISIONS
| 57. |
Advertising veterinary standards for medicines
| (1) |
A person who intends to advertise a veterinary medicine, shall make an application to the Council for authority to advertise.
|
| (2) |
The Council may, on payment of the requisite fee and subject to such conditions as the Council or these Regulations may impose, grant the authorization applied for under paragraph (1).
|
| (3) |
An advertisement for a veterinary medicine shall not be misleading or contain any medicinal claim that is not in the summary of the product characteristics registered with the Council.
|
| (4) |
A veterinary medicine listed under Category I and II shall not be advertised unless —
| (a) |
in the case of a veterinary medicine listed under Category I, the advertisement is aimed at veterinary surgeons; and
|
| (b) |
in the case of a veterinary medicine listed under Category II, the advertisement is aimed at veterinary surgeons or suitably qualified persons as recognized by the Council.
|
|
| (5) |
A veterinary medicine listed in Category III and IV may be advertised to general members of the public.
|
|
| 58. |
Exemptions to Regulations
These Regulations do not apply to —
| (a) |
inactivated autogenous vaccine that is manufactured, on the instructions of a veterinary surgeon, from pathogens or antigens obtained from an animal and used for the treatment of that animal or —
| (i) |
other animals on the same site; |
| (ii) |
animals intended to be sent to those premises; or |
| (iii) |
animals on a site that receives animals from those premises; or |
|
| (b) |
animal foodstuff supplements with no therapeutic claim.
|
|
| 59. |
Discretion of Council
The Council shall have the discretion to —
| (a) |
approve the importation and use of any veterinary medicine for handling emergency situations;
|
| (b) |
cancel the registration of any veterinary medicine that is considered to be harmful to animal and human health and the environment; or
|
| (c) |
restrict the use of any specified veterinary medicine.
|
|
| 60. |
Copy of certificate or authorization
| (1) |
A person who has been issued with a certificate or authorization under these Regulations may, if the certificate or authorization is defaced, damaged or lost, on application to the Council and on payment of the prescribed fee, be issued with a copy of the certificate.
|
| (2) |
The copy of a certificate or authorization issued under paragraph (1) shall —
| (i) |
be issued only where the document is in force at the time the application is made; |
| (ii) |
be valid for the same period as the original document; and |
| (iii) |
bear the words "DUPLICATE COPY". |
|
|
| 61. |
Fees
Any fees payable under this Act is as set in the Fifth Schedule.
|
| 62. |
Supercession
These Regulations shall supersede any other Regulations on matters concerning veterinary medicines.
|
| 63. |
Transition and validity of licences and permits
Upon the commencement of these Regulations any certificate in force shall be deemed to have been issued under these Regulations and shall remain in force until its expiry.
|
FIRST SCHEDULE [r. 8(6)]
CONDUCT OF THE AFFAIRS OF THE COUNCIL
| 1. |
Meetings of the Council.
The Council shall meet at least four times in a year and not more than eight times a year, except in case of an emergency, for the transaction of its business, and such meetings shall be held at such places and times and on such days as the Council may determine.
|
| 2. |
Chairperson and vice-charperson
| (1) |
The Council shall at its first meeting elect its vice-chairperson from among the three members appointed under regulation 8(3)(f).
|
| (2) |
The Chairperson shall preside at all meetings of the Council at which he is present, and in case of his absence the vice-chairperson shall preside, but in the absence of both the chairperson and vice-chairperson, members present and constituting a quorum shall elect one among their number to be the chairperson for purposes of the meeting.
|
|
| 3. |
Quorum
The quorum of a meeting of the Council shall be five, at least three of whom shall be veterinary surgeons.
|
| 4. |
Decisions of Council
The decisions of the Council shall be by a simple majority of the votes of the members present, but in the case of an equality of votes, the Chairperson or person presiding shall have a casting vote.
|
| 5. |
Minutes
| (1) |
The Minutes of each meeting shall be kept in the minute book, after they have been confirmed by the Council and signed by the Chairperson at a subsequent meeting of the Council.
|
| (2) |
The deliberations and minutes of meetings of the Council shall be confidential.
|
|
| 6. |
Vacancy not to invalidate proceedings
No proceedings of the Council shall be invalid by reason only of a vacancy among the members thereof.
|
| 7. |
Council to determine own procedure
Subject to this Schedule, the Council may determine its own procedure.
|
| 8. |
Invitation of other persons
The Council may invite any person to participate in its deliberations but a person who has been invited shall have no right to vote.
|
SECOND SCHEDULE [rr. 32(1) & 33(1).]
CONDUCT OF THE AFFAIRS OF THE COUNCIL
PART I – CLASSES OF VETERINARY MEDICINES
| 1. |
List of veterinary pharmaceuticals.
| (iii) |
Analgesics and anti-inflammatories |
| (iv) |
Drugs acting on the nervous system |
| (vi) |
Other systemic therapeutic agents |
| (vii) |
Local therapeutic agents |
|
| 2. |
| (v) |
Probiotics and enzymes. |
|
| 4. |
| (ii) |
Veterinary medicine administration devices. |
| (iii) |
Any other material and equipment of veterinary relevance. |
|
| 5. |
List of alternative medicines.
| (iii) |
Performance enhancers. |
|
| 6. |
| (v) |
Any other pesticides of veterinary relevance |
| (vi) |
And other ecto-parasiticides |
| (vii) |
substances used for euthanasia |
|
PART II – CATEGORIES OF VETERINARY MEDICINES
(a) Prescription Only Medicine—Veterinary surgeon (abbreviated to POM-V);
Category I A POM-V Controlled Veterinary Medicine
| 11. |
Morphine and its derivatives.
|
| 13. |
Methadone (amidone); its salts.
|
| 16. |
Phenoperidine; its salts.
|
| 19. |
Any other veterinary medicine relevant in the category.
|
Category IB — Other POM - V
CATEGORY II —PRESCRIPTION ONLY MEDICINE (POM-VT)
Prescription Only Medicine—Veterinary surgeon
CATEGORY III — Authorized Veterinary Medicines —General Sales List (AVM-GSL)
CATEGORY IV- Alternative Veterinary Medicine — General Sales List (AItVM-GSL)
THIRD SCHEDULE [rr. 23(1) & 38(6).]
FORMS
APPLICATION FOR REGISTRATION OF A VETERINARY MEDICINE
(to be submitted as one original hard-copy and one electronic copy in MS-Word)
The Registrar
Veterinary Medicines Directorate
KABETE









APPLICATION FOR THE REGISTRATION OF VETERINARY PESTICIDES
[to be submitted as one original hard-copy and one electronic copy (in MS-Word)]
The Director
Veterinary Medicines Directorate
KABETE
Information for Applicants
| 1. |
The application form must be completed by a duly authorized person.
|
| 2. |
Every application must be accompanied by 3 copies of the draft label as per VMD requirements.
|
| 3. |
The applicant may be required to submit:-
| (a) |
a sample of the veterinary pesticide;
|
| (b) |
a sample of the technical grade of its active ingredient;
|
| (c) |
a sample of the laboratory standard of its active ingredient;
|
| (d) |
any other sample as may be required by the Council.
|
|
| 4. |
The application must be accompanied by a technical dossier as per VMD data Requirements.
|
| 5. |
An applicant who is not a resident in Kenya shall appoint as an agent a person who is permanently resident in Kenya and duly recognized by the Veterinary Medicines Directorate.











|
SUMMARY OF THE DATA SUBMITTED TO THE VMD FOR REGISTRATION OF A VETERINARY PESTICIDES
FOURTH SCHEDULE [r. 17.]
CODE OF CONDUCT FOR MEMBERS OF AND EMPLOYEES OF THE DIRECTORATE
| 1. |
Impartiality and independence of members
| (1) |
Every member and employee of the Directorate shall impartially and independently perform the functions of a member in good faith and without fear, favour or prejudice, and without influence from—
| (a) |
the National or County Government;
|
| (d) |
any candidate participating in an election; or
|
| (e) |
any other person or authority.
|
|
|
| 2. |
Independence from political or public office
| (1) |
A member or employee of the Directorate shall not, during tenure of office, be eligible for—
| (a) |
appointment or nomination to a political office; or
|
| (b) |
appointment to another public office.
|
|
| (2) |
A member of the Directorate may not—
| (a) |
by their membership, association, statement, conduct or in any other manner place in jeopardy the perceived independence of the member, or in any other manner harm the credibility, impartiality, independence or integrity of the Directorate;
|
| (b) |
make private use of or profit from any confidential information gained as a result of being a member of the Directorate; or
|
| (c) |
divulge any information to any third party, save in the of official duty.
|
|
|
| 3. |
Disclosure of conflicting interests
| (1) |
If a member or an employee is directly or indirectly interested in any contract, proposed contract or other matter before the Council and is present at any meeting of the Council at which the contract, proposed contract or other matter is the subject of consideration, the member or employee shall, at the meeting and as soon as practicable after the commencement thereof, disclose the fact and shall not take part in the consideration or discussion of, or vote on, any questions with respect to the contract or other matter or be counted in the quorum of the meeting during consideration of the matter.
|
| (2) |
A member or employee whose personal interest conflicts with their official duties shall—
| (a) |
declare the personal interests to their supervisor or other appropriate person or body in writing and comply with any directions given to avoid the conflict; and
|
| (b) |
refrain from participating in any deliberations with respect to the matter.
|
|
| (3) |
No member of staff of the Directorate shall transact business with the Directorate directly or indirectly.
|
|
| 4. |
Professionalism
A member or employee of the Directorate shall—
| (a) |
perform their duties in a manner that promotes and maintains public confidence in the Directorate;
|
| (b) |
treat the public and colleagues with courtesy and respect;
|
| (c) |
discharge all their duties in a professional, timely and efficient manner and in line with the rule of law; and
|
| (d) |
respect the rights and freedom of all persons that he may interact with.
|
|
| 5. |
Improper enrichment
A member or employee of the Directorate shall not—
| (a) |
use their office or organization to improperly enrich themselves or others;
|
| (b) |
accept or request gifts or personal favours from any person who may have a commercial interest with the Directorate or any other interest that may be affected by the normal business of the Service; or
|
| (c) |
use information that is acquired during the course of their duties or connected to their duties for their benefit or for the benefit of others
|
|
| 6. |
Integrity in private affairs
A member or employee shall conduct their private affairs in manner that maintains public confidence in the integrity of their office and the Directorate as a whole and shall—
| (b) |
not neglect their financial obligations;
|
| (c) |
submit an annual declaration of their income, assets and liabilities to the Commission responsible for such declarations from public officers;
|
| (d) |
not engage in political activity that may compromise or be seen to compromise the neutrality of their office, or the Directorate; and
|
| (e) |
not preside over or play a central role in the organization of a fundraising activity.
|
|
| 7. |
Sexual harassment
| (1) |
A member or employee shall not sexually harass a member of the public or colleague.
|
| (2) |
Sexual harassment includes —
| (a) |
making a request or exerting pressure for sexual activity or favours;
|
| (b) |
making intentional or careless physical contact that is sexual in nature; or
|
| (c) |
making gestures, jokes or comments, including innuendoes regarding another person's sexuality.
|
|
|
| 8. |
Nepotism
A member or employee shall not practice favouritism on the grounds of tribe, race, kin, culture, sex or acquaintance or otherwise in performance of their duties.
|
| 9. |
Application of the Public Officers Ethics Act
This Code is in addition to the provisions of the Public Officers Ethics Act and where there is a conflict between the Code and these Regulations, the provisions of the Act shall prevail.
|
| 10. |
Breach of code
Any breach of the Code by a member or officer of the Service shall be treated as gross misconduct.

|
|